Advanced Copper-Catalyzed Oxidative Amidation for High-Purity Pharmaceutical Intermediates
Introduction to Green Oxidative Amidation Technology
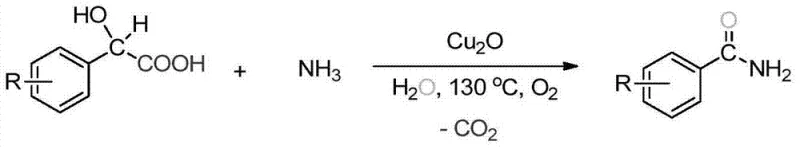
The synthesis of aromatic amides, particularly benzamide derivatives, represents a cornerstone operation in the manufacturing of active pharmaceutical ingredients (APIs) and agrochemical intermediates. Traditionally, this transformation has relied on stoichiometric activation of carboxylic acids, a process fraught with economic and environmental inefficiencies. However, the technological landscape is shifting towards more sustainable catalytic manifolds. Patent CN103772229A introduces a groundbreaking methodology for the preparation of aromatic formamide (benzamide) derivatives utilizing a copper-catalyzed oxidative system. This innovative approach leverages molecular oxygen as the sole oxidant and ammonia as the nitrogen source, effectively bypassing the need for pre-activation of the carboxylic acid substrate. For R&D directors and process chemists, this represents a paradigm shift from atom-uneconomical coupling reactions to a direct, catalytic oxidative decarboxylation strategy that aligns perfectly with modern green chemistry principles.
The core of this technology lies in its simplicity and robustness. By employing inexpensive copper(I) oxide (Cu2O) as the catalyst precursor, the reaction proceeds under relatively mild thermal conditions (100-130 °C) in polar aprotic solvents such as DMSO or DMF. The significance of this patent extends beyond mere academic curiosity; it addresses critical pain points in industrial organic synthesis, specifically the handling of hazardous reagents and the management of toxic waste streams. For procurement managers and supply chain heads, the implication is clear: a route that relies on commodity chemicals (oxygen, ammonia, copper salts) rather than specialized, high-cost coupling reagents offers a tangible pathway to cost reduction and supply chain resilience. This report analyzes the technical merits and commercial viability of this copper-catalyzed protocol, positioning it as a superior alternative for the large-scale production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the established canon of organic synthesis, the construction of the amide bond is frequently achieved through the activation of carboxylic acids. The classical route involves the conversion of the acid to an acid chloride using thionyl chloride (SOCl2) or oxalyl chloride, followed by nucleophilic attack by an amine. Alternatively, peptide coupling reagents such as DCC, EDC, or HATU are employed to activate the acid in situ. While effective on a small scale, these methods present severe drawbacks when translated to commercial manufacturing. The acid chloride route generates stoichiometric amounts of hydrogen chloride (HCl) gas, which is highly corrosive to reactor vessels and necessitates expensive scrubbing systems and specialized metallurgy (e.g., glass-lined or Hastelloy reactors) to prevent equipment failure. Furthermore, the handling of thionyl chloride poses significant safety risks due to its toxicity and reactivity with moisture.
Coupling reagent methodologies, while milder, suffer from poor atom economy and high material costs. Reagents like HATU are prohibitively expensive for multi-kilogram or ton-scale production, drastically inflating the Cost of Goods Sold (COGS). Additionally, these reactions often produce difficult-to-remove urea byproducts, complicating downstream purification and increasing solvent consumption. From a supply chain perspective, reliance on these specialized reagents introduces vulnerability; any disruption in the supply of coupling agents can halt production lines. The cumulative environmental burden of the waste generated—chlorinated byproducts, urea derivatives, and spent solvents—also imposes heavy regulatory compliance costs, making these conventional routes increasingly unsustainable for modern chemical enterprises seeking to minimize their carbon footprint.
The Novel Approach
The methodology disclosed in CN103772229A offers a transformative solution by enabling the direct conversion of aryl-substituted acetic acids (such as phenylacetic acid or mandelic acid derivatives) into benzamides using ammonia and oxygen. This oxidative decarboxylative amidation eliminates the need for any external activating agents or halogenated reagents. The reaction mechanism utilizes atmospheric oxygen (or pure O2) as the terminal oxidant, which is not only the cheapest and most abundant oxidant available but also generates water or carbon dioxide as benign byproducts. Specifically, the process converts the carboxylic acid moiety directly into the amide functionality with the loss of CO2, a feature that simplifies the reaction workup significantly. The absence of corrosive HCl evolution means that standard stainless steel reactors can be utilized, lowering capital expenditure (CAPEX) for plant infrastructure.
Moreover, this novel approach demonstrates exceptional functional group tolerance, a critical parameter for the synthesis of complex drug intermediates. The catalytic system is compatible with a wide array of substituents on the aromatic ring, including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens (fluoro, chloro, bromo) and nitro groups. This versatility allows chemists to synthesize diverse libraries of benzamide derivatives without the need for tedious protecting group strategies. The operational simplicity of the process—a one-pot reaction in a pressure vessel—further enhances its appeal for scale-up. By merging the oxidation and amidation steps into a single catalytic cycle, the process reduces unit operations, solvent usage, and overall processing time, delivering a streamlined manufacturing workflow that is both economically and environmentally superior to traditional activation-based methods.
Mechanistic Insights into Cu2O-Catalyzed Oxidative Decarboxylation
Understanding the mechanistic underpinnings of this transformation is vital for R&D teams aiming to optimize the process for specific substrates. The reaction is proposed to proceed via a copper-catalyzed radical pathway. Initially, the copper(I) species interacts with molecular oxygen to generate reactive copper-oxygen intermediates, possibly involving superoxo or peroxo species. These activated oxygen species facilitate the oxidative decarboxylation of the alpha-substituted acetic acid substrate. In the case of alpha-hydroxy acids (mandelic acids), the presence of the hydroxyl group likely assists in the coordination to the copper center, promoting the single-electron transfer (SET) processes required to cleave the C-C bond adjacent to the carbonyl. This decarboxylation step releases CO2, consistent with the patent's claim of carbon dioxide being the sole byproduct, and generates a transient benzyl or acyl radical/cation species.
This reactive intermediate is subsequently trapped by ammonia (present in excess as ammonium hydroxide) to form the final benzamide product. The regeneration of the active copper catalyst completes the catalytic cycle, ensuring that only sub-stoichiometric amounts of the metal are required (typically 10-20 mol%). Crucially, this mechanism avoids the formation of stable salt byproducts common in coupling reactions. The impurity profile is consequently much cleaner, primarily consisting of unreacted starting material or minor over-oxidation products, which are easily removed during the standard aqueous workup described in the patent (washing with NaHCO3 and brine). This mechanistic clarity assures quality control teams that the risk of heavy metal contamination or persistent organic impurities is minimal, provided standard purification protocols like silica gel chromatography or recrystallization are applied.
How to Synthesize Aromatic Benzamides Efficiently
Implementing this copper-catalyzed oxidative amidation in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The patent outlines a robust protocol involving the use of pressure-resistant sealed tubes or autoclaves, reflecting the need to maintain an oxygen-rich atmosphere at elevated temperatures. The choice of solvent is critical; polar aprotic solvents like DMSO or DMF are preferred as they stabilize the charged intermediates and dissolve both the organic substrate and the inorganic catalyst/ammonia system effectively. The reaction temperature is typically maintained between 100 °C and 130 °C, balancing reaction kinetics with thermal stability. Monitoring the reaction progress via TLC or GC is recommended to determine the precise endpoint, which generally falls within the 24 to 36-hour window depending on the electronic nature of the substrate substituents.
- Charge a pressure-resistant reactor with the aryl acetic acid derivative (e.g., mandelic acid), Cu2O catalyst (10-20 mol%), and aqueous ammonia in a polar aprotic solvent like DMSO or DMF.
- Pressurize the system with oxygen and heat the mixture to 100-130 °C with stirring for 24-36 hours to facilitate oxidative decarboxylation and amidation.
- Upon completion, cool the reaction, extract with ethyl acetate, wash with bicarbonate and brine, dry over sodium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed technology offers compelling strategic advantages that extend beyond simple yield metrics. The primary value driver is the drastic simplification of the raw material portfolio. By replacing expensive, specialty coupling reagents and corrosive chlorinating agents with commodity chemicals like copper oxide, ammonia, and oxygen, manufacturers can significantly insulate their production costs from market volatility. The elimination of hazardous reagents also reduces the regulatory burden associated with storage, handling, and disposal, leading to lower operational expenditures (OPEX) related to environmental health and safety (EHS) compliance. This shift towards inherently safer chemistry not only protects the workforce but also minimizes the risk of production shutdowns due to safety incidents or regulatory audits.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalytic route is substantial. Traditional amide bond formation often accounts for a significant portion of material costs in API synthesis due to the high price of coupling reagents and the stoichiometric waste they generate. This new method utilizes catalytic amounts of inexpensive copper salts and free oxygen from the air, effectively reducing the direct material cost per kilogram of product. Furthermore, the simplified workup procedure—requiring only basic aqueous washes and solvent evaporation—reduces solvent consumption and waste treatment fees. The avoidance of corrosive byproducts also extends the lifespan of reactor equipment, deferring capital replacement costs and reducing maintenance downtime.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available feedstocks. Unlike specialized coupling agents which may have limited suppliers and long lead times, copper oxides and ammonia are produced globally at massive scales, ensuring a stable and continuous supply. This reduces the risk of bottlenecks that can delay project timelines. Additionally, the robustness of the reaction across a wide range of substrates means that a single standardized protocol can be applied to multiple intermediates, simplifying inventory management and operator training. This flexibility allows manufacturing sites to respond more agilely to changes in demand for different pharmaceutical intermediates without retooling or sourcing new, exotic reagents.
- Scalability and Environmental Compliance: From a scale-up perspective, the process is exceptionally well-suited for large-volume production. The exothermic nature of oxidation reactions is manageable under the described conditions, and the use of oxygen gas allows for efficient mass transfer in stirred tank reactors. Environmentally, the process aligns with strict global regulations on waste discharge. With CO2 as the primary byproduct and no halogenated waste streams, the environmental footprint is minimized. This "green" credential is increasingly important for securing contracts with major pharmaceutical companies that have aggressive sustainability goals. The ability to market intermediates produced via a non-corrosive, low-waste pathway adds intangible value to the supply chain, enhancing the manufacturer's reputation as a responsible partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed oxidative amidation technology. These insights are derived directly from the experimental data and claims presented in patent CN103772229A, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into existing manufacturing workflows. The answers reflect the balance between high-yield performance and operational practicality that defines this innovative synthetic route.
Q: What are the primary advantages of this copper-catalyzed method over traditional coupling reagents?
A: This method eliminates the need for expensive and hazardous activating agents like thionyl chloride or carbodiimides (DCC/EDC). It uses molecular oxygen as the terminal oxidant, producing only CO2 as a byproduct, which significantly reduces waste treatment costs and equipment corrosion risks associated with HCl generation.
Q: Does this process tolerate sensitive functional groups on the aromatic ring?
A: Yes, the patent data demonstrates excellent functional group compatibility. Electron-donating groups (e.g., methoxy, methyl, tert-butyl) and electron-withdrawing groups (e.g., fluoro, chloro, nitro, trifluoromethyl) are all tolerated, yielding the corresponding benzamides in moderate to high yields without protecting group strategies.
Q: Is the catalyst system scalable for industrial production?
A: The process utilizes earth-abundant copper oxides (Cu2O) and commodity gases (O2, NH3), making it highly scalable. The absence of corrosive byproducts allows for the use of standard stainless steel reactors, facilitating a smoother transition from laboratory to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzamide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the Cu2O-mediated oxidative amidation described in CN103772229A. As a leading CDMO and supplier of fine chemical intermediates, we possess the technical expertise to translate such patent-protected methodologies into robust, commercial-scale processes. Our team of process chemists is adept at optimizing reaction parameters—such as catalyst loading, oxygen pressure, and temperature profiles—to ensure maximum efficiency and reproducibility. We understand that moving from gram-scale discovery to multi-ton production requires rigorous attention to detail, and our facilities are equipped to handle complex oxidative chemistries safely and effectively.
We invite pharmaceutical and agrochemical companies to leverage our capabilities for the Customized Cost-Saving Analysis of their current amide synthesis routes. By partnering with us, you gain access to our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply chain is both economical and resilient. Our state-of-the-art rigorous QC labs guarantee that all intermediates meet stringent purity specifications, regardless of the synthetic route employed. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can deliver high-purity benzamide derivatives that drive your projects forward.
