Scaling High-Purity Polyaromatic Naphthalene Derivatives via Ruthenium Catalysis for Commercial Applications
The chemical landscape for advanced functional materials is constantly evolving, driven by the need for more efficient and environmentally sustainable synthetic pathways. Patent CN108069934B introduces a groundbreaking methodology for the preparation of polyaromatic substituted naphthalene derivatives, utilizing a ruthenium-catalyzed reaction between biphenyl-type arones and tolane. This innovation represents a significant leap forward in the synthesis of complex aromatic systems, which are critical building blocks for high-performance optoelectronic devices and pharmaceutical intermediates. By leveraging the unique properties of ruthenium catalysis, this method bypasses the traditional reliance on harsh oxidants and expensive ligands, offering a streamlined route that aligns perfectly with modern green chemistry principles. For industry leaders seeking a reliable polyaromatic substituted naphthalene derivative supplier, understanding the mechanistic advantages of this patent is crucial for securing a competitive edge in the supply of high-value electronic chemicals.
Furthermore, the implications of this technology extend beyond mere academic interest, touching upon the core concerns of procurement and supply chain stability. The ability to synthesize these complex molecules under mild conditions without the need for stoichiometric amounts of heavy metal oxidants translates directly into reduced operational risks and lower waste disposal costs. As the demand for high-purity OLED material and semiconductor precursors continues to surge globally, adopting such efficient synthetic strategies becomes not just an option but a necessity for maintaining market leadership. This report delves deep into the technical nuances of this patented process, providing a comprehensive analysis for R&D directors and procurement managers alike who are tasked with optimizing their manufacturing portfolios for the future.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyaromatic substituted naphthalene derivatives has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Conventional cyclometallation processes typically rely on the use of transition metals such as copper or silver, which must be employed in stoichiometric or even excess quantities to act as oxidants to drive the catalytic cycle to completion. This dependency creates a dual burden: firstly, the cost of these precious metal salts is substantial and subject to volatile market fluctuations, directly impacting the cost reduction in electronic chemical manufacturing. Secondly, the presence of these heavy metals in the reaction mixture necessitates rigorous and often costly purification steps to ensure the final product meets the stringent purity specifications required for electronic applications. Residual metal contaminants can severely degrade the performance of organic light-emitting diodes or compromise the safety profile of pharmaceutical intermediates, making the removal process a critical bottleneck.
In addition to the economic and purity concerns, traditional methods often require harsh reaction conditions, including high temperatures and the use of aggressive additives that can lead to poor atom economy and significant environmental impact. The generation of large volumes of metal-containing waste streams poses a serious challenge for environmental compliance and increases the overall carbon footprint of the manufacturing process. For supply chain heads, these factors translate into longer lead times, higher regulatory burdens, and increased complexity in managing vendor relationships. The inability to efficiently scale these traditional routes without incurring prohibitive costs or environmental penalties has long been a pain point for companies aiming to commercialize complex polymer additives or specialty chemicals derived from naphthalene scaffolds. Consequently, there has been an urgent industry-wide demand for a more robust, economical, and green alternative.
The Novel Approach
The methodology disclosed in patent CN108069934B offers a transformative solution by employing a relatively inexpensive ruthenium catalyst, specifically [RuCl2(p-cymene)]2, to facilitate the activation of the aromatic ketone β-H bond. This innovative approach allows for the construction of the six-membered naphthalene ring through a direct cyclization reaction with diphenylacetylene, elegantly circumventing the need for any external oxidants or additives. The reaction proceeds smoothly under mild conditions, utilizing simple bases such as potassium acetate or sodium carbonate in a non-polar solvent like toluene. This simplification of the reaction system is a game-changer for the commercial scale-up of complex polyaromatic substituted naphthalene derivatives, as it drastically reduces the number of reagents required and minimizes the potential for side reactions that could generate difficult-to-remove impurities.
By eliminating the requirement for stoichiometric metal oxidants, this novel route inherently produces a cleaner reaction profile, which significantly eases the downstream purification burden. For a reliable agrochemical intermediate supplier or manufacturer of electronic materials, this means higher overall yields and a more consistent product quality without the need for extensive chromatographic separations or metal scavenging treatments. The use of mild conditions also enhances operational safety, reducing the energy consumption associated with heating and cooling cycles. From a strategic perspective, adopting this ruthenium-catalyzed protocol enables manufacturers to achieve substantial cost savings while simultaneously improving their environmental sustainability metrics. This alignment of economic efficiency with green chemistry principles makes the technology highly attractive for companies looking to future-proof their supply chains against increasingly strict global environmental regulations.
Mechanistic Insights into Ruthenium-Catalyzed Cyclization
The core of this technological breakthrough lies in the sophisticated mechanism of ruthenium-catalyzed C-H activation, which enables the direct functionalization of the aromatic ketone substrate. The catalyst [RuCl2(p-cymene)]2 acts as a precise molecular tool, coordinating with the carbonyl oxygen of the aromatic ketone to facilitate the cleavage of the proximal β-C-H bond. This activation step generates a reactive ruthenacycle intermediate, which is then poised to undergo insertion with the alkyne component, diphenylacetylene. Unlike traditional methods that rely on oxidative addition of aryl halides, this C-H activation pathway is inherently more atom-economical as it does not require pre-functionalized starting materials, thereby reducing the number of synthetic steps and the associated waste generation. The subsequent reductive elimination step releases the desired polyaromatic substituted naphthalene derivative and regenerates the active ruthenium species, allowing the catalytic cycle to continue efficiently without the need for external oxidation.
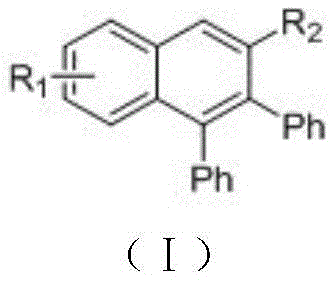
Controlling the impurity profile is paramount for applications in high-purity OLED material and pharmaceutical sectors, and this mechanism offers distinct advantages in that regard. Since the reaction does not involve the use of copper or silver salts, the risk of heavy metal contamination in the final product is virtually eliminated at the source. This is particularly critical for electronic materials where trace metals can act as quenching sites for excitons, drastically reducing the efficiency and lifespan of the device. Furthermore, the mild basic conditions employed in this process minimize the risk of base-sensitive functional group degradation, ensuring that delicate substituents on the aromatic rings remain intact. The selectivity of the ruthenium catalyst ensures that the cyclization occurs specifically at the desired position, reducing the formation of regioisomers that would otherwise complicate the purification process and lower the overall yield of the target compound.
How to Synthesize Polyaromatic Substituted Naphthalene Derivative Efficiently
Implementing this synthesis route in a practical setting requires careful attention to reaction parameters to maximize efficiency and reproducibility. The general procedure involves charging a reaction vessel with the aromatic ketone and diphenylacetylene in a molar ratio of approximately 1:2, ensuring an excess of the alkyne to drive the reaction to completion. The ruthenium catalyst is added at a loading of around 15 mol%, which strikes a balance between catalytic activity and cost-effectiveness for industrial applications. A mixed base system comprising potassium acetate and sodium carbonate is preferred to maintain the optimal pH environment for the C-H activation step without promoting unwanted side reactions. The reaction is conducted in toluene, a solvent that provides excellent solubility for the organic substrates while being easy to recover and recycle, further contributing to the process's green credentials.
- Combine diphenylacetylene and aromatic ketone substrates in a reaction vessel under a nitrogen atmosphere.
- Add the ruthenium catalyst [RuCl2(p-cymene)]2 along with a mixed base system of potassium acetate and sodium carbonate in toluene.
- Heat the mixture to 100°C for 24 hours, then purify the resulting derivative via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers compelling strategic advantages that go beyond simple technical metrics. The elimination of expensive and toxic oxidants like copper and silver salts directly addresses the issue of cost reduction in electronic chemical manufacturing by removing a significant variable cost component from the bill of materials. Moreover, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and shortens the overall production cycle time. This efficiency gain allows for a more agile response to market demands, enabling suppliers to offer more competitive pricing and faster delivery schedules for high-purity polyaromatic substituted naphthalene derivatives. The robustness of the reaction conditions also implies a lower risk of batch failures, enhancing the reliability of the supply chain.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of stoichiometric heavy metal oxidants, which are not only expensive to purchase but also costly to dispose of in an environmentally compliant manner. By utilizing a catalytic amount of ruthenium and simple inorganic bases, the raw material costs are significantly lowered, and the waste treatment burden is drastically reduced. This qualitative shift in the cost structure allows manufacturers to operate with higher margins or pass savings on to customers, creating a strong value proposition in a competitive market. Additionally, the use of common solvents like toluene facilitates easier solvent recovery and recycling, further minimizing operational expenditures associated with solvent procurement and waste management.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable reagents enhances the resilience of the supply chain against disruptions. Unlike specialized ligands or sensitive oxidants that may have limited suppliers or long lead times, the bases and solvents used in this protocol are commodity chemicals with robust global supply networks. This accessibility ensures that production can be maintained consistently without the risk of raw material shortages. Furthermore, the mild reaction conditions reduce the stress on equipment and lower the energy requirements, contributing to a more stable and predictable manufacturing environment. For buyers, this translates into a more dependable source of supply for critical intermediates, reducing the risk of production delays in their own downstream applications.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges related to heat transfer and safety, but the mild nature of this ruthenium-catalyzed reaction mitigates many of these risks. The absence of exothermic oxidation steps and the use of non-hazardous bases make the process inherently safer to operate at large volumes, facilitating a smoother transition from laboratory scale to commercial production. From an environmental perspective, the reduction in heavy metal waste aligns with increasingly stringent global regulations on chemical manufacturing, reducing the regulatory burden and potential liability for the manufacturer. This proactive approach to environmental stewardship not only safeguards the company's reputation but also ensures long-term operational continuity in a regulatory landscape that is becoming progressively stricter regarding industrial emissions and waste.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial aspects of this synthesis technology, based on the detailed disclosures within the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this methodology into their existing production frameworks. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for technical evaluation.
Q: Why is the ruthenium-catalyzed method superior to traditional cyclometallation?
A: Traditional methods often require expensive and toxic oxidants like copper or silver salts to complete the catalytic cycle. This patented ruthenium approach eliminates the need for external oxidants, significantly reducing heavy metal contamination and downstream purification costs.
Q: What are the primary applications of these polyaromatic naphthalene derivatives?
A: These derivatives possess unique electrochemical and photochemical properties, making them ideal for use in organic fluorescent materials, semiconductor components, and as intermediates for novel tyrosine protein kinase inhibitors in pharmaceutical development.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions, simple bases, and avoids hazardous oxidants, which simplifies safety protocols and waste treatment, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyaromatic Substituted Naphthalene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN108069934B for the production of high-value fine chemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial realities. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying that every batch of polyaromatic substituted naphthalene derivatives meets the exacting standards required for optoelectronic and pharmaceutical applications. We understand that consistency and purity are non-negotiable for our clients, and our state-of-the-art facilities are designed to deliver exactly that.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient methodology can optimize your budget. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate the full potential of this technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a product, but a comprehensive strategic alliance focused on driving innovation and efficiency in your manufacturing operations.
