Advanced Solid-Phase Synthesis of Vapreotide: Technical Breakthroughs and Commercial Scalability
Advanced Solid-Phase Synthesis of Vapreotide: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry continuously seeks robust methodologies for the production of complex peptide therapeutics, and the synthesis of Vapreotide stands as a prime example of process evolution. Based on the technical disclosures found in patent CN101712716A, a novel approach utilizing solid-phase peptide synthesis (SPPS) combined with on-resin iodine oxidation has emerged as a superior alternative to traditional liquid-phase methods. This patent details a comprehensive strategy that begins with the loading of Fmoc-Trp(Boc)-OH onto Rink Amide resin, followed by the sequential assembly of the linear peptide chain. The critical innovation lies in the cyclization step, where iodine is employed directly on the solid support to form the essential disulfide bridges, thereby circumventing the operational difficulties and yield losses associated with solution-phase oxidation. For R&D directors and procurement specialists alike, understanding this shift is vital, as it represents a tangible pathway toward cost reduction in API manufacturing while ensuring the high structural fidelity required for somatostatin analogues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclic peptides like Vapreotide relied heavily on liquid-phase strategies, which present inherent challenges regarding scalability and purity control. In traditional liquid-phase oxidation, the linear peptide must first be fully cleaved from the resin and isolated before the cyclization step can occur. This isolation often leads to significant material loss and introduces impurities that complicate subsequent purification efforts. Furthermore, liquid-phase oxidation is prone to intermolecular reactions, leading to the formation of unwanted polymers and dimers rather than the desired intramolecular disulfide bonds. These side reactions not only depress the overall yield but also create a complex impurity profile that requires extensive and costly chromatographic separation. For a reliable peptide supplier, managing these variables in a large-scale reactor is operationally burdensome, often resulting in inconsistent batch-to-batch quality and extended production lead times that strain supply chain continuity.
The Novel Approach
In stark contrast, the methodology outlined in CN101712716A leverages the advantages of solid-phase synthesis to streamline the entire production workflow. By maintaining the peptide on the Rink Amide resin during the critical oxidation step, the effective concentration of the reactive thiol groups is controlled by the resin loading, which inherently favors intramolecular cyclization over intermolecular polymerization. The use of iodine as the oxidizing agent in a solid-phase context allows for precise control over reaction kinetics, with the patent specifying an optimal range of 10 to 80 equivalents, preferably around 20 equivalents. This approach eliminates the need for isolating the unstable linear intermediate, thereby reducing solvent consumption and processing time. The result is a process characterized by simple operation, lower raw material investment, and a total reaction yield that can reach 68%, with crude peptide purity exceeding 85%. This represents a significant leap forward in the commercial scale-up of complex peptide intermediates, offering a more predictable and efficient manufacturing route.
Mechanistic Insights into Solid-Phase Iodine Oxidation
The core of this technical advancement lies in the mechanistic execution of the disulfide bond formation while the peptide is anchored to the solid support. The process initiates with the standard Fmoc-based SPPS cycle, utilizing coupling agents such as DIC and HOBt to attach amino acids sequentially to the growing chain on the Rink Amide resin. Once the linear sequence D-Phe-Cys-Tyr-D-Trp-Lys-Val-Cys-Trp is assembled, the resin-bound peptide undergoes oxidation. The iodine molecules diffuse into the resin matrix and react with the free sulfhydryl groups of the cysteine residues. Because the peptide chains are spatially separated on the resin beads, the probability of two cysteine residues from different chains reacting is minimized, effectively suppressing dimerization. The reaction is typically conducted in solvents like N,N-dimethylformamide (DMF) at a controlled temperature of 35°C for approximately 10 hours. This specific thermal window is crucial; temperatures below 10°C result in incomplete oxidation, while temperatures above 40°C increase the risk of amino acid racemization, particularly at sensitive chiral centers.
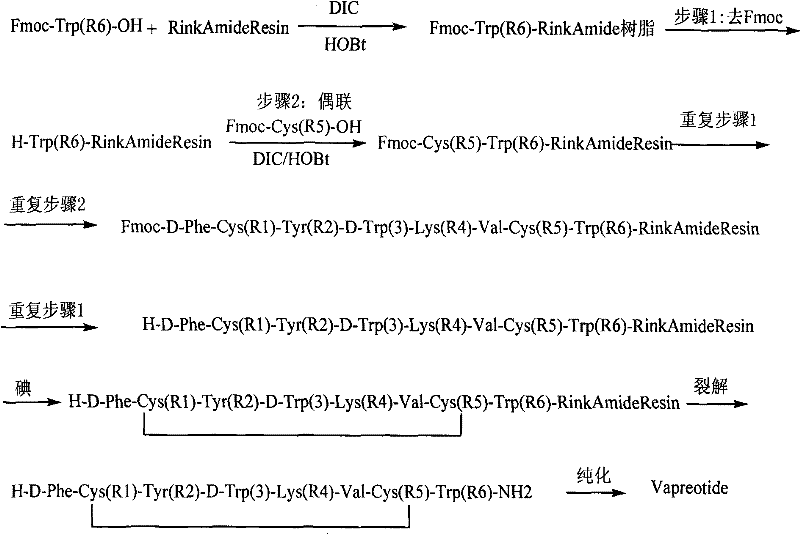
Following the oxidation, the integrity of the cyclic structure is preserved during the cleavage step, which utilizes a cocktail of trifluoroacetic acid (TFA) along with scavengers such as thioanisole, water, phenol, and EDT. The inclusion of these scavengers is mechanistically essential to trap reactive cationic species generated during the acidolysis of the side-chain protecting groups (such as Trt, Boc, and tBu), preventing them from alkylating the electron-rich indole ring of the tryptophan residues or the tyrosine phenol group. This careful orchestration of protection and deprotection chemistry ensures that the final product retains its biological activity and stereochemical purity. For technical teams evaluating high-purity API intermediates, this mechanism demonstrates a sophisticated understanding of peptide stability, ensuring that the final Vapreotide molecule meets the stringent specifications required for clinical applications in treating conditions like acromegaly and gastrointestinal bleeding.
How to Synthesize Vapreotide Efficiently
The synthesis of Vapreotide via this solid-phase route requires precise adherence to the coupling and oxidation parameters defined in the patent to ensure optimal yield and purity. The process begins with the preparation of the resin and proceeds through iterative cycles of deprotection and coupling, culminating in the critical on-resin oxidation and final cleavage. Operators must pay close attention to the stoichiometry of the iodine oxidant and the duration of the reaction to avoid under-oxidation or polymer formation. The following guide outlines the standardized operational framework derived from the patent data, providing a clear roadmap for laboratory and pilot-scale execution. Detailed standardized synthesis steps are provided in the guide below.
- Load Fmoc-Trp(Boc)-OH onto Rink Amide Resin and perform sequential amino acid coupling using DIC/HOBt to build the linear peptide chain.
- Perform solid-phase oxidation of the linear peptide resin using an iodine solution (10-80 equivalents) in DMF at 35°C for approximately 10 hours.
- Cleave the cyclic peptide from the resin using a TFA-based cocktail (e.g., TFA/thioanisole/water/phenol/EDT), followed by precipitation, purification, and freeze-drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this solid-phase iodine oxidation method offers compelling economic and logistical benefits that extend beyond mere technical elegance. The elimination of the liquid-phase isolation step significantly reduces the number of unit operations required, which directly translates to lower labor costs and reduced equipment occupancy time. Furthermore, the improved yield and crude purity mean that less starting material is wasted, and the burden on downstream purification columns is substantially lightened. This efficiency gain is critical for reducing lead time for high-purity peptide intermediates, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on quality standards or regulatory compliance.
- Cost Reduction in Manufacturing: The adoption of DIC/HOBt as the primary coupling system, combined with the streamlined oxidation process, drastically simplifies the production workflow. By avoiding the expensive and time-consuming isolation of linear intermediates, the process reduces solvent usage and waste disposal costs. The ability to achieve high crude purity (over 85%) means that the final purification step is more efficient, requiring less chromatography media and time. Consequently, the overall cost of goods sold (COGS) is significantly optimized, making the final API more competitive in the global marketplace without sacrificing quality.
- Enhanced Supply Chain Reliability: The robustness of the solid-phase method enhances supply chain stability by minimizing the risk of batch failures due to complex liquid-phase handling. The reagents used, such as iodine and standard Fmoc-amino acids, are readily available commodities, reducing the risk of raw material shortages. Additionally, the simplified process flow reduces the dependency on highly specialized operators for intricate liquid-phase manipulations, thereby mitigating human error risks. This reliability ensures a consistent supply of Vapreotide, which is essential for maintaining uninterrupted production schedules for downstream drug formulations.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers distinct advantages. The reduction in solvent volume and the avoidance of hazardous liquid-phase oxidation byproducts contribute to a greener manufacturing footprint. The solid-phase nature of the reaction allows for easier containment and handling of reagents, improving workplace safety. Moreover, the process is inherently scalable; the resin-based approach can be adapted from gram-scale laboratory synthesis to multi-kilogram commercial production with minimal re-optimization, facilitating a smoother technology transfer from R&D to full-scale manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Vapreotide using the patented solid-phase oxidation method. These insights are derived directly from the experimental data and process descriptions found in CN101712716A, providing clarity on yield expectations, purity profiles, and operational parameters. Understanding these details is crucial for stakeholders evaluating the feasibility of this route for their specific supply chain needs.
Q: What is the primary advantage of using solid-phase iodine oxidation for Vapreotide?
A: The primary advantage is the significant simplification of the process compared to traditional liquid-phase oxidation. By performing the oxidation while the peptide is still attached to the resin, the method avoids complex isolation steps, reduces polymer formation, and improves the oxidation yield by approximately 10%-20%.
Q: How does this method impact the purity of the crude peptide?
A: This optimized protocol achieves a crude peptide purity of greater than 85% prior to final purification. The controlled use of iodine (specifically around 20 equivalents) and precise temperature control (35°C) minimizes racemization and side reactions, facilitating easier downstream purification.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the method is highly suitable for scale-up. It utilizes robust solid-phase synthesis techniques with common coupling agents like DIC/HOBt and avoids hazardous or difficult-to-handle liquid-phase cyclization conditions, making it safer and more cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vapreotide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to meet the evolving demands of the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101712716A are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Vapreotide meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of peptide synthesis, delivering products that support your clinical and commercial goals with unwavering consistency.
We invite you to collaborate with us to leverage these process innovations for your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized manufacturing routes can enhance your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in solid-phase peptide synthesis can become a strategic asset to your supply chain.
