Advanced Manufacturing of 17 Alpha-Hydroxy Progesterone via Magnesium-Lithium Complexation
Introduction to Advanced Steroid Intermediate Manufacturing
The pharmaceutical industry constantly seeks robust and scalable pathways for synthesizing critical hormonal intermediates, specifically 17 alpha-hydroxy progesterone and its analogues, which serve as pivotal precursors for various progestogen therapies. Patent CN102268058B introduces a transformative methodology that addresses long-standing inefficiencies in this synthetic landscape by replacing hazardous and costly reagents with a more sustainable magnesium-lithium complex system. This innovation not only enhances the stereoselectivity of the nucleophilic addition to the nitrile group but also simplifies the downstream purification processes required to meet rigorous pharmacopeial standards. By leveraging in-situ generation of reactive species, manufacturers can achieve yields ranging from 80% to 99.5% while maintaining exceptional control over impurity profiles. This technical breakthrough represents a significant leap forward for reliable pharmaceutical intermediates supplier networks aiming to optimize their production capabilities.
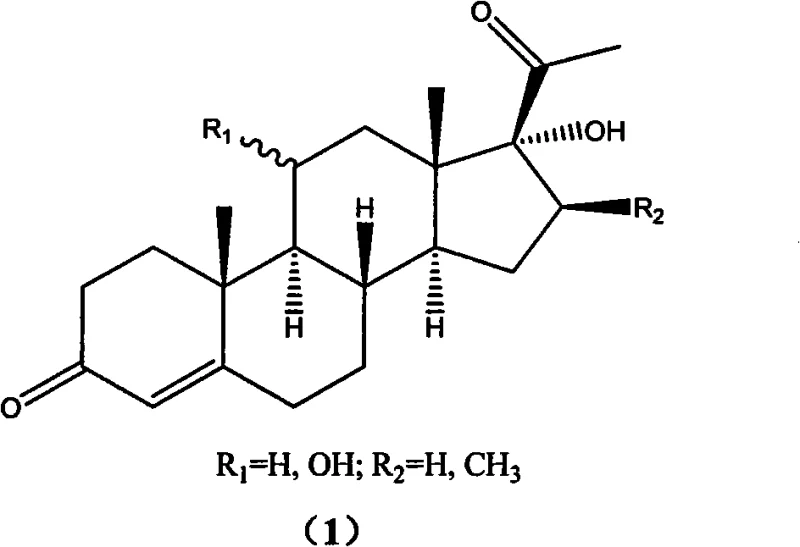
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 17 alpha-hydroxy progesterone analogues has relied heavily on the use of methyllithium (LiCH3) for the critical addition step to the 17-position nitrile group. As illustrated in the conventional reaction pathway, this approach necessitates the use of low-boiling solvents such as diethyl ether, which pose significant safety hazards including high flammability and volatility during large-scale operations. Furthermore, methyllithium is notoriously unstable and expensive, requiring stringent storage conditions that complicate supply chain logistics and increase overall operational expenditures. A major technical drawback of this legacy method is the propensity for configuration reversal at the C17 position, leading to the formation of undesired stereoisomers like compound (5), which complicates purification and reduces the overall yield of the desired therapeutic agent.
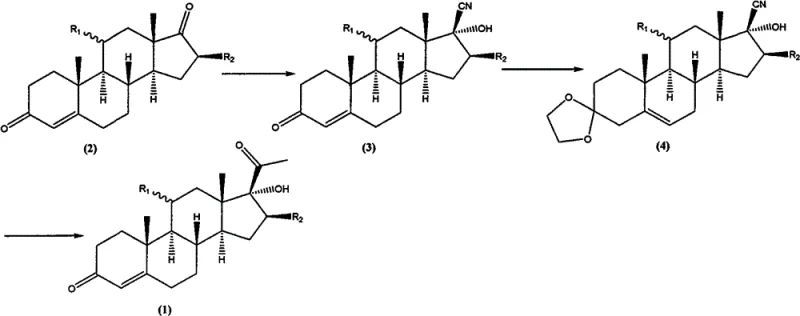
The Novel Approach
The patented methodology fundamentally reengineers this committed step by utilizing a combination of magnesium metal, lithium halide, and halomethane to generate a highly reactive yet stable organometallic complex in situ. Instead of relying on pre-formed methyllithium, the process generates a methylmagnesium halide species that is immediately complexed with lithium halide, creating a superior nucleophile for the addition reaction. This strategic shift eliminates the need for handling pyrophoric reagents and allows for the use of safer, higher-boiling solvents like tetrahydrofuran (THF) which are easier to recover and recycle. The result is a streamlined process that avoids the configuration reversal issues seen in prior art, ensuring that the stereochemical integrity of the steroid backbone is preserved throughout the transformation from the protected nitrile precursor to the final ketone product.
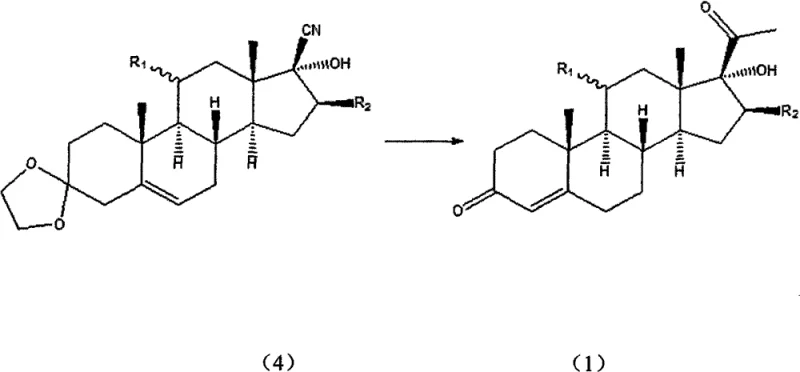
Mechanistic Insights into Magnesium-Lithium Complex Catalysis
The core of this technological advancement lies in the formation of a functionalized metal-complexing reagent, structurally represented as CH3MgX.LiY, where X and Y denote halogens such as chlorine or bromine. When magnesium metal reacts with halomethane in the presence of anhydrous lithium halide, the resulting species exhibits enhanced nucleophilicity and altered steric properties compared to standard Grignard reagents. The lithium cation acts as a Lewis acid coordinator, stabilizing the transition state during the attack on the nitrile carbon and facilitating a more ordered addition trajectory. This coordination effect is crucial for suppressing the formation of the configuration-reversed by-product, thereby directing the reaction exclusively towards the desired 17-alpha stereochemistry. Without the lithium halide additive, the reaction between the steroid nitrile and methyl Grignard reagent proceeds with poor selectivity or fails to react efficiently, highlighting the synergistic role of the bimetallic system.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions that typically arise from the high basicity of methyllithium. The milder nature of the magnesium-lithium complex reduces the risk of deprotonating sensitive positions on the steroid skeleton or attacking other electrophilic sites within the molecule. Consequently, the crude reaction mixture contains significantly fewer organic impurities, which simplifies the workup procedure involving acidic hydrolysis. The hydrolysis step, conducted at moderate temperatures between 20°C and 70°C, effectively cleaves the intermediate imine salt to release the target ketone without degrading the sensitive enone system in the A-ring. This precise control over reaction kinetics ensures that the final product meets the stringent purity specifications required for subsequent pharmaceutical formulation.
How to Synthesize 17 Alpha-Hydroxy Progesterone Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the metal components and the temperature profile during the addition phase to maximize the formation of the active complex. The process begins with the suspension of magnesium chips and lithium halide in a dry aprotic solvent, followed by the controlled introduction of the halomethane to initiate the exothermic formation of the organometallic species. Once the active reagent is generated, the protected steroid nitrile substrate is introduced, allowing the addition to proceed under mild thermal conditions that prevent thermal degradation. Detailed standardized synthetic steps see the guide below.
- Dissolve the protected steroid nitrile (Formula 4) in a solvent such as THF, then add magnesium metal chips and anhydrous lithium halide under inert atmosphere.
- Slowly add halomethane (e.g., methyl chloride or bromide) to the reaction mixture at controlled temperatures between -20°C and 20°C to generate the active metal complex in situ.
- Hydrolyze the reaction mixture with acidic material (e.g., hydrochloric acid) at 20-70°C, separate the organic phase, and purify via recrystallization to obtain the target product with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this magnesium-lithium based protocol offers substantial strategic benefits regarding cost stability and operational safety. By substituting expensive and import-dependent methyllithium with commodity-grade magnesium metal and simple alkyl halides, manufacturers can significantly reduce the raw material cost base associated with steroid intermediate production. The elimination of hazardous low-boiling solvents further lowers the costs related to specialized storage infrastructure and explosion-proof equipment, contributing to a leaner manufacturing overhead. Additionally, the robustness of the reaction conditions allows for greater flexibility in sourcing raw materials, as magnesium and lithium halides are widely available from multiple global suppliers, thereby mitigating the risk of supply disruptions.
- Cost Reduction in Manufacturing: The replacement of high-cost methyllithium with inexpensive magnesium turnings and methyl chloride or bromide results in a drastic reduction in reagent expenses per kilogram of product. Furthermore, the ability to recycle solvents like THF and the simplified purification process due to higher crude purity lead to lower waste disposal costs and reduced consumption of chromatography media. These factors combine to create a highly cost-effective manufacturing process that improves margin potential for high-volume steroid production without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing stable and non-pyrophoric reagents simplifies the logistics of raw material transportation and storage, removing the need for specialized cold-chain shipping or dangerous goods handling protocols. The improved stability of the reaction system also means that production campaigns can be run with greater consistency and fewer interruptions caused by reagent degradation or safety incidents. This reliability ensures a steady flow of high-purity pharmaceutical intermediates to downstream API manufacturers, supporting continuous production schedules and reducing lead times for finished drug products.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic heavy metal catalysts make this process inherently easier to scale from pilot plant to multi-ton commercial production facilities. The reduction in hazardous waste generation, particularly the avoidance of lithium salts in large quantities and volatile ether solvents, aligns with increasingly strict environmental regulations and corporate sustainability goals. This green chemistry approach not only minimizes the environmental footprint but also reduces the regulatory burden associated with waste treatment and emissions monitoring.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method for steroid intermediates. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners.
Q: Why is lithium halide added to the Grignard reaction?
A: The addition of anhydrous lithium halide forms a functionalized metal-complexing reagent (CH3MgX.LiY) which significantly improves reaction preference and yield compared to using methyl Grignard reagent alone, preventing the formation of by-products.
Q: How does this method improve safety over traditional methyllithium routes?
A: Traditional methods rely on expensive and unstable methyllithium which often requires hazardous low-boiling solvents like ether. This novel method uses stable magnesium metal and common halomethanes, drastically reducing fire risks and storage hazards suitable for industrial scale-up.
Q: What purity levels can be achieved with this synthesis route?
A: The process allows for the production of 17 alpha-hydroxy progesterone analogues with purity exceeding 99% after simple purification steps like recrystallization, meeting stringent pharmaceutical intermediate specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 17 Alpha-Hydroxy Progesterone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the magnesium-lithium complex method are seamlessly translated into robust industrial processes. We are committed to delivering high-purity steroid intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage these cost-saving technologies for your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can support your long-term strategic goals.
