Advanced One-Step Microbial Conversion for High-Purity 11-Beta-Hydroxy Steroids
Introduction to Breakthrough Biocatalytic Technology
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex corticosteroid intermediates, and patent CN105779555B presents a transformative approach to this challenge. This intellectual property details a novel method for preparing 11 beta-hydroxy-1, 4-diene-3, 20-diketone steroid compounds through a sophisticated one-step combined fermentation process. By leveraging the synergistic metabolic capabilities of Absidia coerulea AS3.65 and Arthrobacter simplex AS1.94, this technology effectively merges two critical biotransformation steps that were historically performed sequentially. For R&D directors and process chemists, this represents a significant leap forward in biocatalytic engineering, offering a route that simplifies the manufacturing landscape for essential anti-inflammatory drugs like prednisolone and methylprednisolone. The ability to introduce both the 11-beta-hydroxyl group and the 1,2-double bond functionality in a single reactor vessel addresses long-standing inefficiencies in steroid production, promising enhanced throughput and reduced operational complexity for large-scale facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial synthesis of 11-beta-hydroxy-1,4-diene steroids involves a disjointed, multi-stage workflow that is inherently resource-intensive and prone to yield losses. In the conventional paradigm, the C11 beta-hydroxylation reaction is first conducted using mold strains such as Absidia coerulea or Curvularia lunata, requiring specific fermentation conditions optimized solely for fungal growth. Once this step is complete, the product must be isolated, purified, and often chemically protected before being subjected to a second, distinct microbial fermentation using bacteria like Arthrobacter simplex to introduce the C1,2 double bond. This stepwise approach necessitates multiple sterilization cycles, extensive solvent usage for extraction and purification between steps, and significant downtime for equipment turnover. Furthermore, the cumulative yield is often compromised by material losses during each isolation phase, and the handling of intermediate compounds increases the risk of contamination and quality variability, creating a bottleneck for reliable high-purity pharmaceutical intermediates supply chains.
The Novel Approach
In stark contrast, the methodology disclosed in CN105779555B revolutionizes this workflow by enabling a concurrent biotransformation within a unified fermentation system. This innovative strategy employs a co-culture technique where Absidia coerulea and Arthrobacter simplex are inoculated into the same medium, allowing the C11 hydroxylation and C1,2 dehydrogenation reactions to proceed simultaneously. 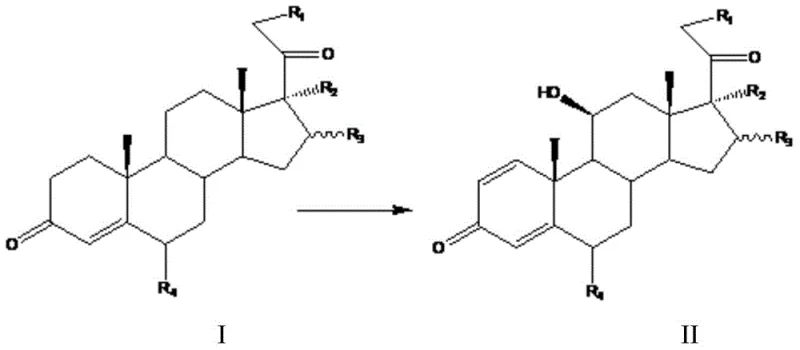 By eliminating the intermediate isolation step, this process drastically reduces the consumption of organic solvents like ethyl acetate and minimizes the energy expenditure associated with repeated heating, cooling, and filtration cycles. The result is a streamlined production line that not only accelerates the time-to-market for critical steroid APIs but also significantly lowers the environmental footprint of the manufacturing process. This consolidated approach ensures that the substrate is converted directly to the final diene-diketone structure with high efficiency, bypassing the logistical and chemical hurdles that plague traditional stepwise fermentation protocols.
By eliminating the intermediate isolation step, this process drastically reduces the consumption of organic solvents like ethyl acetate and minimizes the energy expenditure associated with repeated heating, cooling, and filtration cycles. The result is a streamlined production line that not only accelerates the time-to-market for critical steroid APIs but also significantly lowers the environmental footprint of the manufacturing process. This consolidated approach ensures that the substrate is converted directly to the final diene-diketone structure with high efficiency, bypassing the logistical and chemical hurdles that plague traditional stepwise fermentation protocols.
Mechanistic Insights into Combined Microbial Fermentation
The success of this combined fermentation hinges on the precise orchestration of enzymatic activities from two biologically distinct organisms with naturally divergent optimal growth conditions. Absidia coerulea, a fungus, typically thrives at lower temperatures around 25-28°C and acidic pH levels of 5.3-5.8 to express its 11-beta-hydroxylase enzyme effectively. Conversely, Arthrobacter simplex, a bacterium, prefers slightly higher temperatures of 30-34°C and neutral pH levels near 7.0-7.5 for optimal 1,2-dehydrogenase activity. The technical breakthrough lies in the identification of a "compromise window"—specifically a fermentation temperature of 28°C and a pH range of 5.5-6.5—where both strains retain sufficient metabolic viability and enzymatic specificity. Through rigorous optimization of the fermentation medium, which includes balanced ratios of glucose, peptone, corn steep liquor, and ammonium nitrate, the process ensures that neither organism outcompetes the other to the detriment of the reaction. This delicate balance prevents the formation of unwanted byproducts, such as the 14-alpha-hydroxyl impurities often associated with Curvularia strains, thereby ensuring a cleaner impurity profile for the final active pharmaceutical ingredient.
Furthermore, the substrate scope of this biocatalytic system is remarkably broad, accommodating various substituents at the R1, R2, R3, and R4 positions of the steroid nucleus. As illustrated in specific embodiments, the system successfully converts substrates like Compound 7A into methylprednisolone and Compound 15A into budesonide. 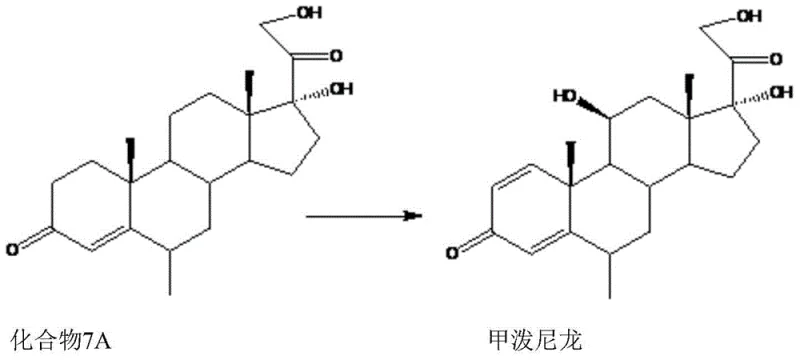 This versatility indicates that the enzyme systems involved possess a high degree of tolerance for different protecting groups, such as acetates or hydroxyls, at the C17 and C21 positions. The mechanism avoids the need for harsh chemical dehydrogenation reagents, relying instead on the regioselective power of microbial enzymes to introduce the conjugated diene system. This biological precision is crucial for maintaining the stereochemical integrity of the molecule, particularly at the chiral centers adjacent to the reaction sites, which is a key concern for R&D teams focused on the structural feasibility and purity of complex steroid intermediates.
This versatility indicates that the enzyme systems involved possess a high degree of tolerance for different protecting groups, such as acetates or hydroxyls, at the C17 and C21 positions. The mechanism avoids the need for harsh chemical dehydrogenation reagents, relying instead on the regioselective power of microbial enzymes to introduce the conjugated diene system. This biological precision is crucial for maintaining the stereochemical integrity of the molecule, particularly at the chiral centers adjacent to the reaction sites, which is a key concern for R&D teams focused on the structural feasibility and purity of complex steroid intermediates.
How to Synthesize 11-Beta-Hydroxy-1,4-Diene-3,20-Diketone Efficiently
Implementing this combined fermentation technology requires strict adherence to the optimized parameters defined in the patent to ensure consistent high-yield performance. The process begins with the preparation of a robust seed culture for both microorganisms, followed by their simultaneous introduction into a production fermenter containing the specially formulated medium. The key to success lies in maintaining the inoculation ratios—typically 15-20% for Arthrobacter simplex and 25-30% for Absidia coerulea—to establish a balanced microbial population from the outset. Detailed standardized synthetic steps see the guide below.
- Prepare a specialized fermentation medium containing glucose, peptone, corn steep liquor, ammonium nitrate, and monopotassium phosphate, adjusting the pH to a compromise range of 5.5 to 6.5 suitable for both fungal and bacterial growth.
- Inoculate the medium with seed cultures of Arthrobacter simplex (15-20% inoculum) and Absidia coerulea (25-30% inoculum) to initiate the combined biocatalytic transformation.
- Maintain fermentation at 28°C with controlled aeration and stirring for approximately 72 hours until substrate conversion is complete, followed by extraction and purification of the target steroid compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-step combined fermentation technology translates into tangible strategic advantages regarding cost structure and operational reliability. By collapsing two distinct manufacturing stages into a single unit operation, the process inherently reduces the capital expenditure required for additional fermentation tanks, centrifuges, and extraction equipment. The elimination of the intermediate isolation step means that vast quantities of organic solvents, which represent a significant variable cost and hazardous waste liability, are no longer required for washing and recrystallizing the hydrocortisone intermediate. This reduction in material usage directly correlates to a substantial decrease in the cost of goods sold (COGS), allowing for more competitive pricing in the global market for corticosteroid intermediates. Moreover, the simplified workflow reduces the dependency on complex logistics for moving semi-finished goods between different production lines or facilities, thereby minimizing the risk of supply chain disruptions.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the removal of unit operations rather than marginal yield improvements alone. By avoiding the separate sterilization, inoculation, and harvest cycles associated with the traditional two-step method, manufacturers can achieve significant savings in utility costs, including steam for sterilization and electricity for agitation and aeration. Additionally, the reduction in solvent consumption lowers the costs associated with solvent recovery systems and waste disposal compliance. This leaner manufacturing model allows for a more efficient allocation of resources, where the same production capacity can generate a higher volume of finished product without proportional increases in overhead, ultimately driving down the unit price for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: From a supply chain perspective, shortening the production cycle time is critical for responding to fluctuating market demands for essential medicines. The one-step fermentation process significantly compresses the lead time required to produce batch quantities of prednisolone and related steroids, as the total fermentation and processing time is reduced by nearly half compared to sequential methods. This agility enables suppliers to maintain lower safety stock levels while still meeting just-in-time delivery requirements for downstream API manufacturers. Furthermore, the robustness of the combined culture method against contamination—due to fewer open handling steps—ensures a higher rate of successful batch completion, guaranteeing a steady and uninterrupted flow of materials to customers who rely on consistent availability for their own drug formulation schedules.
- Scalability and Environmental Compliance: The process described in the patent has been validated at the 30L fermenter scale and is designed with principles that facilitate seamless commercial scale-up of complex biocatalytic reactions to industrial volumes. The use of common, non-toxic media components like corn steep liquor and glucose ensures that raw material sourcing remains stable and unaffected by geopolitical supply shocks. Environmentally, the drastic reduction in organic solvent waste aligns with increasingly stringent global regulations on pharmaceutical manufacturing emissions. By adopting this greener technology, companies can future-proof their operations against tightening environmental compliance standards, avoiding potential fines and reputational damage while appealing to eco-conscious stakeholders and partners in the sustainable chemistry movement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this combined fermentation technology. These insights are derived directly from the experimental data and comparative examples provided in patent CN105779555B, offering clarity on how this method outperforms legacy systems. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this biocatalytic route into their existing manufacturing portfolios.
Q: What is the primary advantage of the combined fermentation method over traditional stepwise processes?
A: The primary advantage is the consolidation of two distinct microbial transformations—C11 beta-hydroxylation and C1,2 dehydrogenation—into a single fermentation vessel. This eliminates the need for intermediate isolation, purification, and sterilization steps between reactions, drastically reducing solvent consumption, energy usage, and overall production time while improving total yield.
Q: How does the process manage the conflicting growth conditions of fungi and bacteria?
A: The process utilizes a carefully optimized medium formulation and environmental conditions that serve as a functional compromise. By maintaining a pH between 5.5 and 6.5 and a temperature around 28°C, the method ensures that Absidia coerulea retains sufficient 11-beta-hydroxylase activity while Arthrobacter simplex maintains effective 1,2-dehydrogenase activity, allowing both enzymes to function concurrently without significant loss of specificity.
Q: Can this technology be applied to steroids other than prednisolone precursors?
A: Yes, the patent demonstrates broad substrate tolerance. Beyond the primary conversion of RSA to prednisolone, the method is effective for synthesizing methylprednisolone from Compound 7A, budesonide from Compound 15A, and various other 11-beta-hydroxy-1,4-diene-3,20-diketone derivatives, making it a versatile platform for producing multiple high-value corticosteroid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prednisolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the combined fermentation technology described in CN105779555B for the production of high-value corticosteroids. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring the complex impurity profiles inherent in steroid fermentation, guaranteeing that every batch meets stringent purity specifications required by global regulatory bodies. We are committed to leveraging advanced biocatalytic strategies to deliver superior quality intermediates that empower our clients to bring life-saving medications to market faster and more cost-effectively.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to explore the full commercial potential of this one-step synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. We encourage you to reach out today to obtain specific COA data for our steroid intermediates and discuss detailed route feasibility assessments that demonstrate how our optimized fermentation processes can enhance your operational efficiency and profitability in the competitive landscape of hormone manufacturing.
