Advanced Solid-Phase Synthesis of Cefotaxime: Technical Breakthroughs and Commercial Scalability
Advanced Solid-Phase Synthesis of Cefotaxime: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry continuously seeks robust manufacturing methodologies that enhance product quality while streamlining operational complexity, particularly for critical antibiotics like cefotaxime sodium. Patent CN108117565B introduces a transformative solid-phase synthesis method that fundamentally restructures the production landscape for this third-generation cephalosporin. By anchoring the 7-ACA starting material onto a solid support, this innovation bypasses the cumbersome purification bottlenecks inherent in traditional solution-phase chemistry. The resulting process not only elevates the chemical purity to exceptional standards but also significantly mitigates environmental impact through reduced solvent usage. For global procurement teams, this represents a pivotal shift towards more reliable and efficient supply chain models for high-value beta-lactam intermediates.
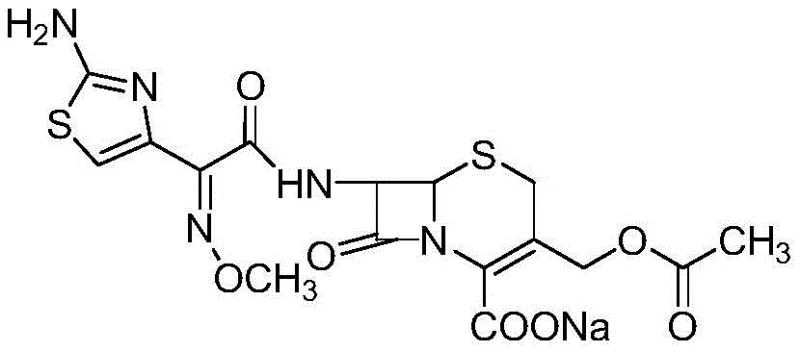
Cefotaxime remains a cornerstone in treating severe bacterial infections, ranging from septicemia to respiratory tract complications, necessitating a supply chain that guarantees unwavering consistency. The structural integrity of the molecule, characterized by its sensitive beta-lactam ring and methoxyimino side chain, demands precise synthetic control to prevent degradation. Traditional manufacturing often struggles with crystallization inconsistencies and residual impurities that complicate downstream formulation. This patented approach addresses these vulnerabilities by leveraging the unique advantages of solid-phase technology, where the growing molecule is physically separated from reaction byproducts at every stage. Consequently, the final active pharmaceutical ingredient exhibits superior stability and bioavailability, meeting the rigorous specifications demanded by international regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of cefotaxime has relied on a two-step solution-phase process that involves reacting 7-ACA with AE-active ester in dichloromethane followed by acidification and salification. This conventional pathway is fraught with technical inefficiencies, primarily due to the difficulty in controlling crystallization conditions which directly impacts the physical form and purity of the final product. The extensive use of organic solvents like dichloromethane not only escalates production costs but also imposes significant environmental burdens regarding waste disposal and solvent recovery. Furthermore, the necessity for multiple post-treatment steps, including drying and extensive purification, introduces opportunities for product degradation and yield loss. These operational complexities create volatility in supply continuity, making it challenging for manufacturers to maintain consistent batch-to-batch quality.
The Novel Approach
In stark contrast, the solid-phase synthesis method described in the patent revolutionizes the workflow by immobilizing the 7-ACA substrate onto a chloromethyl resin carrier before initiating the acylation reaction. This strategic modification allows for the facile removal of excess reagents and soluble byproducts simply by washing the resin, effectively eliminating the need for complex extraction and crystallization procedures during the intermediate stages. The reaction environment is tightly controlled within the solid matrix, which enhances the selectivity of the acylation and minimizes the formation of unwanted isomers or degradation products. By integrating the synthesis and purification phases, the process drastically reduces the overall operational time and resource consumption. This streamlined methodology ensures a more robust and predictable manufacturing outcome, aligning perfectly with the needs of modern cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Solid-Phase Acylation and Cleavage
The core of this technological advancement lies in the meticulous orchestration of the bridging and acylation steps, which dictate the overall success of the synthesis. Initially, 7-ACA is covalently bonded to the solid phase carrier through a nucleophilic substitution reaction in a polar aprotic solvent such as DMF or DMSO. This bridging step is critical as it establishes the anchor point that allows subsequent reactions to occur while the molecule remains insoluble, facilitating easy separation. Following the bridging, the resin-bound intermediate undergoes acylation with 2-(2-amino-4-thiazolyl)-2-(methoxyimino) thiothiazole acetate in the presence of an organic base like triethylamine. The solid support acts as a pseudo-dilution agent, potentially reducing intermolecular side reactions and promoting the formation of the desired amide bond with high stereochemical fidelity.
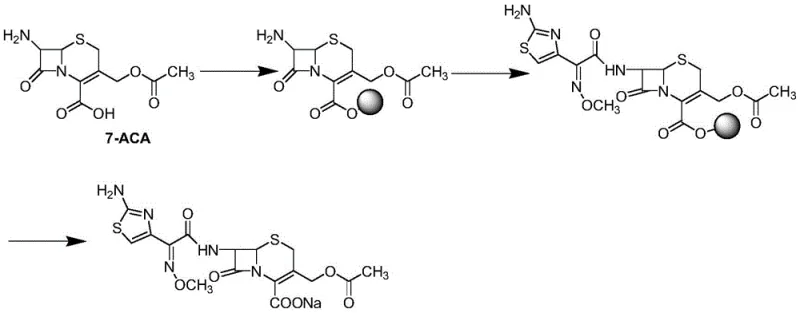
Following the successful construction of the cefotaxime skeleton on the resin, the final and most delicate step involves the cleavage of the product from the solid support. This is achieved through a carefully timed acidolysis process using a mixture of organic solvents and strong acids such as trifluoroacetic acid or trifluoromethanesulfonic acid. The conditions are optimized to break the linker bond without compromising the sensitive beta-lactam ring, which is prone to hydrolysis under harsh acidic conditions. The patent specifies a temperature gradient, cooling the mixture to -20°C during acid addition to suppress exothermic degradation pathways. Once cleaved, the product is released into the solution phase where it can be easily converted to the sodium salt and crystallized, yielding a product with purity exceeding 99.5% and a total yield surpassing 90%.
How to Synthesize Cefotaxime Efficiently
Implementing this solid-phase strategy requires precise adherence to the reaction parameters outlined in the patent to ensure optimal yield and purity profiles. The process begins with the preparation of the resin-bound 7-ACA, followed by the acylation with the side-chain active ester, and concludes with the acid-mediated cleavage and salt formation. Each stage demands careful monitoring of solvent composition, temperature, and reaction time to maintain the integrity of the cephalosporin core. While the conceptual framework is straightforward, the practical execution benefits from standardized protocols that minimize variability. Detailed standardized synthesis steps see the guide below for specific operational parameters.
- Bridge 7-ACA with chloromethyl resin in an organic solvent like DMF to immobilize the starting material onto the solid support.
- Perform acylation by reacting the resin-bound intermediate with AE-active ester in the presence of an organic base such as triethylamine.
- Execute cleavage and separation using a mixed acid solution to release the final cefotaxime product from the solid phase carrier.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid-phase synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of multiple purification and drying steps translates directly into a reduction of utility consumption and labor hours, driving down the overall cost of goods sold without compromising quality. Furthermore, the simplified workflow reduces the dependency on complex equipment setups, allowing for more flexible manufacturing scheduling and faster turnaround times between batches. This operational agility is crucial for maintaining supply continuity in a market often disrupted by raw material fluctuations or regulatory inspections. By adopting this technology, partners can secure a more stable source of high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant reduction in solvent usage and the complete removal of expensive purification media typically required in solution chemistry. By avoiding the need for extensive column chromatography or recrystallization cycles to remove impurities, the manufacturer saves substantially on material costs and waste treatment fees. Additionally, the higher overall yield means that less raw material is required to produce the same amount of final product, further enhancing the economic efficiency of the operation. These factors combine to create a leaner production model that offers substantial cost savings potential for downstream buyers.
- Enhanced Supply Chain Reliability: The robustness of the solid-phase method inherently reduces the risk of batch failures caused by crystallization issues or impurity spikes, which are common pain points in traditional cephalosporin manufacturing. With fewer unit operations involved, there are fewer points of failure in the production line, leading to more predictable lead times and delivery schedules. This reliability is essential for pharmaceutical companies managing just-in-time inventory systems where delays can have cascading effects on drug formulation and market availability. A supplier utilizing this technology can therefore offer a higher degree of assurance regarding order fulfillment and continuity of supply.
- Scalability and Environmental Compliance: Scaling up solid-phase reactions is often more straightforward than optimizing complex solution-phase crystallizations, as the reaction kinetics are less sensitive to mixing efficiencies at larger volumes. The process generates significantly less hazardous waste due to reduced solvent consumption and the absence of heavy metal catalysts or toxic reagents often found in alternative routes. This aligns with increasingly stringent global environmental regulations, reducing the compliance burden and associated costs for the manufacturing facility. Consequently, the technology supports sustainable growth and long-term viability for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this solid-phase synthesis technology. These insights are derived directly from the patent data to provide accurate information for decision-makers evaluating this manufacturing route. Understanding these specifics helps in assessing the feasibility of integrating this method into existing supply chains.
Q: How does solid-phase synthesis improve cefotaxime purity compared to traditional methods?
A: Solid-phase synthesis eliminates complex post-treatment purification steps required in solution chemistry, significantly reducing impurity profiles and achieving purity levels exceeding 99.5%.
Q: What are the key raw materials required for this patented synthesis route?
A: The process primarily utilizes 7-ACA as the core starting material, chloromethyl resin as the solid support, and AE-active ester for the side-chain introduction.
Q: Does this method offer advantages for large-scale commercial production?
A: Yes, by simplifying operation steps and reducing solvent consumption, the method enhances production efficiency and yield, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotaxime Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the solid-phase cefotaxime process can be seamlessly transitioned from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards for safety and efficacy. Our commitment to technical excellence ensures that our clients receive a product that is not only chemically superior but also consistently reliable.
We invite you to collaborate with us to explore how this patented technology can optimize your specific supply chain requirements. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate the tangible value of partnering with a leader in fine chemical innovation. Let us help you secure a competitive advantage through superior chemistry and dedicated service.
