Revolutionizing Aromatic Amide Production: A Green, One-Pot Catalytic Strategy for Commercial Scale-up
The landscape of fine chemical synthesis is undergoing a significant transformation, driven by the urgent need for greener, more atom-economical processes that reduce environmental impact while maintaining high efficiency. A pivotal advancement in this domain is detailed in Chinese Patent CN115108932A, which discloses a novel preparation method for aromatic amide compounds. This technology represents a paradigm shift from traditional acylation methods, utilizing a direct oxidative coupling between aromatic aldehydes and N,N-dimethylformamide (DMF). By leveraging a robust catalytic system involving ruthenium complexes or iodine sources alongside tert-butyl hydroperoxide, this method achieves high yields under remarkably mild conditions. For R&D directors and procurement specialists alike, this innovation offers a compelling alternative to legacy chemistries, promising streamlined operations and reduced hazard profiles in the manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of aromatic amides has relied heavily on the nucleophilic substitution reaction between amines and activated carboxylic acid derivatives, such as acid chlorides or acid anhydrides. While effective, these conventional pathways suffer from severe inherent drawbacks that impact both operational safety and overall process economics. The use of acid chlorides, for instance, necessitates the handling of highly reactive and corrosive reagents that pose significant safety risks to personnel and infrastructure. Furthermore, these reactions invariably generate stoichiometric amounts of hydrogen halide byproducts, which not only corrode reactor equipment but also require extensive neutralization and waste treatment procedures. Additionally, the atom economy of such processes is often suboptimal, as the leaving groups are discarded as waste, leading to higher raw material consumption and increased disposal costs that erode profit margins in competitive markets.
The Novel Approach
In stark contrast, the methodology outlined in the patent data introduces a direct oxidative amidation strategy that bypasses the need for pre-activated acid derivatives entirely. By employing readily available aromatic aldehydes and DMF as the nitrogen source, this route significantly simplifies the synthetic sequence. The reaction proceeds through a catalytic cycle that activates the aldehyde C-H bond, allowing for direct coupling with the amide nitrogen. This approach not only improves atom economy by incorporating more of the starting material into the final product but also eliminates the generation of corrosive halogenated waste streams. The versatility of this method is demonstrated by its tolerance to various substituents on the aromatic ring, including electron-withdrawing and electron-donating groups, ensuring broad applicability across different chemical scaffolds.
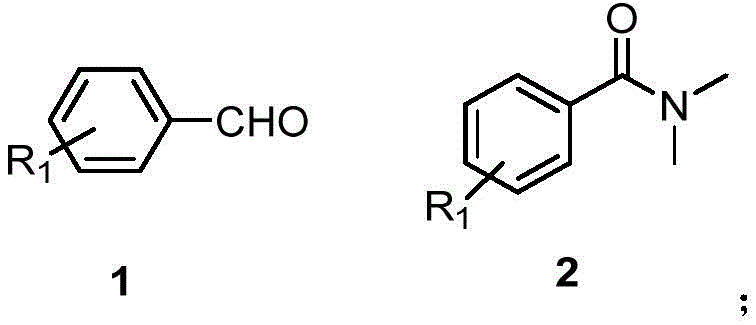
Mechanistic Insights into Ru-Catalyzed Oxidative Amidation
The core of this technological breakthrough lies in the sophisticated interplay between the transition metal catalyst and the oxidant. Specifically, when using tris(2,2'-bipyridine)ruthenium dichloride as the catalyst, the mechanism likely involves a single-electron transfer (SET) process initiated by the oxidant, tert-butyl hydroperoxide (TBHP). The ruthenium center facilitates the generation of radical species that abstract a hydrogen atom from the aldehyde substrate, forming an acyl radical intermediate. This highly reactive species is then trapped by the nitrogen lone pair of the DMF molecule, followed by further oxidation and deprotonation steps to yield the final aromatic amide. The presence of TBHP is critical, as experimental comparisons show that its omission leads to a precipitous drop in yield, confirming its role in driving the catalytic cycle forward and regenerating the active oxidized state of the metal catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over traditional coupling agents. Because the reaction does not involve the formation of unstable mixed anhydrides or highly reactive acid halides, the potential for side reactions such as over-acylation or hydrolysis is minimized. The use of a mild organic base, such as potassium tert-butoxide, in conjunction with the oxidant ensures that the reaction environment remains controlled, preventing the degradation of sensitive functional groups on the aromatic ring. This selectivity is paramount for pharmaceutical applications, where the presence of genotoxic impurities or difficult-to-remove byproducts can halt a drug development program. The ability to achieve high purity directly from the crude reaction mixture reduces the burden on downstream purification processes, thereby enhancing the overall efficiency of the manufacturing workflow.
How to Synthesize N,N-Dimethylbenzamide Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential to maximize yield and reproducibility. The patent specifies a optimized molar ratio of aldehyde to DMF to base to catalyst of 1:1.5:0.5:0.01, which balances reactivity with cost-efficiency. The solvent system, typically a mixture of dimethyl sulfoxide (DMSO) and water, plays a crucial role in solubilizing both the organic substrates and the inorganic base while stabilizing the polar transition states involved in the oxidation. Maintaining the reaction temperature at 60°C for approximately 8 hours provides sufficient thermal energy to overcome the activation barrier without promoting thermal decomposition of the reactants. Detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Prepare the reaction mixture by combining the aromatic aldehyde substrate, N,N-dimethylformamide (DMF), and an organic base such as potassium tert-butoxide in a solvent system of DMSO and water.
- Add the catalytic system, specifically tris(2,2'-bipyridine)ruthenium dichloride, along with tert-butyl hydroperoxide (TBHP) as the terminal oxidant to initiate the oxidative transformation.
- Heat the reaction mixture to 60°C and stir for approximately 8 hours, followed by aqueous workup and purification to isolate the high-purity aromatic amide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this oxidative amidation technology translates into tangible strategic benefits that extend beyond simple yield improvements. By shifting away from hazardous acid chlorides, companies can significantly reduce the costs associated with specialized storage, handling protocols, and hazardous waste disposal. The reliance on commodity chemicals like DMF and simple aromatic aldehydes ensures a stable and resilient supply chain, mitigating the risks of raw material shortages that often plague specialty reagent markets. Furthermore, the simplified workup procedure, which involves a straightforward extraction and evaporation, reduces the demand for labor-intensive purification steps, allowing for faster batch turnover and improved facility utilization rates.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous activating reagents such as thionyl chloride or oxalyl chloride directly lowers the raw material cost per kilogram of the final product. Additionally, the high atom economy of the reaction means that less raw material is wasted as byproduct, further optimizing the cost structure. The reduction in waste treatment complexity also leads to substantial operational savings, as facilities do not need to invest heavily in scrubbing systems for acidic gases or specialized containment for corrosive liquids.
- Enhanced Supply Chain Reliability: The starting materials for this process, including benzaldehydes and DMF, are produced on a massive global scale, ensuring consistent availability and price stability. This contrasts sharply with custom-synthesized coupling agents that may have long lead times or limited supplier bases. By utilizing a robust catalytic system that tolerates minor variations in feedstock quality, manufacturers can maintain continuous production schedules even when facing minor supply fluctuations, thereby securing the continuity of critical pharmaceutical intermediate supplies.
- Scalability and Environmental Compliance: The use of DMSO and water as solvents aligns well with modern green chemistry principles and regulatory expectations regarding volatile organic compound (VOC) emissions. The mild reaction conditions (60°C) reduce energy consumption compared to high-temperature reflux processes, contributing to a lower carbon footprint. This environmental compatibility simplifies the permitting process for new production lines and ensures long-term compliance with increasingly stringent environmental regulations, safeguarding the company against future regulatory risks.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this aromatic amide synthesis protocol. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on catalyst selection, oxidant necessity, and substrate scope. Understanding these nuances is essential for process chemists aiming to adapt this laboratory-scale success to large-scale commercial production environments.
Q: What are the key advantages of this oxidative amidation method over traditional acid chloride routes?
A: Unlike traditional methods that require hazardous acid chlorides and produce corrosive hydrogen halide byproducts, this patent-described method utilizes stable aldehydes and DMF. It operates under mild conditions (60°C) with high atom economy, eliminating the need for complex waste treatment associated with halogenated byproducts.
Q: Why is tert-butyl hydroperoxide (TBHP) critical in this catalytic cycle?
A: Experimental data within the patent indicates that omitting TBHP results in drastically reduced yields (dropping from 85% to 25%). TBHP acts as a synergistic oxidant that regenerates the active catalytic species and facilitates the oxidative cleavage of the C-H bond in the aldehyde, ensuring high conversion rates without requiring excessive amounts of organic base.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable due to its one-pot nature and the use of common solvents like DMSO and water. The absence of sensitive intermediates that require isolation simplifies the workflow, making it ideal for commercial scale-up of complex pharmaceutical intermediates while maintaining stringent purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this oxidative amidation technology for the global pharmaceutical and fine chemical industries. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust manufacturing processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of aromatic amide intermediate delivered meets the highest standards of quality and consistency required by top-tier multinational corporations.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green chemistry approach can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your critical aromatic amide intermediates.
