Advanced Manufacturing of Oxiracetam Intermediates via Optimized Cyclization Routes
Introduction to Next-Generation Oxiracetam Synthesis
The global demand for high-purity nootropic agents continues to surge, driven by an aging population and increasing awareness of cognitive health. At the forefront of this market is Oxiracetam, a potent racetam derivative known for its efficacy in treating senile dementia and memory disorders. However, traditional manufacturing methods have long been plagued by complex multi-step sequences, hazardous reagents, and inconsistent impurity profiles. A pivotal breakthrough in this domain is documented in Chinese Patent CN102134212B, which discloses a novel, streamlined preparation method starting from 4-chloroacetyl acetic ester. This patent represents a significant technological leap, offering a pathway that drastically simplifies the synthetic architecture while enhancing overall product quality. For R&D directors and procurement strategists, understanding this proprietary route is essential for securing a competitive edge in the supply of reliable pharmaceutical intermediates.
The core innovation lies in the strategic selection of starting materials and the optimization of reaction conditions to favor thermodynamic stability and kinetic efficiency. Unlike legacy processes that rely on unstable ketene dimers or explosive azides, this method leverages the robust chemistry of chloroacetyl derivatives. The result is a process that not only improves yield but also aligns with modern green chemistry principles by reducing waste and eliminating high-risk unit operations. As we delve deeper into the technical specifics, it becomes clear why this methodology is becoming the gold standard for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Oxiracetam has been fraught with significant technical and economic hurdles. Early methodologies, such as those described in JP62026267, utilized ketene dimers as starting materials. While conceptually direct, these routes suffered from notoriously low yields during the critical ring-closure step, leading to excessive raw material consumption and inflated production costs. Furthermore, alternative pathways like the one reported in US4118396 relied on iminodiacetic acid derivatives, necessitating lengthy protection and deprotection sequences that complicated the operational workflow and reduced total recovery rates.
Perhaps the most concerning legacy method involves the use of sodium azide, as seen in CN1268611C. This approach introduces severe safety liabilities due to the explosive nature of azide reagents, posing unacceptable risks in large-scale manufacturing environments. Additionally, the lack of hydroxyl protection in these older routes frequently leads to O-acylation byproducts during condensation steps. These impurities are structurally similar to the target molecule, making them exceptionally difficult to remove and ultimately compromising the purity specifications required for injectable grade APIs. The cumulative effect of these drawbacks is a supply chain vulnerable to disruption and quality failures.
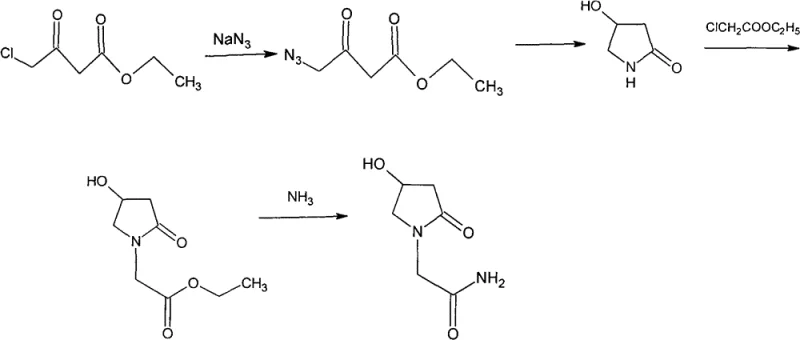
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method disclosed in CN102134212B offers a remarkably elegant solution. By initiating the synthesis with 4-chloroacetyl acetic ester, the process bypasses the need for hazardous azides and complex protecting groups entirely. The route proceeds through a logical sequence of ammonolysis, cyclization, alkylation, reduction, and final aminolysis. Each step is optimized for high conversion and ease of isolation, often relying on simple filtration or crystallization rather than energy-intensive chromatography. This structural simplicity translates directly into operational excellence, allowing manufacturers to achieve higher throughput with lower capital expenditure.
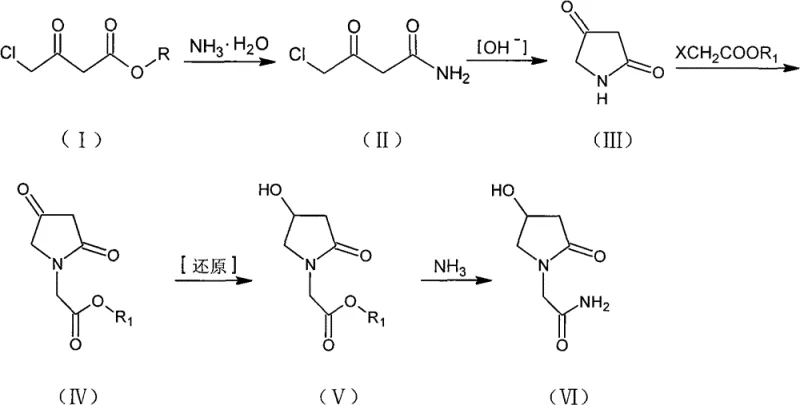
The visual representation of this novel pathway highlights its linearity and efficiency. The transformation from the chloro-ester to the final amide is achieved with minimal functional group manipulation, preserving the integrity of the pyrrolidine ring throughout the sequence. This approach not only mitigates the safety risks associated with azide chemistry but also effectively suppresses the formation of O-acylation impurities. For supply chain heads, this means a more predictable production schedule and a consistent supply of high-purity material that meets stringent regulatory standards without the need for extensive reprocessing.
Mechanistic Insights into Chemoselective Cyclization and Reduction
The success of this synthetic strategy hinges on precise control over reaction mechanisms, particularly during the cyclization and reduction phases. The initial ammonolysis of 4-chloroacetyl acetic ester is conducted at controlled low temperatures, typically between -10°C and 20°C, to prevent hydrolysis and dechlorination side reactions. Following this, the intramolecular cyclization to form pyrrolidine-2,4-dione is critically dependent on pH management. The patent specifies maintaining a pH between 8 and 10 using mild bases like potassium carbonate. This specific alkaline window is crucial; strongly alkaline conditions (pH > 11) promote polymerization side reactions that degrade the intermediate, while insufficient alkalinity fails to drive the nucleophilic substitution of the chloride by the amide nitrogen to completion.
Subsequent steps demonstrate exceptional chemoselectivity. During the N-alkylation phase, the use of sodium hydride in non-polar solvents like toluene ensures that deprotonation occurs exclusively at the imide nitrogen, avoiding unwanted O-alkylation. The reduction step is equally sophisticated, utilizing sodium borohydride to selectively reduce the 4-keto group to a hydroxyl group without affecting the pendant ester functionality. Experimental data from the patent indicates that this reduction can achieve yields approaching theoretical limits (e.g., 97.8% in specific embodiments) when performed in methanol at moderate temperatures. This level of selectivity is vital for maintaining a clean impurity profile, as it eliminates the need for complex downstream purification to remove over-reduced or unreacted species.
Furthermore, the final aminolysis step leverages the differential solubility of the product. By conducting the reaction in low-boiling organic solvents like methanol or ethanol, the resulting Oxiracetam, which has lower solubility in these media compared to the ester precursor, can be easily isolated. This mechanistic understanding allows process chemists to fine-tune conditions for maximum recovery, ensuring that the final API intermediate meets the rigorous purity demands of the pharmaceutical industry.
How to Synthesize Oxiracetam Efficiently
Implementing this advanced synthetic route requires adherence to specific operational parameters to maximize yield and safety. The process begins with the careful addition of 4-chloroacetyl acetic ester to concentrated aqueous ammonia under cooling, followed by a controlled cyclization in alcoholic solvents. The subsequent alkylation and reduction steps demand anhydrous conditions and precise stoichiometric control to prevent side reactions. Detailed standard operating procedures regarding reagent grades, addition rates, and workup protocols are essential for reproducible results. For a comprehensive breakdown of the standardized synthesis steps, please refer to the guide below.
- Perform ammonolysis on 4-chloroacetyl acetic ester using aqueous ammonia at 0-10°C to form 4-chloroacetyl acetamide.
- Execute intramolecular cyclization under alkaline conditions (pH 8-10) using potassium carbonate to generate pyrrolidine-2,4-dione.
- Conduct N-alkylation using sodium hydride and ethyl chloroacetate in toluene to form the ester intermediate.
- Reduce the 4-keto group selectively using sodium borohydride in methanol to yield the hydroxy-ester.
- Complete the synthesis via ammonolysis of the ester group in methanol to afford the final Oxiracetam product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers compelling economic and logistical benefits. The elimination of hazardous reagents like sodium azide removes a significant layer of regulatory compliance burden and insurance cost, while the use of commodity chemicals like 4-chloroacetyl acetic ester ensures a stable and cost-effective raw material base. The simplified workflow reduces the number of unit operations, which directly correlates to lower utility consumption and reduced labor hours per kilogram of product. These factors combine to create a manufacturing process that is not only cheaper to operate but also more resilient to market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this route is the avoidance of expensive and dangerous reagents. By replacing sodium azide with safe ammonolysis and utilizing common bases like potassium carbonate, the direct material costs are significantly lowered. Furthermore, the high selectivity of the reduction step minimizes the loss of valuable intermediates, thereby improving the overall mass balance and reducing the cost of goods sold. The ability to isolate products via simple crystallization rather than complex chromatography further drives down processing expenses, making the final API intermediate much more price-competitive.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of widely available starting materials. 4-chloroacetyl acetic ester and ethyl chloroacetate are bulk chemicals produced by numerous suppliers globally, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate a broad temperature range and do not require exotic catalysts, ensures that production can continue uninterrupted even if minor variations in utility supply occur. This stability is critical for maintaining continuous supply to downstream API manufacturers who operate on tight just-in-time schedules.
- Scalability and Environmental Compliance: From an environmental perspective, this route is markedly superior. The absence of heavy metal catalysts and explosive azides simplifies waste treatment and disposal, lowering the environmental compliance costs associated with hazardous waste management. The process generates fewer byproducts, meaning less solvent is required for purification, which reduces the facility's overall solvent footprint. These attributes make the technology highly scalable, allowing for seamless expansion from pilot plant batches to multi-ton commercial production without encountering the safety or waste disposal barriers that limit older technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical disclosures found within the patent literature, providing a factual basis for decision-making. Understanding these nuances is key for R&D teams evaluating process transfer and for procurement teams assessing supplier capabilities.
Q: Why is the 4-chloroacetyl acetic ester route superior to the sodium azide method?
A: The sodium azide method involves hazardous explosives and often produces O-acylation byproducts that contaminate the final API. The patented route avoids azides entirely, utilizing safer ammonolysis and cyclization steps that inherently minimize toxic impurities and simplify purification.
Q: How does the new process control impurities during the cyclization step?
A: The process strictly controls the pH between 8 and 10 using mild bases like potassium carbonate. This prevents the strong alkaline conditions that typically cause polymerization side reactions, ensuring a cleaner pyrrolidine-2,4-dione intermediate with higher crystallinity.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the route utilizes common solvents like methanol, ethanol, and toluene, and reagents like sodium borohydride which are readily available at industrial scales. The reaction conditions are mild (often room temperature to reflux), making it highly suitable for large-scale manufacturing without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxiracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving needs of the global pharmaceutical market. Our technical team has extensively analyzed the route disclosed in CN102134212B and possesses the expertise to implement this high-efficiency process at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Oxiracetam intermediate we deliver meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and reliability for your organization.
