Industrial Scale-Up of Isoxazolidinedione Compounds for Diabetes Therapeutics
The pharmaceutical industry constantly seeks robust manufacturing routes for critical therapeutic intermediates, particularly for metabolic disorders like diabetes. Patent CN1130361C, filed in late 2003, presents a groundbreaking methodology for the preparation of isoxazolidinedione compounds, specifically designated as formula [11]. These compounds serve as vital precursors in the development of antidiabetic agents. The innovation lies not merely in the creation of the molecule but in the radical optimization of the synthetic pathway to address long-standing industrial pain points regarding safety, environmental compliance, and overall yield efficiency. By transitioning away from hazardous reagents and restricted solvents, this technology offers a blueprint for sustainable chemical manufacturing that aligns with modern regulatory standards.
![General structure of isoxazolidinedione compound [11] used as diabetes therapeutic](/insights/img/isoxazolidinedione-intermediate-synthesis-pharma-supplier-20260308094907-01.webp)
Traditionally, the synthesis of such complex heterocyclic structures relied on methodologies that, while effective on a laboratory bench, proved problematic upon scale-up. The prior art, specifically referenced in international publication WO 95/18125 and the Journal of Medicinal Chemistry (1992), utilized a sequence known as Method A for the early stages and Method B for the later transformations. These conventional approaches were plagued by the use of dichloromethane, a solvent increasingly restricted due to environmental regulations, and highly reactive, dangerous reagents like lithium aluminum hydride and phosphorus oxychloride. The reliance on these materials created significant bottlenecks in supply chain logistics and imposed heavy costs on waste treatment and safety protocols, rendering the older processes economically unviable for large-scale production.
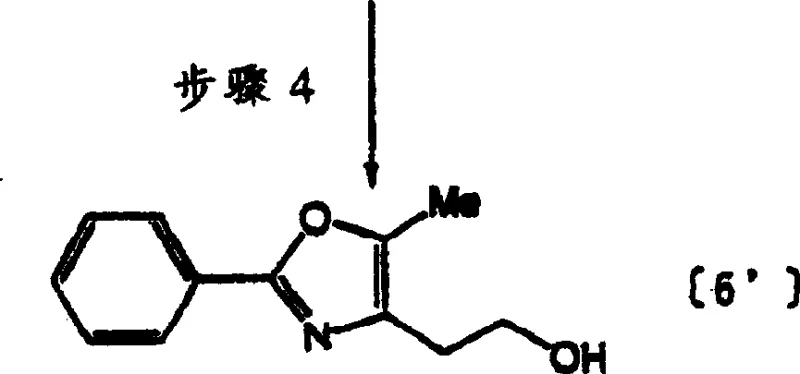
The novel approach detailed in this patent fundamentally re-engineers the synthetic logic to prioritize industrial practicality without sacrificing chemical integrity. In the initial acylation step, the process substitutes the traditional dichloromethane and triethylamine system with a safe, inexpensive water-based solvent and inorganic bases such as potassium or sodium carbonate. This seemingly simple switch results in a dramatic yield improvement, reaching 92-97%, which is a substantial increase of over 10% compared to legacy methods. Furthermore, the cyclization steps avoid the use of corrosive phosphorus oxychloride, opting instead for p-toluenesulfonic acid monohydrate, which facilitates a cleaner reaction profile and eliminates the need for complex post-treatment separations that often lead to product decomposition.
![New optimized synthetic route for isoxazolidinedione compound [11]](/insights/img/isoxazolidinedione-intermediate-synthesis-pharma-supplier-20260308094907-06.webp)
Mechanistically, the most significant breakthrough occurs in the reduction and coupling phases of the synthesis. Conventional wisdom dictated the use of lithium aluminum hydride in diethyl ether for reducing the oxazolyl acetate derivative, a combination notorious for its high flammability and explosion risk in bulk quantities. The patented method ingeniously employs sodium borohydride in tetrahydrofuran with methanol acting as a reduction accelerator. This modification not only mitigates safety hazards but also boosts the reduction yield to an impressive 85-95%. Additionally, the strategy for constructing the final side chain avoids the instability associated with intermediate aldehydes by pre-synthesizing a stable malonate derivative. This allows for a direct coupling reaction that achieves yields of 80-85%, bypassing the low-efficiency 65% yield bottleneck found in previous techniques.
How to Synthesize Isoxazolidinedione Compound Efficiently
The synthesis of this high-value pharmaceutical intermediate requires precise control over reaction conditions to maximize the benefits of the patented improvements. The process begins with the aqueous acylation of L-aspartic acid beta-methyl ester, followed by a telescoped cyclization and decarboxylation sequence in toluene. The subsequent reduction and functionalization steps utilize optimized reagent systems to ensure high purity and throughput. For process chemists looking to implement this route, attention to the specific solvent switches and reagent stoichiometries described in the patent is critical for replicating the reported success rates. The detailed standardized synthesis steps are provided in the guide below.
- React L-aspartic acid beta-methyl ester with an acyl chloride in water using an inorganic base to form the amide derivative.
- Perform cyclization and decarboxylation using acetic anhydride and p-toluenesulfonic acid in toluene to generate the oxazole ring.
- Reduce the ester to an alcohol using sodium borohydride in THF/methanol, followed by mesylation and coupling with a malonate derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into tangible operational efficiencies and risk mitigation. The shift away from regulated solvents like dichloromethane removes a major compliance burden, simplifying the permitting process for manufacturing facilities and reducing the overhead associated with hazardous waste disposal. By utilizing common, bulk-available reagents such as sodium borohydride and inorganic carbonates, the dependency on specialized, high-cost catalysts is eliminated, leading to a more stable and predictable raw material supply chain that is less susceptible to market volatility.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like lithium aluminum hydride and phosphorus oxychloride significantly lowers the raw material costs per kilogram of finished product. Furthermore, the substantial improvements in yield at every stage—from the initial acylation to the final cyclization—mean that less starting material is required to produce the same amount of active intermediate, drastically reducing the cost of goods sold and improving overall margin potential for the final drug product.
- Enhanced Supply Chain Reliability: By replacing restricted solvents with industrially friendly alternatives like toluene and water, the manufacturing process becomes more resilient to regulatory changes and environmental crackdowns. The use of stable intermediates, such as the pre-formed malonate derivative, prevents production delays caused by the degradation of unstable aldehyde precursors, ensuring a consistent and reliable flow of materials to downstream formulation teams.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, avoiding exothermic risks associated with traditional hydride reductions and corrosive chlorinating agents. This inherent safety profile allows for larger batch sizes and continuous processing opportunities, while the reduced generation of toxic by-products aligns with green chemistry principles, facilitating easier approval from environmental health and safety departments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isoxazolidinedione synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of adopting this novel manufacturing route over established prior art methods.
Q: What are the key safety improvements in this synthesis compared to prior art?
A: The process replaces hazardous reagents like lithium aluminum hydride (LiAlH4) and phosphorus oxychloride (POCl3) with safer alternatives such as sodium borohydride and p-toluenesulfonic acid, significantly reducing industrial risk.
Q: How does the new method improve yield in the initial acylation step?
A: By switching from dichloromethane/triethylamine to a water-based solvent system with inorganic bases like sodium carbonate, the yield increases to 92-97%, surpassing previous methods by over 10%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the elimination of restricted solvents like dichloromethane in favor of toluene and water, along with higher yields and simplified workups, makes it highly scalable and compliant with environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoxazolidinedione Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical yields and safety benefits of this patent are fully realized in your supply chain. Our rigorous QC labs and stringent purity specifications guarantee that every batch of isoxazolidinedione intermediate meets the exacting standards required for diabetes therapeutic applications.
We invite you to leverage our technical capabilities to optimize your sourcing strategy for this critical intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing excellence can drive value and security for your pharmaceutical pipeline.
