Advanced D,L-Methionine Manufacturing: Leveraging Patent CN103641758A for Commercial Scale-Up
Advanced D,L-Methionine Manufacturing: Leveraging Patent CN103641758A for Commercial Scale-Up
The global demand for essential amino acids continues to surge, driven by the expanding pharmaceutical, food, and animal nutrition sectors. At the forefront of this chemical evolution is the innovative preparation method for high-purity D,L-methionine detailed in patent CN103641758A. This technology represents a paradigm shift from traditional hydantoin-based synthesis routes, offering a streamlined pathway that utilizes hydrogen cyanide mixed gas prepared via the Andrussow method. By reacting this gas directly with methionaldehyde under alkali catalysis, the process yields a stable 2-hydroxy-4-methylthiobutyronitrile system, which is subsequently converted to 2-amino-4-methylthiobutyronitrile and finally hydrolyzed to the target amino acid. 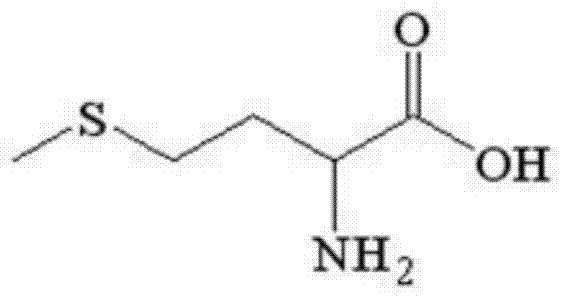 This structural integrity is paramount for its function as a methyl donor and protein building block. The patent underscores a methodology that not only achieves exceptional yields exceeding 97% but also ensures a product purity of 99% with superior bulk density characteristics. For industry stakeholders, this translates to a robust, scalable solution that addresses the chronic supply-demand imbalance in the domestic market, reducing reliance on imports while establishing a new benchmark for cost-effective amino acid manufacturing.
This structural integrity is paramount for its function as a methyl donor and protein building block. The patent underscores a methodology that not only achieves exceptional yields exceeding 97% but also ensures a product purity of 99% with superior bulk density characteristics. For industry stakeholders, this translates to a robust, scalable solution that addresses the chronic supply-demand imbalance in the domestic market, reducing reliance on imports while establishing a new benchmark for cost-effective amino acid manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of D,L-methionine has been dominated by the hydantoin route, as evidenced by numerous patents such as CN1079095C and CN102633699A. While these methods are established, they suffer from inherent inefficiencies that impact both economic viability and environmental compliance. A primary drawback is the generation of substantial volumes of wastewater coupled with low-value byproducts like sodium sulfate, which complicates waste treatment and offers minimal economic recovery. Furthermore, the hydantoin hydrolysis step inevitably leads to the formation of methionine dimers or polymers, which detrimentally affects the overall yield and necessitates complex purification protocols to meet stringent pharmaceutical grade specifications. Additionally, conventional processes often require rigorous rectification and purification of raw materials like 3-methylthiopropionaldehyde and cyanide sources, adding significant operational complexity and energy consumption to the manufacturing workflow.
The Novel Approach
In stark contrast, the methodology disclosed in CN103641758A introduces a disruptive nitrile-based synthesis strategy that circumvents the bottlenecks of the hydantoin pathway. By employing hydrogen cyanide mixed gas derived directly from the mature Andrussow process, the invention eliminates the need for isolating pure liquid HCN, thereby enhancing safety and reducing handling costs. A critical innovation lies in the tolerance for unpurified methionaldehyde, which contains light and heavy components yet reacts efficiently without prior distillation. This approach not only simplifies the operational workflow but also drastically reduces the energy footprint associated with raw material refinement. The resulting intermediate, 2-hydroxy-4-methylthiobutyronitrile, exhibits remarkable stability, allowing for flexible production scheduling and inventory management, while the final hydrolysis step yields high-purity D,L-methionine alongside valuable ammonium sulfate, transforming a waste stream into a revenue-generating agricultural product.
Mechanistic Insights into Andrussow-Based Nitrile Synthesis
The core of this technological advancement rests on a sophisticated sequence of nucleophilic additions and substitutions governed by precise thermodynamic controls. The process initiates with the Andrussow reaction, where methane, ammonia, and oxygen interact over a platinum-rhodium catalyst at temperatures exceeding 1000°C to generate a hydrogen cyanide mixed gas. This gas mixture, after undergoing deamination treatment to remove excess ammonia and water vapor using sulfuric acid, serves as the electrophilic source for the subsequent cyanohydrin formation. In the presence of an organic or inorganic base catalyst, such as pyridine or triethylamine, the cyanide ion attacks the carbonyl carbon of methionaldehyde. This nucleophilic addition occurs optimally at temperatures between 40°C and 45°C and pressures ranging from 0.1 to 0.15 MPa, ensuring a conversion rate of methionaldehyde approaching 100% while maintaining the stability of the resulting 2-hydroxy-4-methylthiobutyronitrile system.
Following the formation of the cyanohydrin intermediate, the synthesis proceeds through a high-pressure ammoniation step where the hydroxyl group is substituted by an amino group. This transformation is conducted in a stainless steel high-pressure reactor using aqueous ammonia at concentrations between 40% and 65%, under pressures of 1 to 3 MPa and temperatures of 40°C to 85°C. The mechanistic efficiency here is critical; the reaction conditions are tuned to maximize the formation of 2-amino-4-methylthiobutyronitrile while minimizing side reactions. Subsequent acid hydrolysis, preferably using sulfuric acid in a two-stage temperature profile, cleaves the nitrile group to the carboxylic acid. The first stage at 45°C to 55°C forms the amide sulfate, while the second stage at 90°C to 110°C completes the hydrolysis to the amino acid. This controlled degradation ensures minimal racemization and high optical purity retention, crucial for downstream applications.
How to Synthesize D,L-Methionine Efficiently
Implementing this synthesis route requires strict adherence to the reaction parameters defined in the patent to ensure reproducibility and safety at scale. The process integrates continuous gas flow technologies with batch reactor systems to optimize throughput. Operators must carefully monitor the composition of the hydrogen cyanide mixed gas, ensuring the removal of ammonia prior to the addition reaction to prevent catalyst poisoning. The stability of the intermediate system allows for decoupling the cyanohydrin formation from the ammoniation step, providing logistical flexibility in plant operations. Detailed standard operating procedures regarding pressure relief, temperature ramping, and pH adjustment during the crystallization phase are essential for achieving the reported bulk density of 0.70 g/mL.
- Generate hydrogen cyanide mixed gas via the Andrussow method, treat with sulfuric acid to remove ammonia, and react with unpurified methional under alkali catalysis to form 2-hydroxy-4-methylthiobutyronitrile.
- React the 2-hydroxy-4-methylthiobutyronitrile system with ammonia under heating (40-85°C) and pressure (1-3MPa) to substitute the hydroxyl group, yielding 2-amino-4-methylthiobutyronitrile.
- Hydrolyze the aminonitrile with inorganic acid (preferably sulfuric acid) in a two-stage temperature process, neutralize, decolorize, and crystallize to obtain high-purity D,L-methionine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers profound strategic advantages that extend beyond simple unit cost metrics. The ability to utilize unpurified raw materials fundamentally alters the cost structure of D,L-methionine manufacturing by removing capital-intensive distillation columns and the associated energy loads. This reduction in processing steps directly correlates to a lower total cost of ownership for the production facility, enabling more competitive pricing in the global market without compromising on quality standards. Furthermore, the generation of high-purity ammonium sulfate as a byproduct, rather than low-value sodium sulfate or saline wastewater, creates an additional revenue stream that can offset production expenses and improve overall margin resilience against raw material price volatility.
- Cost Reduction in Manufacturing: The elimination of raw material rectification steps for methionaldehyde and the use of direct HCN gas significantly lowers utility consumption and equipment maintenance costs. By avoiding the complex purification trains required in traditional hydantoin routes, manufacturers can achieve substantial operational expenditure savings. The process design inherently minimizes waste disposal fees by converting potential effluents into saleable fertilizer-grade ammonium sulfate, thereby turning an environmental liability into an economic asset while streamlining the overall production budget.
- Enhanced Supply Chain Reliability: The stability of the 2-hydroxy-4-methylthiobutyronitrile intermediate is a game-changer for inventory management and production planning. Unlike unstable intermediates that require immediate downstream processing, this system can be stored for extended periods without decomposition, even at ambient temperatures when pH-adjusted. This flexibility allows manufacturers to build buffer stocks of key intermediates, mitigating the risk of supply disruptions caused by upstream feedstock fluctuations or maintenance downtime, thus ensuring a consistent and reliable supply of high-purity D,L-methionine to end-users.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, utilizing standard industrial equipment such as acid towers and high-pressure reactors that are well-understood in the fine chemical industry. The reduction in wastewater volume and the avoidance of heavy metal catalysts in the synthesis steps simplify environmental compliance and permitting processes. The production of a clean ammonium sulfate byproduct aligns with circular economy principles, reducing the ecological footprint of the facility and enhancing the brand reputation of the manufacturer as a sustainable partner in the global amino acid supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced D,L-methionine synthesis technology. These insights are derived directly from the experimental data and beneficial effects recorded in the patent documentation, providing a clear understanding of the process capabilities. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into existing production lines or for procurement specialists assessing the long-term viability of suppliers utilizing this methodology.
Q: What are the primary advantages of this nitrile route over the traditional hydantoin method?
A: Unlike the hydantoin route which generates significant wastewater and low-value sodium sulfate byproducts, this method utilizes direct HCN gas and unpurified methional, eliminating costly rectification steps and producing high-purity ammonium sulfate as a valuable agricultural byproduct.
Q: How does the process ensure high purity and bulk density in the final D,L-methionine product?
A: The process achieves 99% purity and a bulk density of 0.68-0.71 through precise pH control during the nucleophilic addition, the stability of the 2-hydroxy-4-methylthiobutyronitrile intermediate which prevents decomposition, and a specialized two-stage acid hydrolysis followed by flocculant-assisted crystallization.
Q: Is the 2-hydroxy-4-methylthiobutyronitrile intermediate stable for storage?
A: Yes, the patent confirms that the reaction system containing 2-hydroxy-4-methylthiobutyronitrile, along with residual HCN and water, remains stable for long-term storage without decomposition, especially when adjusted to a pH of 2-4, facilitating flexible production scheduling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D,L-Methionine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN103641758A and possess the technical expertise to bring this innovation to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of D,L-methionine meets the exacting standards required for pharmaceutical and high-end feed applications. Our commitment to process optimization means we can leverage the cost-saving mechanisms of this patent to deliver superior value to our global partners.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic implications of switching to this high-efficiency process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven technical capability. Let us collaborate to secure a sustainable and cost-effective supply of high-purity D,L-methionine for your future projects.
