Advanced Catalytic Route for High-Purity L-Menthol Production and Commercial Scale-Up
The global demand for L-menthol, a critical compound in the flavor, fragrance, and pharmaceutical industries, continues to surge, driven by its widespread application in confectionery, oral care, and topical analgesics. However, meeting this demand has historically been constrained by the complexities of chiral synthesis and the limitations of existing catalytic technologies. Patent CN110845305B introduces a transformative methodology for preparing L-menthol utilizing a modified homogeneous catalyst, addressing the persistent bottlenecks of low yield and difficult chiral separation. This innovation represents a significant leap forward for any reliable flavor & fragrance intermediate supplier seeking to optimize production efficiency. The core of this technology lies in a novel organoaluminum catalyst system that facilitates the highly selective ring-closure of citronellal to isopulegol, followed by a robust chemical induction resolution process. By integrating advanced ligand design with practical recovery methods, this patent outlines a pathway that not only enhances stereochemical control but also simplifies the downstream processing required for commercial scale-up of complex fine chemicals.
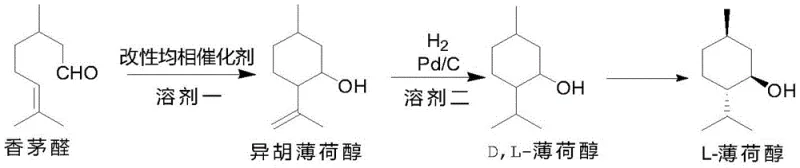
The transition from conventional synthesis routes to this novel approach marks a pivotal shift in manufacturing economics and technical feasibility. Traditional methods often rely on simple Lewis acids like zinc bromide (ZnBr2), which, while industrially established, suffer from inherent limitations such as moderate yields typically around 87% and selectivity capped at roughly 91%. Furthermore, earlier attempts to utilize organoaluminum catalysts were plagued by operational complexities, requiring the addition of ester auxiliary agents and necessitating low-temperature reactions that complicated catalyst recovery. In many prior art scenarios, the recovery process involved quenching the catalyst, which destroyed the ligand structure and prevented reuse, thereby inflating operational costs and waste generation. The novel approach detailed in this patent overcomes these hurdles by employing a specifically engineered ligand derived from 2,6-lutidine and ketone compounds. This new catalyst system operates effectively without auxiliary esters and, crucially, allows for the catalyst to be recovered through simple crystallization, preserving the ligand integrity for potential reuse cycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of menthol has faced significant challenges regarding stereoselectivity and process sustainability. The use of zinc-based catalysts, while common, results in a substantial portion of byproducts such as neo-isopulegol and neoisopulegol, which reduces the overall atom economy and necessitates energy-intensive purification steps to isolate the desired isopulegol intermediate. Additionally, the chiral resolution of the resulting D,L-menthol racemate has traditionally been a costly and loss-prone step. Conventional resolution techniques often involve multiple recrystallizations or enzymatic processes that can be sensitive to reaction conditions, leading to variable optical purity and significant product loss. The inability to efficiently recover expensive chiral auxiliaries or catalysts in these older processes further exacerbates the cost burden, making the production of high-purity L-menthol a capital-intensive endeavor. These inefficiencies create a supply chain vulnerability, where fluctuations in raw material costs or catalyst availability can severely impact production timelines and profit margins for manufacturers.
The Novel Approach
The methodology presented in CN110845305B fundamentally restructures the catalytic landscape for menthol production. By synthesizing a ligand through the reaction of 2,6-dimethylpyridine (2,6-lutidine) with specific ketone compounds using n-butyllithium, the process creates a molecular environment highly conducive to stereoselective cyclization. The subsequent reaction of this ligand with alkyl aluminum (AlR3) generates a modified homogeneous catalyst that exhibits superior performance metrics. Experimental data demonstrates that this system can achieve isopulegol yields exceeding 94% with selectivity rates greater than 98%, a marked improvement over the 81-88% yields seen with comparative catalysts. Moreover, the resolution strategy employs a chemical induction method using (S)-alpha-phenylethylamine, which leverages steric hindrance to selectively precipitate the desired levorotatory salt. This approach ensures that the final L-menthol product maintains high optical purity (ee% ≈ 98%) while minimizing product loss during the separation phases, thereby offering a more sustainable and economically viable route for cost reduction in synthetic menthol manufacturing.
Mechanistic Insights into Organoaluminum-Catalyzed Cyclization
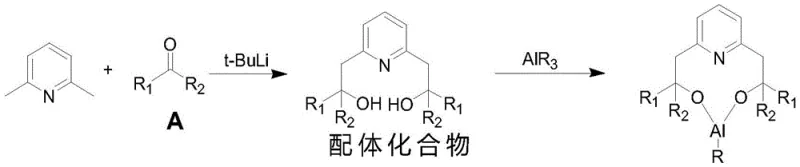
The efficacy of this synthesis hinges on the precise molecular architecture of the modified homogeneous catalyst. The ligand synthesis begins with the deprotonation of the methyl groups on 2,6-lutidine by n-butyllithium, a reaction facilitated by the electron-withdrawing nature of the pyridine nitrogen which activates the ortho-methyl C-H bonds. The resulting lithiated species reacts with a ketone compound to form a diol ligand structure. When this ligand coordinates with the aluminum center of the alkyl aluminum reagent, the two hydroxyl groups bind to the aluminum, creating a rigid, compact cyclic structure. Crucially, the nitrogen atom on the pyridine ring engages in Van der Waals interactions with the aluminum, further stabilizing the complex and defining its spatial geometry. The substituents (R1 and R2) on the ligand framework introduce specific steric hindrance that directs the approach of the citronellal substrate, favoring the formation of the desired isopulegol stereoisomer over unwanted byproducts. This intricate interplay of electronic effects and steric constraints is what drives the exceptional selectivity observed in the cyclization step.
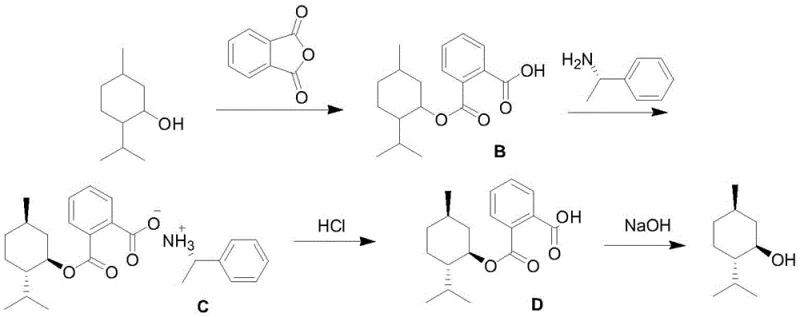
Following the cyclization, the chiral resolution mechanism is equally sophisticated in its simplicity and effectiveness. The process converts the racemic D,L-menthol into a monoester using phthalic anhydride, which modifies the solubility profile of the molecule to facilitate crystallization. The introduction of the chiral resolving agent, (S)-alpha-phenylethylamine, triggers a diastereomeric salt formation. Due to the specific three-dimensional arrangement of the L-menthol derivative, it forms a less soluble salt with the resolving agent compared to the D-isomer, primarily due to steric clashes that prevent the D-isomer from packing efficiently in the crystal lattice. This difference in solubility allows for the selective precipitation of the L-menthol salt, which can then be isolated by filtration. Subsequent acid and base hydrolysis steps cleave the ester and salt bonds without affecting the chiral center, releasing the free L-menthol with high optical purity. This chemical induction method avoids the need for expensive chromatographic separations or enzymatic reactors, making it highly scalable for industrial applications.
How to Synthesize L-Menthol Efficiently
The implementation of this patented technology requires strict adherence to anhydrous conditions and precise temperature control to maximize catalyst activity and product quality. The process is divided into three distinct stages: the preparation of the isopulegol intermediate via catalytic cyclization, the hydrogenation to D,L-menthol, and the final chiral resolution. Each stage utilizes standard chemical engineering unit operations such as distillation, filtration, and crystallization, ensuring compatibility with existing manufacturing infrastructure. For R&D teams looking to replicate or adapt this process, it is essential to focus on the purity of the starting materials, particularly the 2,6-lutidine and the ketone precursors, as impurities can poison the sensitive organoaluminum catalyst. The detailed standardized synthesis steps are outlined below to guide the technical execution of this high-efficiency route.
- Preparation of Isopulegol: React citronellal with a modified homogeneous organoaluminum catalyst in a dry solvent at -10 to 50°C under nitrogen protection to achieve high stereoselectivity.
- Hydrogenation to D,L-Menthol: Subject the resulting isopulegol to catalytic hydrogenation using Pd/C in an alcohol solvent at 0-110°C and 1-15 atm pressure.
- Chiral Resolution: Convert D,L-menthol into a monoester using phthalic anhydride, resolve using (S)-alpha-phenylethylamine to form a chiral salt, and hydrolyze to obtain pure L-menthol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this modified homogeneous catalyst technology offers tangible strategic benefits beyond mere technical superiority. The primary advantage lies in the substantial optimization of raw material utilization and waste reduction. By achieving yields above 94% in the critical cyclization step, the process significantly reduces the volume of unreacted citronellal and byproduct waste that must be managed or recycled. This efficiency translates directly into lower raw material consumption per kilogram of final product, providing a buffer against volatility in the prices of terpene feedstocks. Furthermore, the elimination of ester auxiliary agents, which were required in previous organoaluminum catalytic systems, simplifies the reaction mixture and reduces the number of chemical inputs needed, streamlining the procurement list and reducing inventory complexity.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the dual mechanisms of high yield and catalyst recoverability. In traditional processes, the loss of catalyst ligands during quenching represents a recurring cost that accumulates over large production volumes. In contrast, this novel catalyst can be recovered via crystallization, preserving the valuable ligand structure for potential reuse and drastically cutting down on catalyst replenishment costs. Additionally, the high selectivity (>98%) minimizes the formation of isomers that require expensive separation or disposal, effectively lowering the cost of goods sold (COGS) by improving the overall mass balance of the production line without requiring capital-intensive new equipment.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply continuity. The cyclization reaction operates within a moderate temperature range of -10 to 50°C and does not require extreme cryogenic conditions that strain utility systems. The raw materials, including 2,6-lutidine and common ketones, are commercially available commodities with stable supply chains, reducing the risk of bottlenecks associated with exotic or proprietary reagents. The simplicity of the resolution step, which relies on standard acid-base chemistry and crystallization rather than specialized enzymatic biocatalysts, further ensures that production can be maintained consistently even if specific biological reagents face supply disruptions, thereby securing the continuity of high-purity L-menthol supply.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this process aligns well with modern green chemistry principles. The ability to recover the catalyst reduces the generation of aluminum-containing waste streams, simplifying effluent treatment and lowering compliance costs associated with heavy metal discharge. The process avoids the use of chlorinated solvents in the resolution step where possible, favoring ethers and alcohols which are easier to recover and recycle. The scalability is evidenced by the use of standard hydrogenation and distillation equipment, meaning that scaling from pilot batches to multi-ton commercial production does not require fundamental changes to the reactor design, allowing for rapid capacity expansion to meet market demand spikes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this L-menthol synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the modified homogeneous catalyst improve isopulegol yield compared to traditional Lewis acids?
A: Unlike traditional ZnBr2 catalysts which typically achieve around 87% yield and 91% selectivity, the modified organoaluminum catalyst described in CN110845305B utilizes a specific ligand structure derived from 2,6-lutidine. This creates optimal steric hindrance and Van der Waals interactions that boost isopulegol yield to over 94% with selectivity exceeding 98%.
Q: Is the organoaluminum catalyst recoverable for industrial reuse?
A: Yes, a key advantage of this technology is the catalyst's ability to be recovered via crystallization. The ligand framework forms a compact structure with the aluminum center that allows it to precipitate as colorless crystals upon cooling, avoiding the complex quenching and ligand destruction steps required by previous generations of catalysts.
Q: What is the optical purity (ee%) achievable with this resolution method?
A: The chemical induction method using (S)-alpha-phenylethylamine effectively separates the levorotatory isomer. Experimental data indicates that the final L-menthol product can achieve an optical purity (ee%) of approximately 98%, ensuring high quality for flavor and pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Menthol Supplier
The technological advancements detailed in CN110845305B underscore the potential for highly efficient, cost-effective production of L-menthol, a cornerstone ingredient in the global flavor and pharmaceutical markets. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust industrial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and selectivity demonstrated in patent examples can be replicated reliably at scale. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify optical purity and impurity profiles, guaranteeing that every batch meets the exacting standards required by top-tier multinational corporations.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced catalytic technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific synthetic route can optimize your current sourcing strategy. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a secure and competitive supply of high-purity L-menthol for your future projects.
