Revolutionizing Ulipristal Acetate Production: A Novel Synthetic Route for Commercial Scale-Up
The pharmaceutical landscape for emergency contraception and gynecological treatments has been significantly shaped by the development of selective progesterone receptor modulators, most notably Ulipristal Acetate. A pivotal advancement in the manufacturing of this critical active pharmaceutical ingredient is detailed in patent CN102875629A, which outlines a robust and innovative synthetic methodology. This patent addresses the longstanding challenges associated with the production of complex steroid intermediates, offering a pathway that is not only chemically elegant but also industrially viable. By leveraging a unique sequence involving tosylmethyl isocyanide and controlled oxidation states, the disclosed method circumvents the hazardous reagents and extreme conditions that have historically plagued the synthesis of 19-norpregna derivatives. For global procurement leaders and technical directors, understanding this technological shift is essential for securing a stable supply of high-purity hormonal APIs. The transition from legacy processes to this novel approach represents a substantial opportunity for cost optimization and risk mitigation in the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ulipristal Acetate and its precursors has relied on methodologies that present severe safety and environmental liabilities. As illustrated in the traditional reaction pathways, early methods often utilized osmium tetroxide as a primary oxidant for introducing oxygen functionality into the steroid backbone. 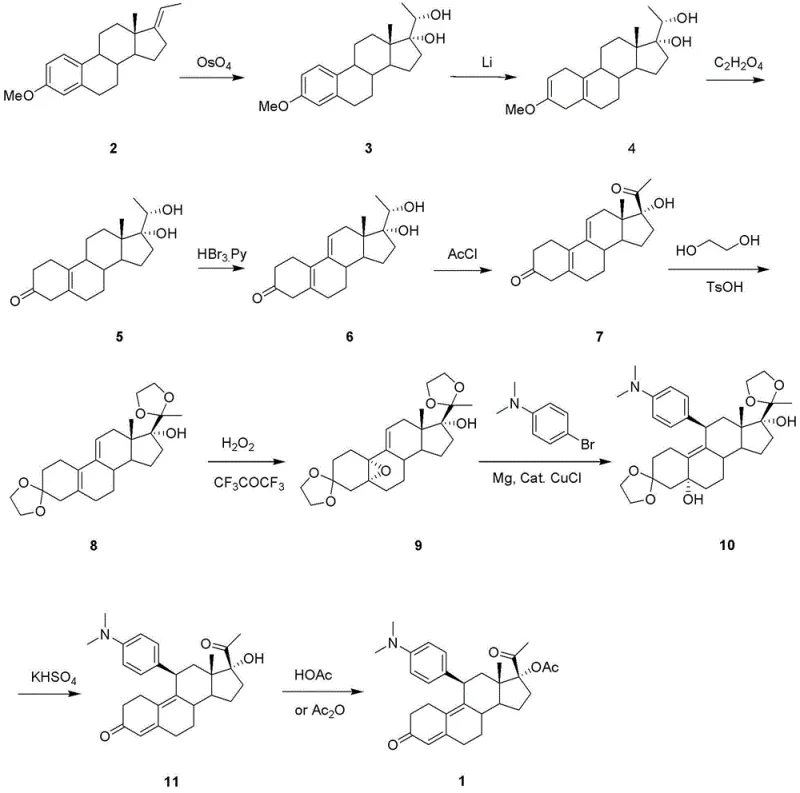 This reagent is notoriously toxic, volatile, and expensive, requiring stringent safety protocols and specialized waste treatment facilities that drive up operational expenditures. Furthermore, alternative routes described in prior art, such as those utilizing lithium metal in conjunction with 4,4'-di-tert-butylbiphenyl, necessitate cryogenic conditions reaching temperatures as low as -70°C. Maintaining such extreme thermal environments on a commercial scale imposes a massive energy burden and introduces significant engineering complexities regarding reactor design and heat transfer. These conventional approaches also frequently suffer from low overall yields due to the multiplicity of steps and the instability of intermediates under harsh reaction conditions, leading to inconsistent batch quality and prolonged production cycles that fail to meet modern supply chain demands.
This reagent is notoriously toxic, volatile, and expensive, requiring stringent safety protocols and specialized waste treatment facilities that drive up operational expenditures. Furthermore, alternative routes described in prior art, such as those utilizing lithium metal in conjunction with 4,4'-di-tert-butylbiphenyl, necessitate cryogenic conditions reaching temperatures as low as -70°C. Maintaining such extreme thermal environments on a commercial scale imposes a massive energy burden and introduces significant engineering complexities regarding reactor design and heat transfer. These conventional approaches also frequently suffer from low overall yields due to the multiplicity of steps and the instability of intermediates under harsh reaction conditions, leading to inconsistent batch quality and prolonged production cycles that fail to meet modern supply chain demands.
The Novel Approach
In stark contrast to the cumbersome legacy techniques, the method disclosed in CN102875629A introduces a streamlined strategy that prioritizes operational simplicity and chemical efficiency. The core innovation lies in the initial functionalization of the steroid nucleus using tosylmethyl isocyanide (TosMIC) under alkaline conditions, which effectively installs the necessary nitrogen-containing handle without the need for hazardous cyanide sources. This is followed by a controlled Grignard addition and a sophisticated oxidation step using trialkyl phosphite in an oxygenated environment, which proceeds at moderate temperatures around -20°C to room temperature. 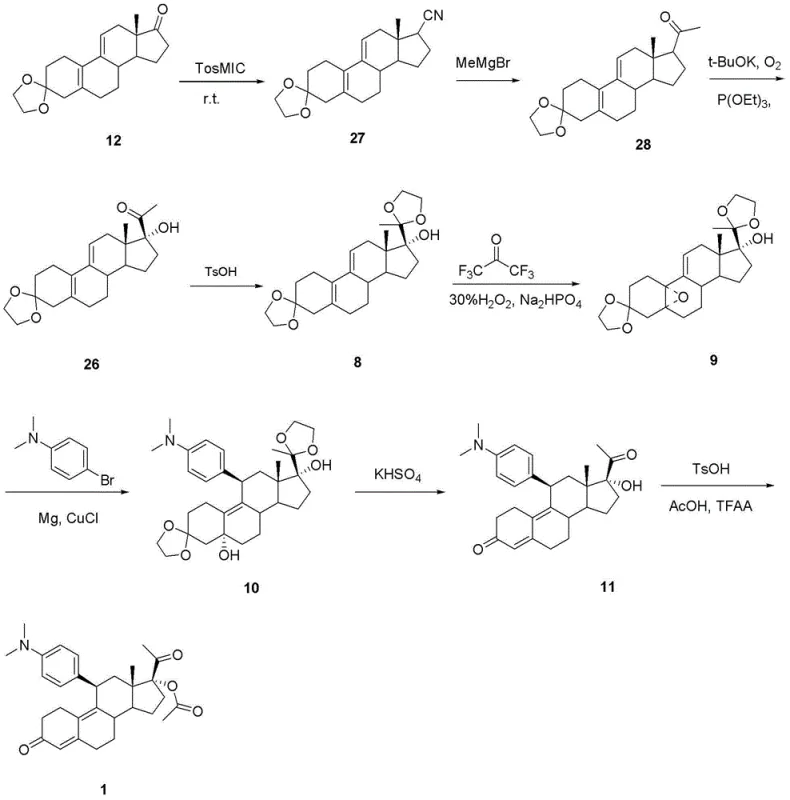 By eliminating the requirement for cryogenic lithium reductions and toxic heavy metal oxidants, this new route drastically simplifies the post-reaction workup and purification processes. The result is a synthetic pathway that is not only safer for personnel and the environment but also offers superior scalability, allowing manufacturers to transition from laboratory benchtop quantities to multi-ton commercial production with minimal process re-engineering. This shift enables a more reliable pharmaceutical intermediates supplier to maintain consistent output levels regardless of market fluctuations.
By eliminating the requirement for cryogenic lithium reductions and toxic heavy metal oxidants, this new route drastically simplifies the post-reaction workup and purification processes. The result is a synthetic pathway that is not only safer for personnel and the environment but also offers superior scalability, allowing manufacturers to transition from laboratory benchtop quantities to multi-ton commercial production with minimal process re-engineering. This shift enables a more reliable pharmaceutical intermediates supplier to maintain consistent output levels regardless of market fluctuations.
Mechanistic Insights into TosMIC-Mediated Steroid Functionalization
The mechanistic elegance of this synthesis is rooted in the versatile reactivity of the TosMIC reagent, which serves as a masked acyl anion equivalent. In the initial step, the deprotonated TosMIC attacks the C17 ketone of the protected steroid starting material, forming an intermediate that subsequently eliminates the tosyl group to yield the 17-cyano derivative. This transformation is critical as it establishes the carbon framework required for the subsequent side-chain elaboration while maintaining the stereochemical integrity of the steroid nucleus. The reaction is conducted in a glycol dimethyl ether solvent system with potassium tert-butoxide as the base, ensuring a homogeneous reaction mixture that facilitates efficient mass transfer. Following the formation of the nitrile, the introduction of a methyl group via methyl magnesium bromide proceeds through a nucleophilic addition to the cyano group, generating an imine salt that is hydrolyzed to the corresponding methyl ketone. This sequence avoids the use of organolithium reagents which are prone to side reactions and require strict moisture exclusion, thereby enhancing the robustness of the process.
Further downstream, the conversion of the methyl ketone to the 17-alpha-hydroxy-20-ketone motif is achieved through an oxidative rearrangement mediated by trialkyl phosphite and molecular oxygen. This step is particularly noteworthy for its ability to introduce the oxygen functionality at the C17 position with high regioselectivity, a transformation that is often difficult to achieve without affecting other sensitive sites on the molecule. The reaction conditions are meticulously controlled, utilizing a mixed solvent system of DMF and tert-butanol to solubilize both the organic substrate and the inorganic oxidants. The subsequent protection of the C20 carbonyl as an ethylene ketal ensures that the ketone remains inert during the epoxidation and Grignard addition steps that follow. This strategic use of protecting groups, combined with the mild oxidation protocol, minimizes the formation of by-products and impurities, resulting in a crude product profile that is significantly cleaner than that obtained from conventional routes. Such purity is paramount for meeting the stringent specifications required for high-purity OLED material or pharmaceutical grade intermediates.
How to Synthesize Ulipristal Acetate Efficiently
The implementation of this synthetic route requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of the 17-cyano intermediate, followed by the Grignard addition to form the methyl ketone, and concludes with the oxidative functionalization and final deprotection steps. Each stage has been optimized to balance reaction kinetics with safety considerations, ensuring that the process is amenable to standard chemical manufacturing equipment. The detailed standardized synthesis steps see the guide below for specific molar ratios and temperature profiles that have been validated to ensure reproducibility.
- React 3,3-(ethylenedioxy)estra-5(10),9(11)-dien-17-one with tosylmethyl isocyanide under alkaline conditions to form the 17-cyano derivative.
- Treat the cyano intermediate with methyl magnesium bromide to generate the methyl ketone functionality.
- Perform oxidation using trialkyl phosphite in an oxygenated alkaline environment to yield the 17-alpha-hydroxy-20-ketone structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic methodology translates directly into tangible business benefits and risk reduction. The primary advantage lies in the substantial cost savings achieved by eliminating the need for exotic and hazardous reagents such as osmium tetroxide and cryogenic lithium. By substituting these with commercially abundant and inexpensive chemicals like TosMIC and trialkyl phosphites, the raw material cost base is significantly lowered, allowing for more competitive pricing structures in the final API market. Moreover, the simplification of the process workflow reduces the consumption of utilities such as energy for cooling and heating, further driving down the manufacturing overheads. This cost reduction in steroid manufacturing is not merely theoretical but is grounded in the fundamental chemistry of the route, which requires fewer unit operations and less complex waste treatment infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts and cryogenic reagents removes a major cost driver from the production budget. Traditional methods often require specialized containment and disposal procedures for toxic waste, which incur significant regulatory compliance costs. By shifting to a greener chemistry platform, manufacturers can avoid these penalties and reduce the overall cost of goods sold. Additionally, the higher yields observed in key steps mean that less starting material is wasted, improving the atom economy of the process. This efficiency gain allows for a more favorable margin structure, enabling suppliers to offer better value to their clients while maintaining profitability in a competitive market.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials ensures that production schedules are not disrupted by shortages of niche reagents. Reagents like TosMIC and common Grignard solutions are stocked by multiple global vendors, reducing the risk of single-source dependency. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to variations in ambient temperature or minor fluctuations in reagent quality. This stability is crucial for maintaining continuous production runs and meeting tight delivery windows. Reducing lead time for high-purity hormonal APIs becomes achievable when the manufacturing process is decoupled from complex and fragile operational requirements, ensuring a steady flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: The mild reaction conditions facilitate a smoother transition from pilot plant to full-scale commercial production. Processes that operate at ambient or moderately elevated temperatures are easier to control in large reactors, minimizing the risk of thermal runaways or hot spots. This inherent safety profile aligns with increasingly stringent environmental regulations, positioning the manufacturer as a responsible partner in the pharmaceutical supply chain. The reduction in hazardous waste generation simplifies the permitting process for new manufacturing lines and reduces the long-term liability associated with environmental remediation. Consequently, this route supports the commercial scale-up of complex steroid intermediates with a lower environmental footprint, appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. They are derived from the specific advantages and operational details outlined in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their own production needs or for sourcing partners who can execute it effectively.
Q: How does the new synthetic route improve safety compared to conventional methods?
A: The novel method eliminates the use of highly toxic osmium tetroxide and cryogenic lithium reductions, replacing them with safer TosMIC chemistry and moderate temperature Grignard reactions.
Q: What are the yield advantages of this patented process?
A: The process demonstrates high efficiency, with key steps achieving yields such as 75% for the cyano formation and 80% for the methyl ketone conversion, significantly outperforming older multi-step routes.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the route avoids extreme conditions like -70°C and uses readily available reagents, making it highly scalable and cost-effective for commercial manufacturing of steroid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ulipristal Acetate Supplier
The technical potential of the synthetic route described in CN102875629A is immense, offering a clear path toward more efficient and sustainable production of this vital medication. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovative chemistry to life. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of intermediate or API meets the highest international standards. We understand the critical nature of supply continuity in the pharmaceutical sector and have built our operations to prioritize reliability and quality above all else, making us a trusted partner for long-term projects.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this route impacts your bottom line. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your volume requirements. Our team is ready to provide the technical support and commercial flexibility needed to secure your position in the market with high-quality, cost-effective solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
