Advanced Manufacturing of Ulipristal Acetate: Technical Breakthroughs and Commercial Scalability
The pharmaceutical landscape for emergency contraception has been significantly transformed by the introduction of Ulipristal Acetate, a selective progesterone receptor modulator with superior efficacy profiles. As detailed in the pivotal patent CN102942612A, recent advancements in synthetic methodology have addressed long-standing challenges regarding yield, purity, and industrial safety associated with this critical active pharmaceutical ingredient. This technical insight report analyzes the novel synthetic pathway that circumvents the use of hazardous reagents while enhancing overall process efficiency, offering a robust solution for global supply chains. The molecular architecture of Ulipristal Acetate, characterized by its specific stereochemistry at the C11 and C17 positions, demands precise chemical control to ensure therapeutic consistency and regulatory compliance across international markets. 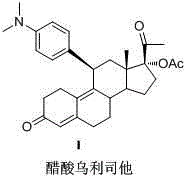
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Ulipristal Acetate relied on synthetic routes that presented substantial operational risks and economic inefficiencies, as exemplified by earlier patents such as US5929262A. These legacy methodologies frequently necessitated the utilization of acetone cyanohydrin, a reagent classified as highly toxic and strictly controlled, thereby imposing severe regulatory burdens and safety protocols on manufacturing facilities. Furthermore, the reliance on flammable metallic lithium and expensive chloromethyl chlorosilane introduced significant cost volatility and fire hazards that are untenable for modern large-scale chemical production environments. The structural complexity of the steroid backbone often led to poor selectivity during key addition reactions, resulting in difficult-to-separate impurities that compromised the final purity profile and increased downstream processing costs. 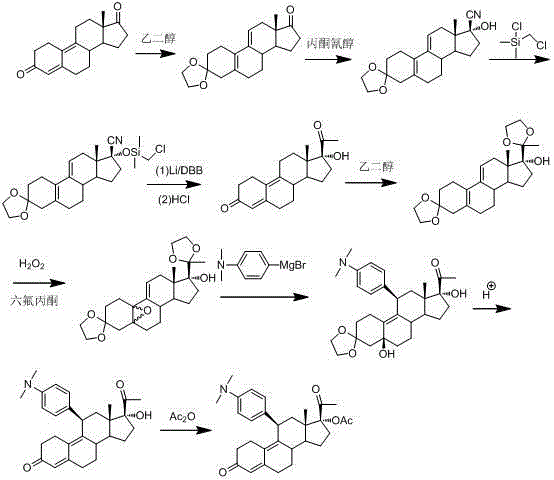
The Novel Approach
In stark contrast, the innovative methodology disclosed in CN102942612A introduces a strategically redesigned synthetic sequence that prioritizes both safety and atom economy without compromising on yield. By replacing hazardous cyanohydrin derivatives with safer peroxide-based oxidation systems and eliminating the need for pyrophoric metals, the new process drastically reduces the environmental footprint and operational risk profile of the manufacturing plant. The strategic implementation of catalytic systems allows for milder reaction conditions, which not only preserves the integrity of sensitive functional groups but also minimizes energy consumption associated with extreme temperature controls. This paradigm shift in process chemistry enables manufacturers to achieve higher throughput rates while maintaining stringent quality standards required for pharmaceutical intermediates destined for regulated markets.
Mechanistic Insights into CuCl-Catalyzed Grignard Addition
The cornerstone of this enhanced synthetic route lies in the sophisticated application of transition metal catalysis to govern regioselectivity during the carbon-carbon bond-forming steps. Specifically, the inclusion of cuprous chloride (CuCl) during the Grignard addition of 4-dimethylaminophenylmagnesium bromide serves as a critical control element that directs the nucleophilic attack preferentially towards the C17 carbonyl moiety. This catalytic intervention effectively mitigates the steric hindrance issues that typically plague steroid synthesis, ensuring that the desired alpha-configuration is obtained with high fidelity and minimal formation of beta-isomer byproducts. The mechanistic pathway involves the formation of an organocopper intermediate which is less basic and more nucleophilic than the corresponding Grignard reagent, thereby preventing unwanted side reactions at other electrophilic sites within the complex molecular framework. 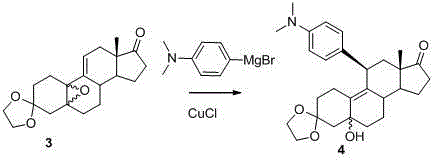
Furthermore, the initial epoxidation step utilizing hydrogen peroxide in the presence of hexafluoroacetone demonstrates a remarkable ability to establish the necessary oxygenation pattern with exceptional diastereoselectivity. The reaction conditions, maintained between -20°C and 40°C under alkaline environments, facilitate the formation of the 5α,10α-epoxy intermediate which is crucial for the subsequent stereochemical outcomes of the synthesis. This precise control over the oxidation state prevents the formation of over-oxidized species or ring-opened degradation products that could act as genotoxic impurities in the final drug substance. The robustness of this oxidative protocol ensures that the process remains scalable, as the exothermic nature of peroxide reactions is effectively managed through the specific solvent systems and dosing strategies outlined in the patent documentation.
How to Synthesize Ulipristal Acetate Efficiently
The execution of this novel synthesis requires strict adherence to the optimized parameters regarding temperature, stoichiometry, and workup procedures to maximize the recovery of the target compound. The process begins with the careful preparation of the epoxy intermediate, followed by the catalytic Grignard addition which sets the critical stereocenters required for biological activity. Subsequent steps involving ethynylation and deprotection are designed to be telescoped where possible, reducing the number of isolation events and thereby minimizing material loss and solvent usage throughout the production campaign. Detailed standardized operating procedures for each transformation step are essential to ensure batch-to-batch consistency and compliance with current Good Manufacturing Practices (cGMP).
- Perform selective epoxidation of the starting steroid material using hydrogen peroxide and hexafluoroacetone under alkaline conditions to form the key epoxy intermediate.
- Execute a highly regioselective Grignard addition using 4-dimethylaminophenylmagnesium bromide catalyzed by cuprous chloride to introduce the aryl group at the C11 position.
- Complete the synthesis through ethynylation, deprotection, and final acetylation steps to obtain high-purity Ulipristal Acetate suitable for pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this novel synthetic route offers compelling advantages that directly address the core concerns of cost stability and supply continuity for high-value pharmaceutical intermediates. By eliminating the dependency on scarce or heavily regulated raw materials such as acetone cyanohydrin, manufacturers can secure a more resilient supply chain that is less susceptible to geopolitical disruptions or sudden regulatory bans. The simplification of the purification process, driven by higher reaction selectivity, translates into reduced consumption of chromatography media and solvents, which are often significant cost drivers in the production of complex steroid molecules.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous reagents inherently lowers the raw material cost base, while the improved yield profile reduces the effective cost per kilogram of the finished active ingredient. Qualitative analysis suggests that the elimination of heavy metal catalysts removes the need for costly scavenging steps and extensive testing for residual metals, further streamlining the production budget. Additionally, the higher overall yield means that less starting material is required to produce the same amount of final product, optimizing the utilization of capital-intensive reactor capacity.
- Enhanced Supply Chain Reliability: The use of commercially abundant and stable reagents ensures that production schedules are not dictated by the lead times of specialty chemicals with limited supplier bases. This accessibility allows for better inventory management and the ability to ramp up production quickly in response to market demand fluctuations without facing bottlenecks in raw material sourcing. The robustness of the chemical process also reduces the likelihood of batch failures, ensuring a consistent flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by reducing waste generation and avoiding the use of substances of very high concern (SVHC). This alignment with environmental regulations facilitates smoother regulatory approvals and reduces the liability associated with waste disposal and emissions monitoring in manufacturing jurisdictions. The scalability is further supported by the use of common solvents and standard reaction conditions that do not require specialized equipment beyond typical pharmaceutical manufacturing infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology in an industrial setting. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature to assist decision-makers in evaluating the feasibility of adoption. Understanding these nuances is critical for aligning technical capabilities with business objectives in the competitive pharmaceutical market.
Q: How does the new synthesis method improve safety compared to prior art?
A: The novel method eliminates the use of highly toxic reagents like acetone cyanohydrin and flammable metals such as lithium, significantly reducing industrial safety hazards and waste treatment costs.
Q: What is the impact of the CuCl catalyst on reaction selectivity?
A: The presence of cuprous chloride in the Grignard addition step enhances regioselectivity at the C17 carbonyl group, minimizing byproduct formation and simplifying downstream purification processes.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process avoids complex protection-deprotection sequences and uses readily available reagents, making it highly adaptable for commercial scale-up from 100 kgs to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ulipristal Acetate Supplier
At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced synthesis technology to life for our global partners. Our commitment to delivering high-purity Ulipristal Acetate is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch meets the highest international standards for safety and efficacy. We understand the critical nature of supply chain reliability in the pharmaceutical sector and have invested heavily in redundant systems and qualified raw material sources to guarantee uninterrupted service.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific project requirements and cost structures. Request a Customized Cost-Saving Analysis today to quantify the potential efficiencies for your organization, and ask for specific COA data and route feasibility assessments tailored to your regulatory needs. Together, we can accelerate the availability of high-quality hormonal therapeutics to patients worldwide through superior chemical manufacturing excellence.
