Advanced Solid-Phase Synthesis of Vapreotide: Enhancing Yield and Purity for Commercial Scale-up
Introduction to Advanced Vapreotide Manufacturing
The pharmaceutical industry constantly seeks robust methodologies for synthesizing complex peptide therapeutics, and Patent CN101712716B presents a significant advancement in the production of Vapreotide, a potent somatostatin analog used for treating acute variceal bleeding and other endocrine disorders. This intellectual property outlines a refined solid-phase synthesis strategy that fundamentally alters the oxidation step, transitioning from cumbersome liquid-phase techniques to a more efficient solid-phase iodine oxidation protocol. By anchoring the peptide chain to a Rink Amide resin and utilizing specific coupling agents like DIC and HOBt, the process achieves a total reaction yield of approximately 68% with a crude peptide purity exceeding 85%. For global procurement leaders and R&D directors, this patent represents a critical pathway for securing a reliable API intermediate supplier capable of delivering high-quality materials with consistent batch-to-batch reproducibility. The technical nuances described herein provide a blueprint for cost reduction in pharmaceutical manufacturing by minimizing purification burdens and maximizing throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of cyclic peptides like Vapreotide has relied heavily on liquid-phase oxidation methods, which introduce substantial operational complexities and inefficiencies into the manufacturing workflow. In liquid-phase systems, the oxidation of linear precursors to form the critical disulfide bonds often requires rigorous control of concentration and stoichiometry to prevent intermolecular polymerization, which drastically lowers yields. Furthermore, liquid-phase reactions typically necessitate multiple extraction and isolation steps to remove excess oxidants and by-products, leading to significant solvent consumption and extended processing times. These conventional approaches often struggle with scalability, as the risk of forming difficult-to-remove polymeric impurities increases with batch size, thereby complicating the downstream purification process and inflating the overall cost of goods. For supply chain managers, these inefficiencies translate into longer lead times and higher vulnerability to production bottlenecks, making the search for alternative synthetic routes a strategic priority.
The Novel Approach
The methodology disclosed in Patent CN101712716B revolutionizes this landscape by employing a solid-phase synthesis approach where the oxidation step is performed directly on the resin-bound peptide. This innovative technique utilizes iodine as a mild yet effective oxidant within a solid-phase matrix, effectively circumventing the intermolecular interactions that lead to polymerization in liquid systems. By keeping the peptide anchored to the Rink Amide resin during cyclization, the process ensures that the reaction remains intramolecular, thereby significantly enhancing the selectivity for the desired cyclic monomer. This shift not only simplifies the operational workflow by eliminating intermediate isolation steps but also improves the oxidation yield by a reported margin of 10% to 20% compared to traditional methods. The result is a streamlined process characterized by simple reaction operations, lower raw material investment, and a high total yield, positioning this technology as a superior choice for the commercial scale-up of complex peptide APIs.
Mechanistic Insights into Solid-Phase Iodine Oxidation
The core chemical innovation of this patent lies in the precise optimization of the solid-phase oxidation conditions, specifically utilizing iodine in N,N-dimethylformamide (DMF) to facilitate the formation of the 2-7 disulfide linkage essential for Vapreotide's bioactivity. The mechanism involves the exposure of the linear peptide-resin to an iodine solution, where the thiol groups of the cysteine residues are selectively oxidized to form the disulfide bond while the peptide remains immobilized. Critical to this success is the control of reaction parameters; the patent specifies a temperature range of 10°C to 40°C, with an optimal setpoint of 35°C. Operating below 10°C results in incomplete oxidation with yields dropping below 50%, whereas temperatures exceeding 40°C induce amino acid racemization, generating chiral impurities that are notoriously difficult to separate. Additionally, the reaction time is tightly controlled between 8 to 12 hours, preferably 10 hours, to ensure complete conversion without degrading the peptide backbone or forming side products.
Furthermore, the stoichiometry of the oxidant plays a pivotal role in the purity profile of the final product. The patent dictates an iodine consumption of 10 to 80 times the molar weight of the peptide, with a preferred ratio of 20 times. This specific excess ensures that the oxidation potential is sufficient to drive the reaction to completion without being so excessive that it causes oxidative damage to other sensitive residues like tryptophan or tyrosine. The use of DMF as the solvent is also strategic, as it effectively swells the polystyrene-based resin, allowing the iodine reagent to penetrate the polymer matrix and access the reactive sites efficiently. This meticulous balance of reagent concentration, temperature, and time results in a crude peptide purity greater than 85%, significantly reducing the load on subsequent purification columns and demonstrating a deep understanding of high-purity API intermediate production dynamics.
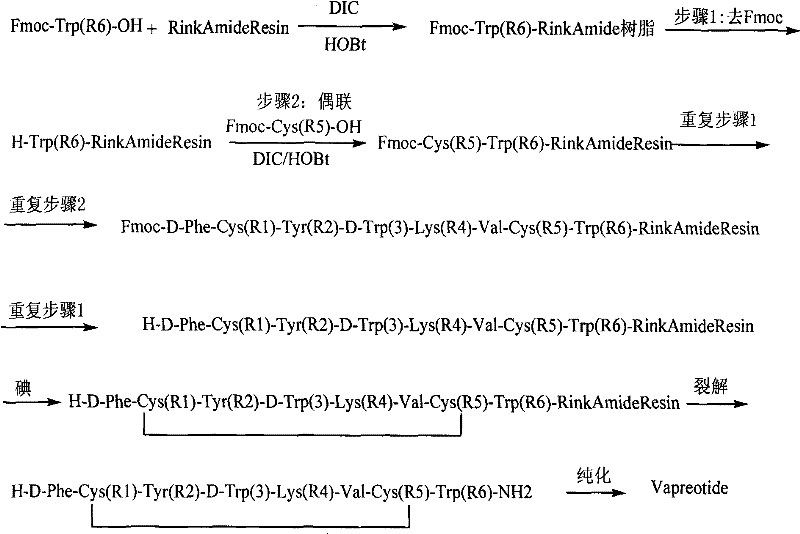
How to Synthesize Vapreotide Efficiently
Implementing this synthesis route requires adherence to a strict sequence of solid-phase peptide synthesis (SPPS) protocols followed by the specialized on-resin oxidation step. The process begins with the loading of the C-terminal amino acid onto the Rink Amide resin, followed by the iterative addition of protected amino acids using activated esters generated by DIC and HOBt. Once the full linear sequence is assembled, the resin is subjected to the iodine oxidation treatment under the optimized conditions previously discussed. This standardized approach allows for the reproducible manufacture of Vapreotide with minimal variability. For detailed operational parameters, including specific reagent weights, washing protocols, and cleavage cocktail formulations, please refer to the standardized synthesis guide below which encapsulates the critical steps derived from the patent embodiments.
- Load Fmoc-Trp(Boc)-OH onto Rink Amide Resin using DIC/HOBt coupling agents to initiate the peptide chain assembly.
- Perform sequential solid-phase coupling of the remaining amino acids (Cys, Tyr, Lys, Val) following the specific Vapreotide sequence.
- Oxidize the linear peptide resin using an iodine solution in DMF at 35°C for approximately 10 hours to form the disulfide bridge.
- Cleave the cyclic peptide from the resin using a TFA-based cocktail, followed by precipitation, HPLC purification, and freeze-drying.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this solid-phase iodine oxidation method offers profound benefits for procurement managers and supply chain heads focused on efficiency and cost containment. The primary advantage stems from the drastic simplification of the manufacturing process, which eliminates the need for complex liquid-phase isolation and purification steps prior to cyclization. By performing the oxidation on the solid support, the process inherently reduces the volume of solvents required for workup and minimizes the loss of product during transfer operations, leading to substantial cost savings in raw materials and waste disposal. Moreover, the improved yield and purity profile mean that less starting material is needed to produce a given amount of final API, directly lowering the cost of goods sold (COGS) and enhancing the overall economic viability of the project. This efficiency is crucial for maintaining competitive pricing in the global marketplace for somatostatin analogs.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction in solvent usage for intermediate isolations contribute to a leaner manufacturing budget. The process relies on readily available reagents like iodine and standard coupling agents, avoiding the volatility and high costs associated with specialized noble metal catalysts. Additionally, the higher crude purity reduces the burden on preparative HPLC, extending column life and reducing the consumption of expensive chromatographic solvents, which collectively drives down the operational expenditure for large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of the solid-phase method ensures consistent output quality, which is vital for maintaining uninterrupted supply to downstream drug product manufacturers. Since the reagents involved are commodity chemicals with stable global supply chains, the risk of raw material shortages is significantly mitigated. The simplified workflow also reduces the dependency on highly specialized operator skills for complex liquid-phase manipulations, allowing for more flexible staffing and reduced training overheads, thereby strengthening the resilience of the production schedule against labor fluctuations.
- Scalability and Environmental Compliance: Solid-phase synthesis is inherently scalable, allowing for the transition from laboratory gram quantities to multi-kilogram commercial batches with minimal process re-engineering. The use of iodine, which can be recovered and recycled, along with the reduced generation of hazardous liquid waste streams, aligns well with modern environmental, health, and safety (EHS) standards. This compliance facilitates smoother regulatory approvals and reduces the environmental footprint of the manufacturing facility, appealing to partners who prioritize sustainable sourcing and green chemistry initiatives in their supply chain audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Vapreotide using the patented solid-phase oxidation method. These insights are derived directly from the experimental data and process descriptions found in Patent CN101712716B, providing clarity on the feasibility and advantages of this route. Understanding these details is essential for technical teams evaluating the potential for technology transfer or contract manufacturing partnerships. The answers reflect the specific optimizations regarding temperature, stoichiometry, and purification that define the success of this methodology.
Q: What are the advantages of solid-phase iodine oxidation for Vapreotide?
A: Solid-phase iodine oxidation significantly simplifies the process compared to traditional liquid-phase methods. It avoids complex isolation steps, improves oxidation yield by 10%-20%, and reduces the formation of polymeric impurities, leading to a crude peptide purity of over 85%.
Q: What are the optimal conditions for the oxidation step?
A: The patent specifies using iodine as the oxidant in N,N-dimethylformamide (DMF) solvent. The optimal temperature is maintained at 35°C for about 10 hours, with an iodine molar ratio of roughly 20 times that of the peptide resin to ensure complete cyclization without racemization.
Q: How is the final Vapreotide product purified?
A: After cleavage from the resin using a TFA/thioanisole/water/phenol/EDT cocktail, the crude peptide is precipitated in ether. The final high-purity product is obtained through preparative high-pressure liquid chromatography (HPLC) followed by freeze-drying.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vapreotide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in Patent CN101712716B to meet the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering Vapreotide and related peptide intermediates with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the highest international standards for identity, potency, and impurity profiles. Our infrastructure is designed to support the complex requirements of solid-phase peptide synthesis, providing a secure and compliant environment for your most valuable assets.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific project needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this solid-phase oxidation protocol for your supply chain. We encourage you to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and reliability in your API sourcing strategy. Let us be your trusted partner in navigating the complexities of peptide manufacturing.
