Advanced Manufacturing of High-Purity Glimepiride for Global Pharmaceutical Supply Chains
Advanced Manufacturing of High-Purity Glimepiride for Global Pharmaceutical Supply Chains
The global demand for effective antidiabetic medications continues to surge, placing immense pressure on the supply chains of critical active pharmaceutical ingredients (APIs) like Glimepiride. As a third-generation sulfonylurea, Glimepiride represents a cornerstone therapy for Type II diabetes, yet its manufacturing has historically been plagued by challenges related to purity, crystal form control, and the removal of persistent structural impurities. The technical disclosure found in patent CN110885306A presents a transformative approach to these longstanding issues, offering a robust pathway to achieve pharmaceutical-grade purity exceeding 99.9%. This innovation is not merely a laboratory curiosity but a commercially viable strategy that addresses the critical pain points of R&D directors and procurement specialists alike. By re-engineering the solvent systems used in both the intermediate purification and the final coupling stages, this method effectively mitigates the risk of degradation products and ensures the consistent production of the therapeutically superior Form II crystal lattice. For international pharmaceutical manufacturers, adopting such advanced synthetic methodologies is essential for maintaining regulatory compliance and securing a reliable supply of high-quality medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
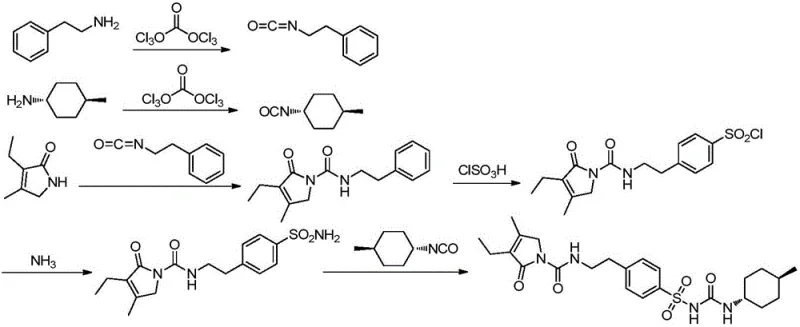
Traditional synthetic routes for Glimepiride, often derived from early Hoechst processes, typically rely heavily on acetone as the primary reaction solvent for the final urea coupling step. While acetone offers the advantage of minimizing certain side reactions, it suffers from significant thermodynamic limitations regarding the solubility of the key starting material, 1-[4-[2-(3-ethyl-4-methyl-2-oxo-3-pyrroline-1-carboxamido)-ethyl]-benzenesulfonamide. This poor solubility leads to incomplete conversion of the starting sulfonamide, resulting in substantial residual amounts of this intermediate remaining in the crude product. This is a critical quality failure because the unreacted sulfonamide is not only a difficult-to-remove impurity due to its similar crystal properties to the final API but also acts as a primary degradation product that can accumulate over the shelf-life of the drug. Furthermore, conventional purification strategies often require multiple, yield-depleting recrystallization steps to attempt to remove ortho- and meta-sulfonylated isomers, drastically reducing the overall economic efficiency of the manufacturing process and complicating waste stream management.
The Novel Approach
The methodology disclosed in CN110885306A fundamentally alters the reaction landscape by introducing a sophisticated mixed solvent system comprising acetone and 2-butanone. This strategic substitution leverages the superior solvation capabilities of 2-butanone to ensure that the starting sulfonamide intermediate remains fully dissolved and reactive throughout the coupling process. By driving the reaction to near-total completion, the process inherently minimizes the presence of the problematic starting material in the crude mixture, thereby simplifying downstream purification. Additionally, the patent details a highly efficient purification protocol for the sulfonamide intermediate itself, utilizing a 2-butanone and water mixture that selectively crystallizes the desired product while leaving isomeric impurities in the mother liquor. This dual-stage optimization—first in the intermediate prep and then in the final coupling—creates a synergistic effect that elevates the purity profile of the final API to unprecedented levels without sacrificing yield.
Mechanistic Insights into Solvent-Engineered Crystallization and Coupling
The core mechanistic advantage of this novel process lies in the precise manipulation of solubility parameters to control reaction kinetics and crystal nucleation. In the synthesis of the critical sulfonamide intermediate, the use of a 2-butanone and water system creates a solvent environment where the target molecule has a steep solubility curve relative to temperature. Upon cooling, the desired compound precipitates rapidly and selectively, while structurally similar impurities, such as ortho- and meta-isomers, remain solvated due to their different polarity profiles. This phenomenon effectively acts as a molecular filter, removing impurities at the source rather than attempting to separate them from the final API. When transitioning to the final urea formation step, the introduction of 2-butanone alongside acetone modifies the dielectric constant of the reaction medium. This change enhances the nucleophilic attack of the sulfonamide nitrogen on the isocyanate carbon, facilitating a smoother transition state and reducing the activation energy required for the coupling. Consequently, the reaction proceeds more vigorously at reflux temperatures, ensuring that the equilibrium shifts decisively towards the product side.
Furthermore, the control of the crystal polymorph is a critical aspect of this mechanism. Glimepiride exists in multiple crystalline forms, with Form II being the preferred polymorph for pharmaceutical formulation due to its enhanced dissolution rate and bioavailability. The specific thermal cycling and solvent composition described in the patent—particularly the final slurry in dichloromethane at controlled temperatures between 15°C and 20°C—provide the necessary thermodynamic conditions to stabilize the Form II lattice. This prevents the spontaneous conversion to the less soluble Form I, which could compromise the drug's efficacy. The rigorous control over these physical parameters ensures that every batch produced meets the stringent specifications required by global regulatory bodies, eliminating the variability that often plagues generic API manufacturing.
How to Synthesize High-Purity Glimepiride Efficiently
The implementation of this advanced synthesis route requires strict adherence to the optimized solvent ratios and thermal profiles established in the patent data. The process begins with the meticulous preparation of the sulfonamide intermediate, where the 2-butanone/water crystallization must be performed with precision to maximize isomer rejection. Following this, the final coupling reaction demands a nitrogen-inert atmosphere to prevent moisture interference with the isocyanate reagent. The detailed operational parameters, including specific reflux times and cooling rates, are critical for reproducibility. For a comprehensive breakdown of the exact reagent quantities, temperature ramping schedules, and workup procedures required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Synthesize the key sulfonamide intermediate via chlorosulfonation and subsequent amination, utilizing a 2-butanone/water system for high-efficiency purification to remove isomers.
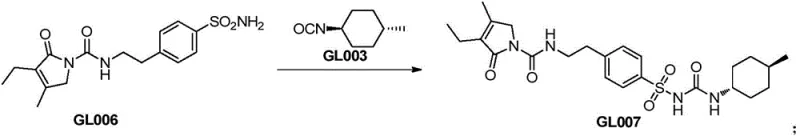
- Prepare the final Glimepiride API by reacting the purified sulfonamide with trans-4-methylcyclohexyl isocyanate in a mixed acetone/2-butanone solvent system.
- Execute a controlled crystallization and slurry process using dichloromethane to ensure the formation of the stable Form II crystal lattice with >99.9% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refined manufacturing process translates directly into tangible operational benefits and risk mitigation. The primary value proposition lies in the drastic simplification of the purification workflow. By achieving higher crude purity through solvent engineering, the need for extensive, yield-killing recrystallization cycles is significantly reduced. This streamlining of the production process leads to a substantial reduction in manufacturing costs, as less solvent is consumed, less energy is expended on heating and cooling cycles, and labor hours are optimized. Moreover, the increased overall yield means that more saleable product is generated from the same amount of raw materials, effectively lowering the cost of goods sold (COGS) and improving margin potential for the final drug product.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic separations and the reduction in recrystallization steps result in significant cost savings. By utilizing common, industrially available solvents like 2-butanone and acetone, the process avoids the expense of exotic reagents. The higher yield directly correlates to better raw material utilization, ensuring that the production of this high-purity API remains economically competitive in the global market.
- Enhanced Supply Chain Reliability: A robust manufacturing process that is less sensitive to minor variations in reaction conditions ensures consistent batch-to-batch quality. This reliability is crucial for maintaining uninterrupted supply to downstream formulation partners. The ability to consistently produce the correct crystal form (Form II) reduces the risk of batch rejection due to polymorphic instability, thereby securing the continuity of supply for critical diabetes medications.
- Scalability and Environmental Compliance: The solvent systems employed are well-suited for large-scale industrial reactors, facilitating easy scale-up from pilot plants to commercial metric-ton production. Furthermore, the reduction in solvent usage and waste generation aligns with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals without compromising on product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Glimepiride using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this process outperforms legacy methods in terms of purity and efficiency.
Q: How does the new solvent system improve Glimepiride purity compared to conventional acetone methods?
A: The novel mixed solvent system (acetone/2-butanone) offers superior solvation properties for the starting sulfonamide intermediate compared to pure acetone. This ensures complete consumption of the starting material during the coupling reaction, preventing residual intermediate carryover which is a major degradation product and difficult-to-remove impurity in the final API.
Q: What specific crystal form of Glimepiride is produced by this method?
A: This manufacturing process specifically yields Glimepiride Crystal Form II. This polymorph is highly desirable in pharmaceutical formulations due to its superior dissolution rate and bioavailability characteristics compared to Form I, ensuring consistent therapeutic efficacy.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. By optimizing the crystallization steps to avoid complex chromatographic purifications and utilizing robust solvent systems like 2-butanone and water, the method minimizes processing time and equipment constraints, making it ideal for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glimepiride Supplier
At NINGBO INNO PHARMCHEM, we understand that the integrity of your final pharmaceutical product depends entirely on the quality of its ingredients. Our technical team has extensively analyzed advanced synthesis routes like the one described in CN110885306A, and we possess the infrastructure to implement these high-purity protocols at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical assessment of crystal forms and isomeric impurities, guaranteeing that every gram of Glimepiride we supply meets the highest global pharmacopeial standards.
We invite you to collaborate with us to optimize your supply chain for antidiabetic APIs. By leveraging our technical expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain security.
