Advanced Carbazolyl POSS Monomer Synthesis for High-Performance Optoelectronic Applications
Advanced Carbazolyl POSS Monomer Synthesis for High-Performance Optoelectronic Applications
The rapid evolution of the optoelectronic industry demands materials that combine robust thermal stability with superior luminescent properties, a challenge addressed comprehensively in patent CN108440759B. This groundbreaking intellectual property introduces a novel carbazolyl POSS (Polyhedral Oligomeric Silsesquioxane) monomer, synthesized through a sophisticated palladium-catalyzed Heck coupling reaction. By integrating the rigid, electron-rich carbazole moiety into the inorganic-organic hybrid POSS cage, the invention creates a material platform that significantly outperforms traditional organic fluorophores in terms of thermal endurance and structural integrity. For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, this technology represents a pivotal shift towards high-purity carbazolyl POSS monomer solutions that can withstand the rigorous demands of next-generation display and sensing applications. The strategic incorporation of nitrogen-containing heterocycles not only enhances fluorescence intensity but also opens new avenues for gas adsorption and environmental remediation, positioning this monomer as a versatile building block for advanced functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of silsesquioxane cages has relied heavily on less selective methodologies such as Friedel-Crafts reactions or oxidative coupling polymerization, which often suffer from significant drawbacks in industrial scalability and product consistency. These traditional pathways frequently result in broad molecular weight distributions and uncontrolled substitution patterns on the POSS cage, leading to batch-to-batch variability that is unacceptable for high-end electronic manufacturing. Furthermore, conventional methods often require harsh reaction conditions that can degrade sensitive functional groups, limiting the scope of compatible substituents and reducing the overall yield of the desired monomeric species. The lack of precise control over the spatial arrangement of organic groups around the inorganic core compromises the material's crystallinity and thermal performance, creating bottlenecks in the commercial scale-up of complex polymer additives. For supply chain heads, these inefficiencies translate into unpredictable lead times and increased waste generation, undermining the economic viability of large-scale production runs.
The Novel Approach
In stark contrast, the methodology disclosed in CN108440759B utilizes a highly selective Heck coupling strategy that ensures precise attachment of carbazole derivatives to the octavinylsilsesquioxane (OVS) core. This approach leverages the specific reactivity of vinyl groups on the POSS cage with aryl halides, facilitated by a optimized palladium catalyst system, to achieve a well-defined architecture with minimal side reactions. The use of 1-bromo-4(N-carbazolyl)benzene as a precursor allows for the introduction of the fluorescent carbazole unit with high fidelity, preserving the integrity of the conjugated system essential for optoelectronic performance. 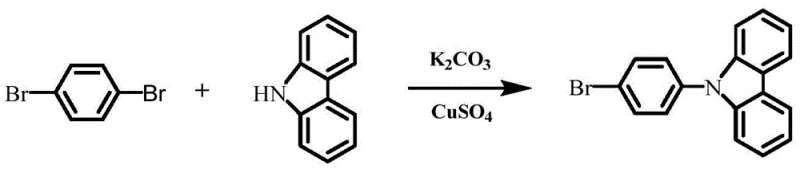 This precision synthesis not only enhances the thermal stability of the resulting monomer but also simplifies the downstream purification process, as the reaction profile is cleaner and more predictable than legacy methods. By adopting this novel approach, manufacturers can achieve cost reduction in display & optoelectronic materials manufacturing through improved yields and reduced solvent consumption during workup.
This precision synthesis not only enhances the thermal stability of the resulting monomer but also simplifies the downstream purification process, as the reaction profile is cleaner and more predictable than legacy methods. By adopting this novel approach, manufacturers can achieve cost reduction in display & optoelectronic materials manufacturing through improved yields and reduced solvent consumption during workup.
Mechanistic Insights into Pd-Catalyzed Heck Coupling
The core of this technological advancement lies in the intricate catalytic cycle of the palladium-mediated Heck reaction, which facilitates the carbon-carbon bond formation between the vinyl groups of OVS and the brominated carbazole derivative. The mechanism initiates with the oxidative addition of the aryl bromide to the zero-valent palladium species, generated in situ from palladium acetate and the tri-o-methylphenyl phosphine ligand. This step is critical for activating the relatively inert aryl halide, allowing it to coordinate with the metal center before inserting into the vinyl double bond of the silsesquioxane cage. The subsequent beta-hydride elimination restores the double bond character and releases the functionalized POSS product, while the palladium hydride species is regenerated by the base, triethylamine, to close the catalytic loop. 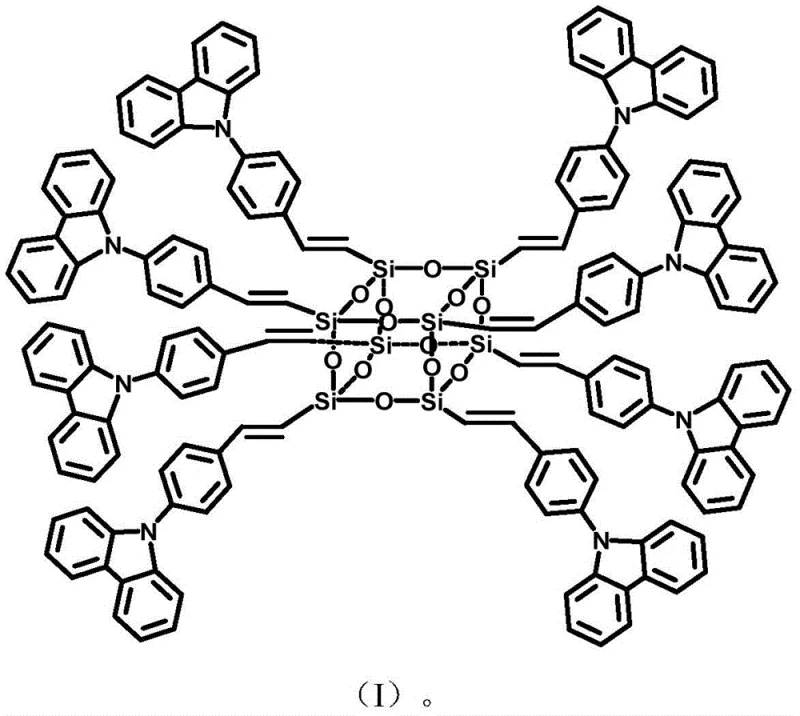 Understanding this mechanistic pathway is vital for R&D teams aiming to replicate the high purity and consistent fluorescence properties reported in the patent, as slight deviations in ligand choice or base strength can alter the turnover frequency and selectivity.
Understanding this mechanistic pathway is vital for R&D teams aiming to replicate the high purity and consistent fluorescence properties reported in the patent, as slight deviations in ligand choice or base strength can alter the turnover frequency and selectivity.
Furthermore, the impurity control mechanism inherent in this system is driven by the steric and electronic properties of the ligand environment surrounding the palladium center. The use of tri-o-methylphenyl phosphine provides a specific steric bulk that discourages the formation of homocoupling byproducts or over-substitution on the POSS cage, which are common pitfalls in multi-vinyl functionalization reactions. This selectivity ensures that the final product maintains a high degree of structural uniformity, which is directly correlated with its enhanced thermal stability and sharp fluorescence emission profiles. For quality assurance professionals, this means that the material exhibits a narrow polydispersity and consistent elemental composition, as verified by the close alignment of theoretical and experimental elemental analysis data. Such rigorous control over the molecular architecture is essential for applications requiring precise energy level alignment, such as in organic light-emitting diodes (OLEDs) or chemical sensors.
How to Synthesize Carbazolyl POSS Monomer Efficiently
Executing the synthesis of this high-value monomer requires strict adherence to the optimized reaction parameters outlined in the patent to ensure maximum conversion and ease of isolation. The process begins with the preparation of the reaction mixture under an inert nitrogen atmosphere, combining the OVS core and the carbazole precursor in dimethylformamide (DMF) solvent with triethylamine serving as the acid scavenger. 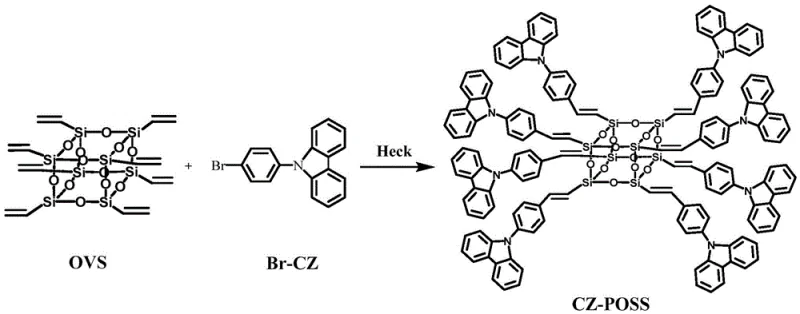 Critical to the success of the operation is the maintenance of the reaction temperature between 90-110°C for a duration of 40-50 hours, which provides sufficient thermal energy to drive the coupling to completion without degrading the sensitive organic components. Following the reaction, the workup procedure involves cooling, filtration, and rotary evaporation, followed by precipitation in methanol to remove soluble impurities and unreacted starting materials. The detailed standardized synthesis steps see the guide below.
Critical to the success of the operation is the maintenance of the reaction temperature between 90-110°C for a duration of 40-50 hours, which provides sufficient thermal energy to drive the coupling to completion without degrading the sensitive organic components. Following the reaction, the workup procedure involves cooling, filtration, and rotary evaporation, followed by precipitation in methanol to remove soluble impurities and unreacted starting materials. The detailed standardized synthesis steps see the guide below.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial strategic advantages for procurement managers and supply chain heads looking to optimize their material sourcing strategies. The streamlined nature of the Heck coupling process eliminates the need for multiple protection and deprotection steps often required in alternative synthetic routes, thereby drastically simplifying the manufacturing workflow and reducing overall operational costs. This efficiency gain is compounded by the use of readily available starting materials and a catalyst system that, while precious metal-based, operates with high turnover numbers, minimizing the total metal loading required per batch. For organizations focused on cost reduction in electronic chemical manufacturing, this translates to a more favorable cost-of-goods-sold profile without compromising on the premium quality required for high-tech applications.
- Prepare the reaction system by mixing 1-bromo-4(N-carbazolyl)benzene and octavinylsilsesquioxane (OVS) in DMF solvent with triethylamine as an acid absorbent.
- Add the palladium catalyst system, specifically palladium acetate and tri-o-methylphenyl phosphine ligand, under a nitrogen atmosphere to prevent oxidation.
- Heat the mixture to 90-110°C for 40-50 hours, followed by purification via column chromatography using dichloromethane and petroleum ether to isolate the final monomer.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and the high selectivity of the reaction significantly reduce solvent usage and waste generation, leading to lower environmental compliance costs and disposal fees. By avoiding the use of expensive transition metal removal resins often needed for less selective catalysts, the downstream purification process becomes more economical and faster. The robust nature of the reaction conditions also allows for the use of standard stainless steel reactors rather than specialized glass-lined equipment, further decreasing capital expenditure requirements for scale-up.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as DMF, triethylamine, and commercially available palladium salts ensures that the supply chain is resilient against raw material shortages or geopolitical disruptions. The simplicity of the isolation procedure, involving standard filtration and chromatography techniques, means that production can be easily transferred between different manufacturing sites without extensive requalification. This flexibility reduces lead time for high-purity photoelectric intermediates, allowing buyers to respond more agilely to market demand fluctuations and inventory requirements.
- Scalability and Environmental Compliance: The process is inherently scalable due to the homogeneous nature of the reaction mixture and the absence of exothermic spikes that are difficult to manage in large vessels. The use of triethylamine as a base generates manageable salt byproducts that can be easily separated, minimizing the environmental footprint associated with aqueous waste streams. This alignment with green chemistry principles supports corporate sustainability goals and ensures long-term regulatory compliance in jurisdictions with strict environmental standards.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and performance of this carbazolyl POSS technology in industrial settings. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this monomer into their existing product lines or research pipelines. The clarity on thermal limits and synthetic accessibility removes ambiguity for engineering teams planning pilot plant trials.
Q: What are the thermal stability characteristics of the carbazolyl POSS monomer?
A: According to patent CN108440759B, the synthesized monomer exhibits exceptional thermal stability, with thermogravimetric analysis indicating a 5% mass loss temperature at approximately 377°C, making it suitable for high-temperature processing.
Q: Why is the Heck coupling method preferred over traditional Friedel-Crafts reactions for this material?
A: The Heck coupling route offers superior control over the functionalization of the POSS cage, resulting in higher purity and better-defined structures compared to the broader product distributions often seen in Friedel-Crafts alkylation.
Q: Can this monomer be used for heavy metal adsorption applications?
A: Yes, the nitrogen-containing carbazole structure enhances the electron cloud density, significantly improving the interaction with adsorbates and increasing capacity for dye molecules and heavy metal ions in wastewater treatment scenarios.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazolyl POSS Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of material consistency and performance in the fast-paced electronics sector, which is why we have invested heavily in mastering complex synthetic pathways like the one described in CN108440759B. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality ensures that the carbazolyl POSS monomers supplied meet the exacting requirements for fluorescence intensity and thermal stability demanded by top-tier device manufacturers.
We invite you to collaborate with us to unlock the full potential of this advanced material for your specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and drive innovation in your product development.
