Scalable Whole-Cell Biocatalysis for High-Purity L-Menthol and D-Menthol Production
The global demand for high-purity chiral compounds, particularly in the flavor, fragrance, and pharmaceutical sectors, has driven significant innovation in resolution technologies. Patent CN1978659A introduces a transformative method for preparing L-menthol through the stereoselective hydrolysis of DL-fatty acid menthyl esters using a whole-cell biological process. Unlike traditional chemical resolution methods that rely on expensive chiral resolving agents or complex enzymatic immobilization techniques, this invention leverages the intrinsic stability of microbial cells, such as Rhizopus chinensis and Candida parapsilosis, to achieve efficient kinetic resolution. This approach not only simplifies the downstream processing but also aligns with modern green chemistry principles by reducing toxic waste and energy consumption. For procurement and R&D leaders, this technology represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value terpene alcohols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of optically pure L-menthol has been plagued by significant technical and economic hurdles associated with conventional resolution techniques. Traditional chemical methods often utilize optically active reagents, such as alkaloids or camphor sulfonic acid derivatives, to form diastereomeric salts or esters that can be separated by fractional crystallization. These processes are inherently inefficient, requiring multiple recrystallization steps that drastically reduce overall yield and generate substantial amounts of chemical waste. Furthermore, the resolving agents used are often prohibitively expensive and difficult to recover, leading to inflated production costs. Alternatively, earlier biocatalytic approaches relied on isolated enzymes that required rigorous purification and immobilization on solid supports to enhance stability. These immobilization procedures are technically demanding, often result in significant loss of enzymatic activity, and limit the substrate concentration due to mass transfer restrictions, thereby hindering large-scale commercial viability.
The Novel Approach
The methodology described in patent CN1978659A circumvents these bottlenecks by employing a direct whole-cell catalytic system. By utilizing intact microbial cells rather than purified enzymes, the process eliminates the need for cumbersome enzyme purification and immobilization steps, which are major cost drivers in biocatalysis. The cellular matrix naturally protects the intracellular esterases, providing a robust microenvironment that enhances operational stability and extends the catalyst's half-life to approximately 240 hours. This novel approach allows for significantly higher substrate concentrations, ranging from 0.1 to 15 g/100ml, compared to the dilute conditions often required for immobilized enzymes. Consequently, this leads to higher volumetric productivity and a simplified workflow that is far more amenable to industrial scale-up, offering a reliable pathway for the cost reduction in flavor and fragrance intermediate manufacturing.
Mechanistic Insights into Whole-Cell Stereoselective Hydrolysis
The core of this technology lies in the exquisite stereoselectivity of specific microbial strains towards the ester bond of DL-fatty acid menthyl esters. The process begins with the esterification of racemic DL-menthol to form DL-menthyl acetate or other fatty acid esters. When exposed to the whole-cell biocatalyst, the intracellular esterases exhibit a strong preference for hydrolyzing the ester bond of one specific enantiomer, typically the L-isomer precursor, while leaving the D-isomer ester largely untouched. This kinetic resolution is governed by the precise fit of the substrate within the enzyme's active site, where steric hindrance prevents the binding or hydrolysis of the unwanted enantiomer. The result is a reaction mixture containing free L-menthol and unreacted D-fatty acid menthyl ester, which can be easily separated due to their distinct physical properties.
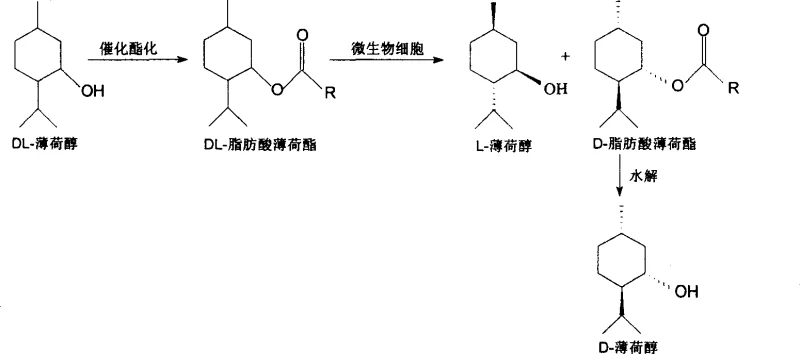
Following the biocatalytic step, the unreacted D-fatty acid menthyl ester serves as a valuable intermediate rather than waste. As illustrated in the reaction pathway, this byproduct can be subjected to a subsequent chemical hydrolysis step to yield high-purity D-menthol. This dual-product strategy maximizes atom economy, ensuring that both enantiomers of the starting racemic material are converted into commercially valuable products. The optical purity of the resulting L-menthol consistently reaches between 90% e.e. and 100% e.e., meeting the stringent quality standards required for fine chemical applications. Furthermore, the robustness of the whole-cell system allows it to maintain high selectivity even under varying pH and temperature conditions, providing a wide operating window that is critical for maintaining consistency in large-scale reactors.
How to Synthesize L-Menthol Efficiently
The synthesis of L-menthol via this whole-cell biocatalytic route involves a streamlined sequence of esterification, biological resolution, and separation. The process begins with the preparation of the substrate, DL-menthyl acetate, followed by incubation with lyophilized microbial cells in a buffered aqueous medium. The reaction conditions are carefully optimized to balance conversion rates with optical purity, typically operating at mild temperatures between 20°C and 40°C.
- Prepare DL-fatty acid menthyl ester substrate, typically DL-menthyl acetate, via catalytic esterification of DL-menthol.
- Conduct stereoselective hydrolysis using screened whole-cell microorganisms (e.g., Rhizopus chinensis) in a buffered aqueous system at controlled pH and temperature.
- Separate the resulting L-menthol product from the unreacted D-fatty acid menthyl ester, which can be subsequently hydrolyzed to recover high-purity D-menthol.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain managers and procurement officers, the adoption of this whole-cell biocatalytic process offers profound strategic advantages over legacy manufacturing routes. The elimination of enzyme immobilization and purification steps translates directly into a simplified supply chain with fewer raw material dependencies and reduced processing time. By removing the need for expensive chiral resolving agents and complex downstream purification trains, manufacturers can achieve substantial cost savings in production overhead. Additionally, the ability of the whole-cell catalyst to tolerate higher substrate concentrations means that reactor volumes can be utilized more efficiently, increasing throughput without proportional increases in capital expenditure. This efficiency is crucial for maintaining competitive pricing in the volatile flavor and fragrance market.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic simplification of the biocatalyst preparation. By bypassing the enzyme purification and immobilization stages, producers avoid the high costs associated with chromatography media, support materials, and activity loss during processing. This streamlined workflow significantly lowers the variable cost per kilogram of product. Furthermore, the reusability of the whole-cell biomass for multiple batches extends the effective life of the biocatalyst, distributing the initial fermentation costs over a larger volume of product and driving down the unit cost significantly.
- Enhanced Supply Chain Reliability: The robustness of the whole-cell system contributes to greater supply chain stability. Unlike isolated enzymes which may suffer from batch-to-batch variability or rapid deactivation during storage, lyophilized whole cells demonstrate excellent stability and can be stored for extended periods without significant loss of activity. The process tolerance to a wide range of substrate concentrations and reaction conditions reduces the risk of batch failures due to minor process deviations. This reliability ensures consistent delivery schedules and reduces the need for safety stock, allowing for a leaner and more responsive inventory management strategy.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this biological process offers a cleaner alternative to chemical resolution. The mild reaction conditions and aqueous-based system minimize the generation of hazardous organic waste and reduce energy consumption associated with heating and cooling. The ability to convert the D-isomer byproduct into valuable D-menthol further enhances the sustainability profile by maximizing resource utilization. These factors facilitate easier regulatory compliance and align with the increasing corporate mandates for green chemistry and sustainable sourcing in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this whole-cell hydrolysis technology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating the technology's fit for your specific application requirements.
Q: What are the advantages of using whole-cell biocatalysis over isolated enzymes for menthol resolution?
A: Whole-cell biocatalysis eliminates the complex and costly steps of enzyme purification and immobilization. This approach preserves natural enzyme stability within the cellular environment, reduces activity loss, and allows for broader substrate tolerance, significantly lowering production costs while maintaining high operational stability.
Q: What optical purity and conversion rates can be achieved with this method?
A: The process achieves high stereoselectivity, producing L-menthol with an optical purity ranging from 90% e.e. to 100% e.e. and conversion rates typically between 40% and 50%. The byproduct, D-fatty acid menthyl ester, can also be converted to D-menthol with 80% to 100% e.e., maximizing raw material utilization.
Q: Is the biocatalyst reusable for industrial scale-up?
A: Yes, the whole-cell catalysts demonstrate excellent reusability. Experimental data indicates that the lyophilized cells can be recycled and reused for multiple batches (at least 4 cycles) with only minimal decline in transformation efficiency, ensuring consistent performance for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Menthol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced biocatalytic technologies to meet the evolving demands of the global fine chemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the whole-cell hydrolysis of menthyl esters can be successfully translated into robust industrial operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of L-menthol and related intermediates meets the highest international standards for optical purity and chemical composition.
We invite potential partners to engage with our technical procurement team to discuss how this novel biocatalytic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your transition to high-efficiency, bio-based manufacturing is seamless and commercially viable.
