Advanced Synthesis of Carbazolyl POSS Monomers for High-Performance Optoelectronic Applications
Advanced Synthesis of Carbazolyl POSS Monomers for High-Performance Optoelectronic Applications
The rapid evolution of the organic light-emitting diode (OLED) and photoelectric material sectors demands intermediates that combine exceptional thermal stability with superior luminescent properties. Patent CN108440759B introduces a groundbreaking methodology for synthesizing a novel carbazolyl POSS (Polyhedral Oligomeric Silsesquioxane) monomer, addressing critical limitations in current hybrid material architectures. By strategically integrating carbazole derivatives into the rigid inorganic-organic POSS cage via a palladium-catalyzed Heck coupling reaction, this technology yields a monomer with significantly enhanced fluorescence and robustness. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this patent represents a pivotal shift towards more efficient, high-performance building blocks for next-generation display and sensing technologies. The detailed chemical pathway outlined in the documentation provides a clear roadmap for scaling these complex hybrid molecules without compromising purity or structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of silsesquioxane cages has relied heavily on methods such as Friedel-Crafts reactions, oxidative coupling polymerization, or free radical polymerization. These conventional approaches often suffer from significant drawbacks, including harsh reaction conditions that can degrade the delicate POSS cage structure or lead to uncontrolled cross-linking. Furthermore, achieving high degrees of functionalization with bulky aromatic groups like carbazole is notoriously difficult using older methodologies, often resulting in low yields and complex impurity profiles that are costly to remove. The lack of precise control over the substitution pattern on the POSS core frequently leads to heterogeneous mixtures, which negatively impacts the optical clarity and thermal performance of the final electronic material. Additionally, many traditional routes require expensive or hazardous reagents that complicate waste management and increase the overall environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology disclosed in CN108440759B utilizes a highly selective Heck coupling reaction to attach 1-bromo-4(N-carbazolyl)benzene units directly to the vinyl groups of octavinylsilsesquioxane (OVS). This approach operates under milder thermal conditions, typically between 90°C and 110°C, preserving the structural fidelity of the inorganic core while ensuring robust covalent bonding of the organic fluorophores. The use of a specific palladium catalyst system, comprising palladium acetate and tri-o-methylphenyl phosphorus ligands in a DMF solvent, facilitates a clean transformation with minimal side reactions. This precision synthesis not only simplifies the downstream purification process but also guarantees a homogeneous product distribution, which is essential for consistent performance in optoelectronic devices. By leveraging this modern cross-coupling strategy, manufacturers can achieve cost reduction in electronic chemical manufacturing through higher yields and reduced solvent consumption during purification.
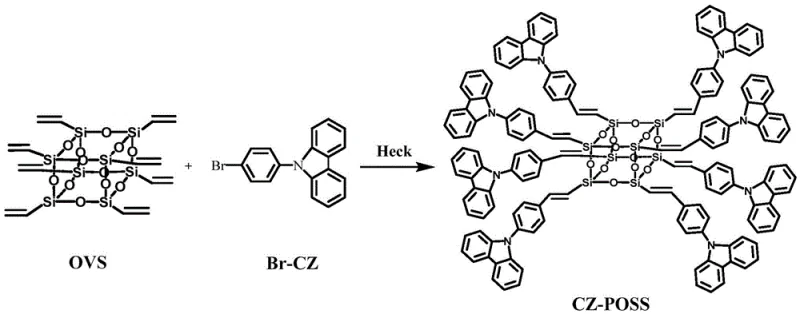
The visual representation of the synthetic route highlights the elegance of transforming simple vinyl-functionalized silica cages into sophisticated fluorescent monomers. This direct coupling avoids the multi-step protection and deprotection sequences often required in alternative syntheses, thereby streamlining the entire production timeline. The ability to tune the ratio of reactants allows for precise control over the degree of substitution, offering flexibility in tailoring the material's physical properties for specific applications such as gas adsorption or biological sensing.
Mechanistic Insights into Palladium-Catalyzed Heck Coupling
The core of this innovation lies in the mechanistic efficiency of the palladium-catalyzed Heck reaction, which proceeds through a well-defined catalytic cycle involving oxidative addition, migratory insertion, and beta-hydride elimination. In this specific system, the palladium(0) species generated in situ from palladium acetate and the phosphine ligand undergoes oxidative addition with the carbon-bromine bond of the carbazole derivative. This step is critical as it activates the aryl halide for subsequent coupling with the electron-deficient vinyl groups on the POSS cage. The presence of the bulky tri-o-methylphenyl phosphorus ligand plays a dual role: it stabilizes the active palladium center against aggregation and modulates the electronic environment to favor the desired coupling over competing homocoupling reactions. Following the coordination and insertion of the vinyl group into the palladium-aryl bond, the system eliminates hydrogen bromide, driven by the base triethylamine, to regenerate the active catalyst and release the functionalized product.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based methods. Because the reaction is catalytic and highly specific to the vinyl and aryl halide functionalities, there is minimal risk of random polymerization of the vinyl groups on the POSS cage, which would otherwise lead to insoluble gels or ill-defined oligomers. The use of a stoichiometric excess of the carbazole precursor ensures that the reaction drives to completion, minimizing the presence of partially substituted intermediates that could act as quenchers in fluorescence applications. Furthermore, the choice of DMF as a solvent ensures excellent solubility of both the polar inorganic core and the hydrophobic organic arms, maintaining a homogeneous reaction phase that promotes uniform kinetics. This level of mechanistic control is paramount for producing high-purity OLED material batches that meet the stringent specifications required by top-tier display manufacturers.
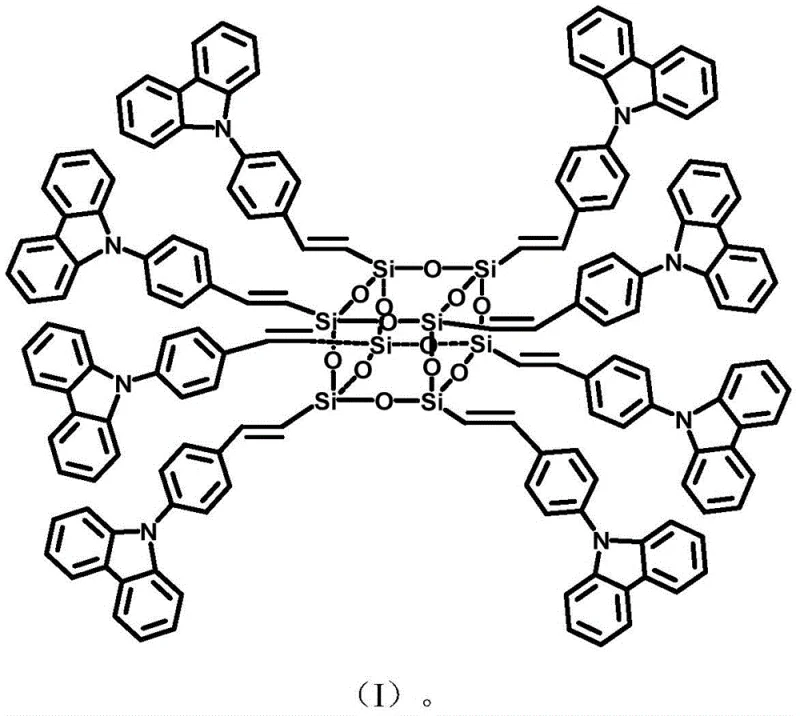
How to Synthesize Carbazolyl POSS Monomer Efficiently
The synthesis protocol described in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing the importance of inert atmosphere conditions and precise temperature control. To achieve optimal results, operators must strictly adhere to the specified molar ratios, particularly maintaining an OVS to bromo-carbazole ratio between 1:1.1 and 1:2 to drive the equilibrium towards the fully substituted product. The reaction mixture, containing the catalyst, ligand, and base, requires thorough degassing to prevent oxidation of the palladium species, which could deactivate the catalyst and lower yields. Following the extended reaction period of 40 to 50 hours, the workup involves a straightforward filtration and solvent removal process, followed by precipitation in methanol to isolate the crude solid. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining octavinylsilsesquioxane (OVS), 1-bromo-4(N-carbazolyl)benzene, palladium acetate catalyst, tri-o-methylphenyl phosphorus ligand, and triethylamine in DMF solvent under nitrogen atmosphere.
- Heat the reaction mixture to a temperature range of 90-110°C and maintain stirring for 40-50 hours to ensure complete Heck coupling conversion.
- Purify the crude product by cooling, filtering, rotary evaporation, precipitation in methanol, and silica gel column chromatography using dichloromethane and petroleum ether eluents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Heck coupling methodology presents substantial opportunities for optimizing the sourcing of advanced electronic intermediates. The primary raw materials, octavinylsilsesquioxane (OVS) and carbazole derivatives, are commercially available from established chemical suppliers, ensuring a stable and continuous supply chain without reliance on exotic or restricted precursors. The simplicity of the reaction setup, which utilizes standard glass-lined reactors and common solvents like DMF, means that existing manufacturing infrastructure can be easily adapted for production without requiring significant capital expenditure on specialized equipment. This ease of translation from lab to plant significantly reduces the lead time for high-purity electronic chemicals, allowing companies to respond rapidly to market demands for new display technologies.
- Cost Reduction in Manufacturing: The elimination of complex multi-step synthesis routes and the use of a highly efficient catalytic system drastically reduce the overall processing time and energy consumption. By avoiding the need for cryogenic conditions or ultra-high pressure reactors, the operational costs associated with utility usage are significantly lowered. Furthermore, the high selectivity of the Heck reaction minimizes the formation of byproducts, which reduces the volume of solvents and adsorbents required for purification, leading to substantial cost savings in waste treatment and raw material utilization.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with downstream device manufacturers. Since the process does not rely on unstable intermediates or sensitive reagents that require cold-chain logistics, the risk of supply disruption due to transportation issues is markedly decreased. The ability to store the key starting materials under ambient conditions further enhances inventory management flexibility, allowing for just-in-time production strategies that optimize working capital.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional stoichiometric methods, aligning with increasingly strict global environmental regulations. The use of triethylamine as a base produces manageable salt byproducts that can be easily separated and disposed of, reducing the burden on effluent treatment facilities. Additionally, the high thermal stability of the final product implies that it can withstand rigorous downstream processing conditions, reducing scrap rates during the fabrication of final electronic devices and contributing to a more sustainable manufacturing lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of carbazolyl POSS monomers. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this material into their existing product lines and assess the potential return on investment for adopting this new technology.
Q: What are the key advantages of introducing carbazole groups into POSS structures?
A: Introducing carbazole derivatives into the POSS cage structure significantly enhances the fluorescence intensity and thermal stability of the resulting monomer. The rigid conjugated system of carbazole improves electron cloud density, which boosts interaction with adsorbates, making it ideal for gas storage, sensing, and organic electronics applications.
Q: Why is the Heck coupling reaction preferred for functionalizing OVS in this patent?
A: The Heck coupling reaction allows for precise control over the functionalization of the octavinylsilsesquioxane (OVS) core under relatively mild conditions (90-110°C). Compared to free radical polymerization or Friedel-Crafts reactions, this method offers better selectivity and preserves the integrity of the sensitive POSS cage while attaching large aromatic groups.
Q: How does this synthesis method impact the scalability of OLED material production?
A: The process utilizes commercially available starting materials like OVS and bromo-carbazole derivatives with standard palladium catalysis. The purification steps involve common techniques like filtration and column chromatography, which can be adapted for larger scale production, ensuring a reliable supply chain for high-purity electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazolyl POSS Monomer Supplier
As the demand for high-performance optoelectronic materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis capabilities and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision and reliability. We understand that the commercial scale-up of complex polymer additives and electronic intermediates requires meticulous attention to detail, which is why our facilities are equipped with state-of-the-art rigorous QC labs to verify every batch against stringent purity specifications. Whether you require custom modifications to the POSS cage or large-volume supply of the standard monomer, our technical expertise ensures seamless integration into your manufacturing workflow.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall material costs. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your product development timelines and secure a competitive advantage in the global market.
