Advanced Ruthenium Catalysis for Scalable Polyaromatic Naphthalene Derivative Production
Advanced Ruthenium Catalysis for Scalable Polyaromatic Naphthalene Derivative Production
The rapid evolution of organic electronics and targeted cancer therapies has intensified the demand for high-purity polyaromatic scaffolds, specifically naphthalene derivatives with complex substitution patterns. Patent CN108069934B introduces a groundbreaking methodology for preparing these valuable compounds through the reaction of biphenyl-type arones and tolanes catalyzed by ruthenium. This technology represents a significant leap forward in synthetic efficiency, utilizing a relatively inexpensive ruthenium catalyst to activate the aromatic ketone β-H bond, thereby constructing the six-membered naphthalene ring directly. Unlike traditional methods that rely on harsh conditions and stoichiometric oxidants, this approach operates under mild conditions using simple bases, offering a greener and more economically viable pathway for producing high-purity pharmaceutical intermediates and electronic chemical materials.
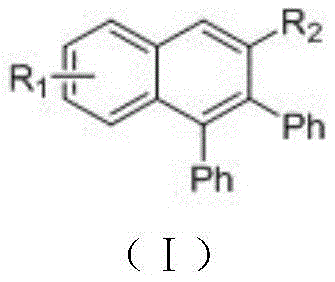
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclometallated polyaromatic systems has been plagued by significant operational and environmental hurdles. Conventional protocols often necessitate the use of aryl halides or aryl acids as starting materials, which require pre-functionalization steps that increase both cost and waste generation. Furthermore, existing transition metal-catalyzed C-H activation strategies frequently depend on the addition of specific ligands or equivalent amounts of metal salts, such as copper or silver, to act as oxidants to complete the catalytic circulation. These heavy metal salts not only escalate the raw material costs but also introduce severe environmental pollution concerns due to the difficulty in removing trace metal residues from the final active pharmaceutical ingredients or electronic materials. The reliance on harsh reaction conditions and toxic additives creates a bottleneck for reliable agrochemical intermediate supplier networks and pharmaceutical manufacturers aiming for green chemistry compliance.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a streamlined ruthenium-catalyzed cyclization that eliminates the need for external oxidants and complex ligand systems. By employing [RuCl2(p-cymene)]2 as the catalyst, the method facilitates the direct activation of the C-H bond on the aromatic benzene ring under mild thermal conditions. This innovation allows for the direct coupling of aromatic ketones with diphenylacetylene (tolane) to form the desired naphthalene core with high atom economy. The reaction proceeds efficiently in non-polar solvents like toluene at 100°C, utilizing a simple dual-base system of potassium acetate and sodium carbonate. This simplification of the reaction matrix drastically reduces the complexity of downstream purification, making it an ideal solution for cost reduction in electronic chemical manufacturing where purity is paramount.
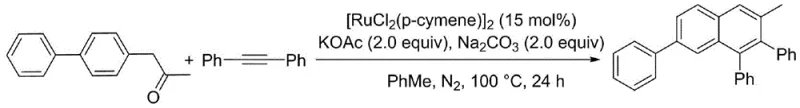
Mechanistic Insights into Ruthenium-Catalyzed Cyclization
The core of this technological breakthrough lies in the unique ability of the ruthenium catalyst to mediate β-H activation without auxiliary oxidants. The mechanism likely involves the initial coordination of the ruthenium species to the carbonyl oxygen of the biphenyl-type aromatic ketone, directing the metal center to the proximal C-H bond. Through a concerted metalation-deprotonation (CMD) pathway facilitated by the acetate base, the C-H bond is cleaved to form a stable ruthenacycle intermediate. Subsequent insertion of the alkyne (tolane) into the ruthenium-carbon bond followed by reductive elimination constructs the new carbon-carbon bonds required for the naphthalene ring system. This mechanistic pathway avoids the high-energy intermediates associated with radical processes, thereby minimizing the formation of polymeric byproducts and ensuring a cleaner reaction profile suitable for the commercial scale-up of complex polymer additives and fine chemicals.
From an impurity control perspective, the absence of stoichiometric oxidants like copper or silver salts is a critical advantage for R&D directors focused on quality. Heavy metal residues are notoriously difficult to reduce to ppm levels required by regulatory bodies for drug substances. By utilizing a catalytic amount of ruthenium (15 mol%) and avoiding additional metal oxidants, the impurity profile of the crude product is significantly simplified. The mild reaction temperature of 100°C further suppresses thermal decomposition pathways that often lead to tar formation in high-temperature cyclizations. This results in a robust process where the primary impurities are likely unreacted starting materials, which are easily removed via standard column chromatography or crystallization, ensuring the delivery of high-purity OLED material precursors with consistent batch-to-batch reproducibility.
How to Synthesize Polyaromatic Substituted Naphthalene Efficiently
The synthesis protocol outlined in the patent provides a reproducible framework for generating these complex architectures. The process begins with the precise weighing of diphenylacetylene and the specific aromatic ketone substrate, maintaining a molar ratio of 1:2 to drive the equilibrium towards the product. The reaction is conducted in a sealed tube under a nitrogen atmosphere to prevent oxidative degradation of the sensitive intermediates. The addition of the dual base system is crucial; while single bases showed lower yields in optimization studies, the combination of KOAc and Na2CO3 in toluene proved optimal, achieving yields up to 65% in model systems. Detailed standardized synthesis steps see the guide below.
- Combine diphenylacetylene and the specific aromatic ketone substrate in a sealed reaction vessel under an inert nitrogen atmosphere.
- Add the ruthenium catalyst [RuCl2(p-cymene)]2 along with a dual base system consisting of potassium acetate and sodium carbonate in toluene solvent.
- Heat the reaction mixture to 100°C for 24 hours to facilitate cyclization, followed by column chromatography purification to isolate the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed route offers tangible strategic benefits beyond mere technical novelty. The elimination of expensive and toxic oxidants translates directly into a simplified supply chain, reducing the number of SKUs that need to be sourced and qualified. Furthermore, the use of toluene, a commodity solvent, instead of specialized polar aprotic solvents or chlorinated hydrocarbons, aligns with modern sustainability goals and reduces solvent recovery costs. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or moisture, enhancing the reliability of supply for critical intermediates used in tyrosine protein kinase inhibitors and other high-value therapeutics.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of stoichiometric heavy metal oxidants with a catalytic system based on relatively cheap ruthenium. By removing the requirement for silver or copper salts, the raw material cost per kilogram of product is significantly lowered. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces labor hours and consumable costs associated with purification. This logical deduction of cost savings makes the process highly attractive for the mass production of fine chemical intermediates where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: The starting materials, specifically biphenyl-type aromatic ketones and tolanes, are commercially available building blocks with established global supply chains. Unlike custom-synthesized aryl halides which may have long lead times, these substrates can often be sourced from multiple vendors, mitigating the risk of supply disruption. The mild reaction conditions also imply that the process can be run in standard glass-lined or stainless steel reactors without requiring exotic high-pressure or cryogenic equipment, further ensuring that production schedules can be met consistently without unexpected downtime.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is facilitated by the homogeneous nature of the catalytic system and the use of common solvents. The absence of heavy metal waste streams simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. This aligns perfectly with increasingly stringent global environmental regulations, allowing manufacturers to maintain their social license to operate while expanding capacity. The process generates minimal hazardous waste, supporting the industry's shift towards greener synthesis routes for advanced materials and pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium-catalyzed technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: Does this ruthenium-catalyzed method require expensive oxidants?
A: No, a key advantage of the method described in patent CN108069934B is that it operates without the need for additional oxidants or ligands, relying solely on simple bases like KOAc and Na2CO3 to drive the catalytic cycle.
Q: What are the primary applications of these naphthalene derivatives?
A: These polyaromatic substituted naphthalene derivatives serve as critical intermediates for novel tyrosine protein kinase inhibitors in pharmaceuticals and as blue light-emitting materials in organic electronics and semiconductor applications.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes relatively inexpensive ruthenium catalysts and mild reaction conditions (100°C in toluene), avoiding heavy metal salt waste, which makes it scientifically reasonable and economically practical for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyaromatic Substituted Naphthalene Supplier
As the demand for sophisticated organic semiconductors and targeted oncology drugs continues to surge, the ability to manufacture complex naphthalene derivatives efficiently is a key competitive differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this chemical evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of ruthenium-catalyzed reactions, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that the transition from bench-scale discovery to commercial supply requires a partner who values both technical precision and operational excellence.
We invite R&D leaders and procurement specialists to collaborate with us to evaluate the feasibility of integrating this advanced synthesis route into your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data for our catalog of naphthalene derivatives and to discuss route feasibility assessments that could accelerate your time-to-market for next-generation electronic and pharmaceutical products.
