Advanced Glimepiride Manufacturing: Eliminating Chloroform for Scalable API Production
Advanced Glimepiride Manufacturing: Eliminating Chloroform for Scalable API Production
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with environmental sustainability, particularly for high-volume antidiabetic agents like Glimepiride. Patent CN101486674B introduces a refined preparation method that addresses critical bottlenecks in the traditional manufacturing of this sulfonylurea derivative. By leveraging a direct condensation strategy between a mature sulfonamide intermediate and an isocyanate derivative, this technology circumvents the need for hazardous chlorosulfonic acid sulfonation steps found in legacy processes. The innovation lies not only in the chemical transformation but also in the downstream processing, which substitutes toxic chloroform with safer solvent systems like acetone and water. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for a reliable glimepiride supplier, ensuring that the final active pharmaceutical ingredient meets stringent regulatory standards for residual solvents while maintaining economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Glimepiride has relied on routes documented in literature such as Drugs of the Future (1992), which involve multiple high-risk chemical transformations. A primary drawback of these conventional methods is the utilization of chlorosulfonic acid for the sulfonation of the phenethyl precursor, a reagent that generates substantial corrosive waste and requires specialized handling equipment to manage exothermic risks. Furthermore, the final purification stages in these legacy processes typically necessitate recrystallization using chloroform, a solvent classified as a probable human carcinogen with strict limits on residual levels in drug substances. The difficulty in completely removing chloroform from the crystal lattice often leads to batch failures or requires extensive additional drying cycles, inflating production costs and extending lead times. Additionally, the multi-step nature of building the sulfonylurea core from basic starting materials increases the cumulative yield loss and complicates the impurity profile, making it challenging to achieve the high-purity glimepiride required for modern generic and branded formulations without expensive chromatographic interventions.
The Novel Approach
In stark contrast, the methodology outlined in CN101486674B streamlines the synthesis by utilizing pre-functionalized intermediates, specifically Compound A (a sulfonamide) and Compound B (trans-4-methyl-cyclohexyl isocyanate), effectively bypassing the dangerous sulfonation step entirely. This approach focuses on the final urea bond formation as the key value-add step, conducted in acetone with potassium carbonate acting as a promoter. The elimination of chlorosulfonic acid removes a major safety hazard and reduces the burden on waste treatment facilities, directly contributing to cost reduction in API manufacturing. Moreover, the purification protocol ingeniously exploits the solubility differences of the product salt; by dissolving the crude reaction mixture in hot water and filtering out insoluble impurities, the process achieves a high degree of purification before the final acidification. This water-based workup significantly lowers the reliance on volatile organic compounds during the bulk purification phase, aligning with green chemistry principles and simplifying the solvent recovery infrastructure needed for commercial scale-up of complex sulfonylureas.
Mechanistic Insights into Potassium Carbonate-Promoted Urea Formation
The core chemical transformation in this patent is the nucleophilic addition of the sulfonamide nitrogen to the electrophilic carbon of the isocyanate group, a reaction that is thermodynamically favorable but kinetically slow without activation. Potassium carbonate serves a dual purpose in this mechanism: firstly, it acts as a base to deprotonate the slightly acidic sulfonamide proton of Compound A, generating a more nucleophilic sulfonamidate anion in situ. This anionic species attacks the electron-deficient carbon of the isocyanate in Compound B, leading to the formation of the urea linkage that characterizes the Glimepiride structure. The use of a mild inorganic base like potassium carbonate, rather than strong organic bases or alkali metals, ensures that the sensitive pyrrolinone ring and the sulfonyl group remain intact, minimizing degradation pathways that could generate difficult-to-remove impurities. The reaction is performed under anhydrous conditions in acetone to prevent the hydrolysis of the isocyanate, which would otherwise consume the reagent and form urea byproducts, thereby preserving the atom economy of the process.
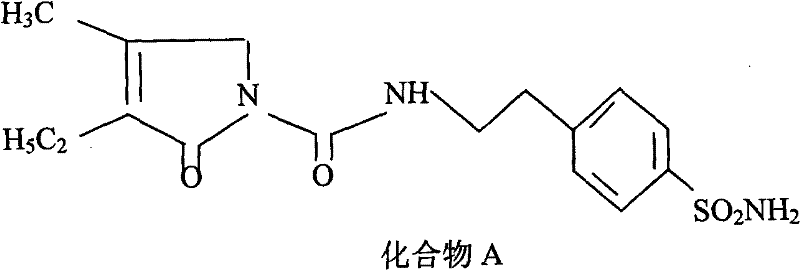
Following the condensation, the mechanistic advantage extends to the isolation strategy, where the resulting Glimepiride exists initially as a potassium salt or a mixture with the base, which exhibits distinct solubility properties compared to the neutral molecule. By adding pure water, the potassium salt dissolves readily, allowing for the physical separation of unreacted organic starting materials or non-polar side products via filtration. The subsequent addition of activated carbon during this aqueous phase targets colored impurities and trace organic contaminants through adsorption, a step that is far more efficient in water than in organic solvents due to the polarity match. Finally, acidification with dilute hydrochloric acid reprotonates the sulfonamide nitrogen, causing the neutral Glimepiride to precipitate out of the aqueous solution in a highly pure crystalline form. This sequence demonstrates a sophisticated understanding of physical organic chemistry, leveraging pKa differences and solubility switches to achieve purification without the need for energy-intensive distillation or hazardous solvent exchanges.
How to Synthesize Glimepiride Efficiently
The operational simplicity of this patented route makes it highly attractive for technology transfer and rapid implementation in existing multipurpose reactors. The process begins with the suspension of Compound A and potassium carbonate in acetone, followed by heating to promote the initial activation of the nucleophile. Once the temperature stabilizes between 50°C and 60°C, Compound B is introduced slowly to control the exotherm and ensure complete conversion to the urea derivative. After the reaction reaches completion, indicated by the consumption of the isocyanate, the mixture is cooled and filtered to separate the bulk inorganic salts. The real innovation occurs in the aqueous workup, where the filter cake is dissolved in heated water, treated with activated carbon, and filtered again to yield a clear solution ready for precipitation.
- Condense Compound A and Compound B in acetone with potassium carbonate at 50-60°C to form the crude urea linkage.
- Filter the reaction mixture and dissolve the crude product in heated pure water, treating with activated carbon to remove impurities.
- Acidify the aqueous solution with dilute hydrochloric acid at low temperature to precipitate the crude Glimepiride solid.
- Recrystallize the crude solid in acetone at 50-70°C, followed by hot filtration and vacuum drying to obtain the pure final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the solvent inventory; by replacing chloroform and chlorosulfonic acid with acetone, water, and dilute hydrochloric acid, facilities can reduce their exposure to highly regulated hazardous materials. This shift not only lowers the cost of compliance and waste disposal but also mitigates the risk of supply disruptions associated with the sourcing of restricted reagents. Furthermore, the use of water as a primary purification solvent represents a significant cost reduction in API manufacturing, as water is exponentially cheaper and more abundant than high-grade organic solvents, and its removal via drying requires less energy than stripping high-boiling organics. The robustness of the process, which tolerates standard industrial equipment without requiring glass-lined reactors specifically for corrosive acid handling, enhances the overall asset utilization rate and flexibility of the production line.
- Cost Reduction in Manufacturing: The elimination of chloroform from the recrystallization step removes the need for expensive solvent recovery systems designed for halogenated hydrocarbons, which often require specialized metallurgy and rigorous emission controls. Additionally, the high efficiency of the aqueous purification step reduces the volume of organic mother liquor that needs to be processed, leading to substantial savings in waste treatment fees. The use of equimolar amounts of reactants minimizes raw material waste, and the mild reaction conditions reduce energy consumption for heating and cooling cycles. By avoiding the multi-step synthesis from basic precursors and starting from mature intermediates, the overall processing time is shortened, allowing for more batches to be produced per year within the same facility footprint, thereby driving down the fixed cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved because the key starting materials, Compound A and Compound B, are stable, commercially available intermediates that do not suffer from the volatility of supply often seen with reactive acid chlorides or isocyanates stored over long periods. The process does not rely on single-source specialty catalysts or rare metals, ensuring that production can continue even during global shortages of specific fine chemicals. The simplified workflow reduces the number of unit operations, which in turn decreases the probability of mechanical failure or operator error causing a batch loss. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing suppliers to respond more quickly to market demand fluctuations without compromising on quality or delivery schedules.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with increasingly strict global regulations regarding residual solvents in drug substances, particularly Class 2 solvents like chloroform. The switch to acetone and water ensures that the final product easily passes ICH Q3C guidelines without extensive secondary processing, facilitating faster regulatory approval and market entry. The aqueous waste streams generated are easier to treat biologically compared to halogenated organic waste, reducing the environmental footprint of the manufacturing site. This green profile enhances the brand reputation of the manufacturer and future-proofs the supply chain against tightening environmental legislation, ensuring long-term operational continuity and social license to operate in key pharmaceutical manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Glimepiride synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the process capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing production portfolios or sourcing strategies.
Q: How does this patent method improve solvent safety compared to traditional Glimepiride synthesis?
A: Traditional methods described in prior art often utilize chlorosulfonic acid for sulfonation and chloroform for recrystallization, leading to significant toxic solvent residues. This patented process eliminates the use of chloroform entirely, replacing it with acetone and water for purification, thereby drastically reducing hazardous waste and ensuring lower residual solvent levels in the final API.
Q: What is the role of potassium carbonate in the condensation reaction?
A: Potassium carbonate acts as a mild inorganic base that facilitates the nucleophilic attack of the sulfonamide nitrogen on the isocyanate carbon. It helps deprotonate the sulfonamide group in Compound A, increasing its nucleophilicity without the harshness of stronger bases, which minimizes side reactions and simplifies the downstream neutralization and filtration steps.
Q: Why is the aqueous dissolution step critical for purity?
A: The unique solubility profile of the Glimepiride potassium salt allows it to dissolve in hot water while many organic impurities remain insoluble or are adsorbed by activated carbon. This aqueous workup serves as a highly effective purification stage before acidification, removing inorganic salts and organic byproducts more efficiently than traditional organic solvent extractions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glimepiride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for maintaining competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101486674B are fully realized in practical, large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities and residual solvents, guaranteeing that every batch of Glimepiride meets the highest international pharmacopoeia standards. Our commitment to technical excellence allows us to navigate the complexities of urea condensation chemistry and aqueous workups with precision, delivering consistent quality that R&D directors can trust for their formulation development.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages specific to your supply chain context. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with a solid foundation of technical and commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
