Advanced Manufacturing of Oxiracetam: A Cost-Effective Route for Pharmaceutical Intermediates
Introduction to Novel Oxiracetam Manufacturing Technology
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for producing nootropic agents, with Oxiracetam standing out as a critical compound for cognitive enhancement therapies. The patent CN101693685A, published in April 2010, discloses a groundbreaking method for preparing 4-hydroxylethylpyrrolidone-2-acetamide, commonly known as Oxiracetam, which addresses significant bottlenecks in traditional synthesis routes. This innovative approach leverages chloroacetoacetic ester and aminoacetonitrile hydrochloride as primary feedstocks, executing a sequence of hydrogenation, substitution, and cyclization reactions to achieve the target molecule with superior efficiency. By shifting away from complex halogenated acid derivatives, this technology offers a streamlined pathway that enhances both product quality and process economics, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to optimize their portfolios.
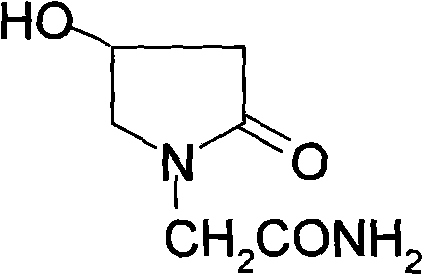
The strategic value of this patent lies in its ability to bypass the tedious purification steps often required in legacy methods, thereby reducing the overall environmental footprint and operational expenditure. For R&D directors and procurement managers alike, understanding the nuances of this four-step synthesis is crucial for evaluating potential partnerships and securing a stable supply of high-purity active pharmaceutical ingredients. The method not only promises higher yields but also ensures that the final product meets stringent medicinal standards, a prerequisite for any serious player in the global nootropics market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industry largely relied on methods such as those described in Japanese Patent JP62026267, which utilized 3-hydroxy-4-halo butanoic acid derivatives reacting directly with glycine amide equivalents. While conceptually straightforward, this conventional approach suffers from severe practical drawbacks that hinder commercial viability. The reaction profile is plagued by numerous side reactions, leading to a final product mixture that is incredibly complex and difficult to separate. Consequently, even with meticulous purification efforts, the resulting purity often fails to reach the rigorous requirements necessary for pharmaceutical applications. Furthermore, the reaction kinetics are sluggish, necessitating reaction times exceeding 20 hours, which drastically reduces throughput and increases energy consumption. The ultimate yield in these legacy processes is notoriously low, creating a supply bottleneck that cannot satisfy the growing demands of commercial scale production for cognitive health medications.
The Novel Approach
In stark contrast, the novel approach detailed in CN101693685A introduces a paradigm shift by utilizing chloroacetoacetic ester as the foundational building block. This route is characterized by its operational simplicity and the use of readily available, low-cost raw materials that are abundant in the global chemical market. The process is divided into distinct, manageable stages: an initial reduction to form a chloro-hydroxy ester, followed by a substitution reaction with aminoacetonitrile, and finally a cyclization and hydrolysis sequence. This modular design allows for precise control over each transformation, minimizing the formation of unwanted by-products and simplifying downstream processing. Compared with the prior art, this method boasts a significantly higher total recovery rate and superior product quality, making it far more suitable for the industrialized scale production required by modern supply chains. The elimination of prolonged reaction times and complex purification protocols translates directly into enhanced manufacturing efficiency and reduced lead times for high-purity pharmaceutical intermediates.
Mechanistic Insights into Borohydride-Mediated Reduction and Cyclization
The core of this synthesis strategy relies on a carefully orchestrated sequence of organic transformations, beginning with the selective reduction of the ketone functionality in chloroacetoacetic ester. 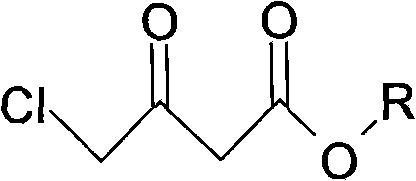
In the first critical step, chloroacetoacetic ester is subjected to reduction using a hydroborate agent, specifically potassium borohydride, within an alcoholic solvent system. The reaction is conducted under strictly controlled low-temperature conditions, ranging from -5°C to 15°C, with optimal results observed between 3°C and 8°C. This thermal regulation is vital to prevent the reduction of the ester group or other undesirable side reactions, ensuring high chemoselectivity for the hydroxyl group formation. The molar ratio of the hydroborate to the ester is maintained between 1:4 and 1:5, providing a slight excess of reducing power to drive the reaction to completion while maintaining economic efficiency. The result is the formation of a chloro-hydroxy butyrate intermediate, which serves as the scaffold for the subsequent nitrogen incorporation.
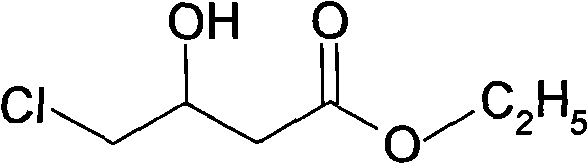
Following the reduction, the process moves to a nucleophilic substitution where the chloride leaving group is displaced by the amino group of aminoacetonitrile hydrochloride or sulfate. This step occurs at elevated temperatures between 60°C and 100°C, facilitating the necessary activation energy for the substitution to proceed efficiently. The resulting intermediate then undergoes an intramolecular cyclization, driven by the nucleophilic attack of the nitrogen on the ester carbonyl, forming the pyrrolidone ring structure. Finally, the nitrile group is hydrolyzed under mild alkaline conditions at 40°C to 60°C to yield the acetamide side chain. This mechanistic pathway effectively constructs the complex heterocyclic core of Oxiracetam in a linear, high-yielding fashion, demonstrating a sophisticated understanding of functional group compatibility and reaction kinetics.
How to Synthesize Oxiracetam Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to maximize yield and purity, as outlined in the patent embodiments. The process begins with the preparation of the chloro-hydroxy intermediate, followed by the substitution and cyclization steps which can be performed sequentially in the same solvent system to minimize handling losses. The final hydrolysis step converts the nitrile precursor into the desired amide, completing the molecular architecture of the nootropic agent. For detailed standard operating procedures and exact stoichiometric calculations, please refer to the standardized synthesis guide below which breaks down the laboratory-scale protocol into actionable manufacturing steps.
- Reduce chloroacetoacetic ester with potassium borohydride at low temperature (-5 to 15°C) to form chloro-hydroxy butyrate.
- Perform nucleophilic substitution with aminoacetonitrile hydrochloride in alcoholic solvent at 60-100°C.
- Execute cyclization and subsequent alkaline hydrolysis at 40-60°C to yield the final 4-hydroxy-2-pyrrolidone-2-acetamide product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads in the fine chemical sector. The primary driver of value is the utilization of chloroacetoacetic ester and aminoacetonitrile salts, which are commodity chemicals with stable pricing and widespread availability. This reliance on basic feedstocks mitigates the risk of supply disruptions often associated with specialized or exotic reagents, ensuring a continuous flow of materials for production schedules. Furthermore, the simplicity of the technical process means that it can be executed in standard stainless steel reactors without the need for high-pressure equipment or exotic catalysts, significantly lowering the barrier to entry for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the elimination of expensive transition metal catalysts and the reduction of energy-intensive long-duration reactions. By shortening the reaction time from over 20 hours in legacy methods to a fraction of that time, manufacturers can achieve substantial cost savings in utilities and labor. Additionally, the higher total recovery rate implies that less raw material is wasted per kilogram of finished product, directly improving the cost of goods sold (COGS). The avoidance of complex purification sequences further reduces the consumption of solvents and adsorbents, contributing to a leaner and more cost-efficient production model that enhances overall profitability.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route translates into greater reliability for the supply chain, as the process is less sensitive to minor fluctuations in reaction conditions compared to the finicky prior art. The use of common alcoholic solvents like ethanol or methanol simplifies logistics and solvent recovery, reducing the dependency on specialized supply lines. This operational stability allows for more accurate forecasting of production output, enabling suppliers to commit to tighter delivery windows and maintain consistent inventory levels for their clients. For global buyers, this means a reduced risk of stockouts and a more predictable procurement cycle for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scalability is inherent in the design of this process, as the reaction conditions are mild and the exotherms are manageable, allowing for safe scale-up from pilot plants to multi-ton commercial facilities. The simplified workflow generates less hazardous waste and consumes fewer resources per unit of product, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The ability to produce high-purity material with minimal by-product formation reduces the burden on waste treatment facilities, making this method not only commercially attractive but also environmentally responsible. This dual benefit positions manufacturers adopting this technology as preferred partners for eco-conscious pharmaceutical companies seeking green chemistry solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Oxiracetam using this patented methodology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: What are the primary advantages of this Oxiracetam synthesis method over prior art?
A: This method utilizes cheap and easily obtainable raw materials like chloroacetoacetic ester, avoiding the complex side reactions and low yields associated with older methods such as JP62026267. It offers a significantly shorter reaction time and higher total recovery rates suitable for industrial production.
Q: What are the critical reaction conditions for the reduction step?
A: The reduction of chloroacetoacetic ester requires strict temperature control between -5°C and 15°C, optimally 3-8°C, using potassium borohydride as the reducing agent in an alcoholic solvent to ensure high selectivity and yield.
Q: Is this process scalable for commercial manufacturing?
A: Yes, the process is designed for commercial scale-up with simple operational steps, standard equipment requirements, and the use of common solvents like ethanol, making it highly viable for large-scale pharmaceutical intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxiracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthesis routes like CN101693685A in delivering high-quality nootropic agents to the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Oxiracetam produced adheres to the highest international standards for pharmaceutical use. We are committed to leveraging our technical expertise to optimize this cost-effective route, delivering value through both superior product quality and operational excellence.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to explore the full commercial potential of this technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific rigor and commercial reliability.
