Advanced Rhodium-Catalyzed Synthesis of Terminal Alkenyl Indoles for Pharmaceutical Manufacturing
Advanced Rhodium-Catalyzed Synthesis of Terminal Alkenyl Indoles for Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to access complex heterocyclic scaffolds, particularly those containing alkenyl functionalities which serve as versatile handles for further derivatization. Patent CN112375068B introduces a groundbreaking methodology for the preparation of terminal alkenyl indole derivatives, addressing critical bottlenecks in modern organic synthesis. This technology leverages a sophisticated Rhodium-catalyzed C-H activation strategy to directly install a terminal vinyl group onto the indole core using vinyltriethoxysilane. Unlike traditional approaches that rely on pre-functionalized starting materials, this invention enables the direct transformation of readily available indole substrates into high-value intermediates. The significance of this development lies in its ability to streamline the production of bioactive molecules, where the terminal alkene moiety can be subsequently exploited for cross-metathesis, dihydroxylation, or epoxidation reactions to generate diverse libraries of drug candidates.
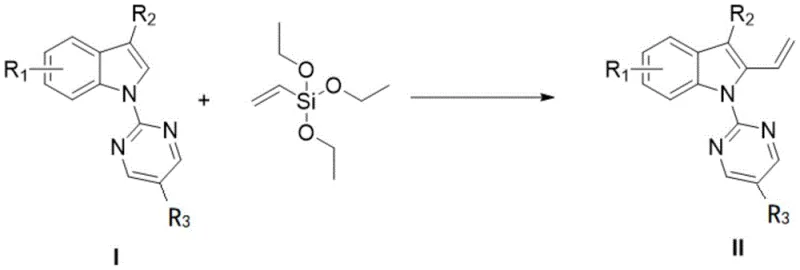
The core innovation described in the patent involves reacting a substituted indole compound (Compound I) with vinyltriethoxysilane in the presence of a specific catalyst system and organic solvent to yield the target terminal alkenyl indole (Compound II). The reaction conditions are meticulously optimized to ensure high regioselectivity and yield, operating typically at temperatures between 80-120 °C. This process represents a paradigm shift from stoichiometric reagents to catalytic efficiency, aligning perfectly with the principles of green chemistry and sustainable manufacturing. For R&D directors and process chemists, this offers a powerful tool to accelerate lead optimization campaigns by providing rapid access to structurally diverse alkenyl indoles without the burden of lengthy synthetic sequences.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkenyl indoles has been plagued by inefficiencies inherent in classical organic transformations. Traditional methods often necessitate the prior installation of reactive functional groups, such as halogens or aldehydes, onto the indole skeleton before the olefin can be introduced. For instance, the Wittig reaction requires the preparation of phosphorus ylides and indole-2-carbaldehydes, generating substantial stoichiometric waste in the form of phosphine oxides which are notoriously difficult to remove. Alternatively, the hydrogenation of alkynylindoles using palladium on carbon catalysts poses risks of over-reduction or incomplete selectivity, leading to impurity profiles that complicate downstream purification. Furthermore, reduction coupling methods utilizing TiCl4 and Zinc are moisture-sensitive and generate large volumes of hazardous metal waste, creating significant environmental and disposal challenges for large-scale operations. These legacy processes fail to meet the modern demands of step economy and atom economy, resulting in higher production costs and longer lead times for critical pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in patent CN112375068B circumvents these historical limitations by employing a direct C-H bond olefination strategy. By utilizing vinyltriethoxysilane as the olefin source, the process eliminates the need for pre-halogenation or formylation steps, effectively collapsing multiple synthetic operations into a single catalytic event. The use of a Rhodium(III) catalyst system allows for the activation of inert C-H bonds under relatively mild thermal conditions, typically around 90 °C in solvents like 1,2-dichloroethane. This approach not only simplifies the operational workflow but also drastically reduces the consumption of raw materials and reagents. The terminal nature of the resulting alkene is particularly valuable, as it provides an unsubstituted double bond that is highly amenable to subsequent functionalization, such as ring-closing metathesis to form carbazoles or dihydroxylation to create chiral diols. This novel route exemplifies how modern catalysis can transform complex molecule synthesis into a more streamlined and economically viable process.
Mechanistic Insights into Rhodium-Catalyzed C-H Olefination
The success of this transformation relies on a sophisticated catalytic cycle driven by the Rhodium(III) species, specifically the dimer [RhCp*Cl2]2. The mechanism initiates with the coordination of the indole nitrogen to the rhodium center, directing the metal to the proximal C2 position for C-H activation. This step is facilitated by the presence of additives like AgF, which likely assist in halide abstraction to generate the active cationic rhodium species. Once the rhodacycle intermediate is formed via concerted metalation-deprotonation, the vinyltriethoxysilane coordinates to the metal center. Subsequent migratory insertion of the vinyl group into the Rh-C bond extends the carbon framework, setting the stage for the formation of the new C-C bond. The cycle is completed by beta-hydride elimination or a similar protodemetalation step, releasing the terminal alkenyl indole product and regenerating the active catalyst. The inclusion of Cu(OAc)2 serves as a terminal oxidant or co-catalyst to maintain the oxidation state of the metal and drive the reaction to completion, ensuring high turnover numbers even with complex substrates.
From an impurity control perspective, this catalytic system offers distinct advantages over non-directed methods. The directing group effect of the pyrimidine or indole nitrogen ensures that functionalization occurs exclusively at the C2 position, minimizing the formation of regioisomers that are common in electrophilic aromatic substitution reactions. Furthermore, the mild reaction conditions prevent the decomposition of sensitive functional groups such as esters, nitriles, or halogens that might be present on the indole ring (R1, R2, or R3 substituents). The patent data demonstrates excellent tolerance for a wide range of substituents, including electron-withdrawing groups like nitro and cyano, as well as electron-donating groups like methoxy and alkyl chains. This robustness implies that the impurity profile of the crude product is relatively clean, primarily consisting of unreacted starting material rather than complex side-products, thereby facilitating easier purification via standard silica gel chromatography.
How to Synthesize Terminal Alkenyl Indole Derivatives Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The process begins with the precise weighing of the indole substrate and vinyltriethoxysilane, typically in a molar ratio of 1:(2-4) to drive the equilibrium towards the product. The catalyst loading is critical, with the patent specifying a molar ratio of Cu(OAc)2:AgF:[RhCp*Cl2]2 relative to the substrate of approximately (2-3):(2-3):(0.01-0.1). The reaction is conducted in an organic solvent, with 1,2-dichloroethane (DCE) being the preferred medium, often mixed with DMF to enhance solubility. Detailed standardized synthesis steps, including specific workup procedures and purification protocols, are outlined in the technical guide below to ensure reproducibility across different laboratory scales.
- Combine Compound I (indole derivative), vinyltriethoxysilane, and the catalyst system comprising [RhCp*Cl2]2, Cu(OAc)2, and AgF in an organic solvent such as 1,2-dichloroethane.
- Heat the reaction mixture to a temperature between 80-120 °C, preferably 90 °C, and maintain stirring for 12 to 24 hours under an inert atmosphere until TLC indicates completion.
- Cool the mixture to room temperature, concentrate under reduced pressure to obtain a crude product, and purify via silica gel column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Rhodium-catalyzed technology translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of pre-functionalization steps means fewer raw materials need to be sourced and managed, reducing the complexity of the supply chain and mitigating the risk of bottlenecks associated with specialized reagents like phosphorus ylides or alkynyl precursors. Moreover, the use of vinyltriethoxysilane, a commodity chemical, as the olefin source ensures a stable and cost-effective supply of key reagents. The simplified workflow also reduces the demand on reactor time and labor, allowing manufacturing facilities to increase throughput without significant capital expenditure on new equipment. These factors collectively contribute to a more resilient supply chain capable of meeting the fluctuating demands of the pharmaceutical market.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic reduction in synthetic steps. By bypassing the need for separate halogenation or formylation reactions, manufacturers save significantly on reagents, solvents, and energy consumption associated with those additional unit operations. The high atom economy of the C-H activation means that a larger proportion of the input mass ends up in the final product, reducing waste disposal costs. Additionally, the avoidance of expensive palladium catalysts or stoichiometric titanium reagents further lowers the direct material costs, making the production of high-purity terminal alkenyl indoles more economically competitive.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of commercially available and stable starting materials. Indoles and vinyltriethoxysilane are widely produced commodities with established global supply networks, reducing the dependency on niche suppliers who might face production disruptions. The robustness of the reaction conditions, which tolerate a variety of functional groups, means that the same process can be applied to a broad library of substrates without needing to requalify entirely new synthetic routes for each analog. This flexibility allows for rapid scale-up and ensures consistent availability of diverse intermediates, crucial for maintaining continuous clinical trial supplies or commercial manufacturing runs.
- Scalability and Environmental Compliance: Scaling this process is straightforward due to the use of standard organic solvents like DCE and DMF, which are familiar to process engineers and compatible with existing infrastructure. The reaction operates at moderate temperatures (90 °C), avoiding the extreme cryogenic or high-pressure conditions that complicate scale-up and increase safety risks. From an environmental standpoint, the reduction in waste generation aligns with increasingly stringent regulatory requirements for green manufacturing. The simplified purification process, often achievable via standard column chromatography or crystallization, minimizes the volume of solvent waste, supporting corporate sustainability goals and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this terminal alkenylation technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on reaction scope and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this method into their existing process development pipelines.
Q: What are the primary advantages of using vinyltriethoxysilane for indole olefination?
A: Vinyltriethoxysilane serves as an efficient olefinating agent that allows for direct C-H bond activation without the need for pre-functionalized halogen or aldehyde groups on the indole ring. This significantly improves atom economy and reduces the number of synthetic steps compared to traditional Wittig or hydrogenation methods.
Q: Which catalyst system is required for this terminal alkenylation reaction?
A: The process utilizes a robust transition metal catalyst system centered around a Rhodium(III) complex, specifically [RhCp*Cl2]2. This is supported by Copper(II) acetate (Cu(OAc)2) as an oxidant and Silver Fluoride (AgF) as an additive to facilitate the catalytic cycle and ensure high conversion rates.
Q: How does this method improve upon conventional alkenyl indole synthesis?
A: Conventional methods often require harsh conditions or multi-step sequences involving alkynyl precursors or phosphorus ylides. This novel approach achieves direct functionalization at the C2 position of the indole under relatively mild conditions (90 °C), offering superior step economy and simplifying the purification process for high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terminal Alkenyl Indole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant is seamless and efficient. Our commitment to quality is unwavering, with stringent purity specifications enforced through our rigorous QC labs equipped with state-of-the-art analytical instrumentation. We understand that the integrity of your pharmaceutical intermediates is paramount, and our team of expert chemists is dedicated to optimizing every parameter of the synthesis to deliver products that meet the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data for our terminal alkenyl indole portfolio and discuss route feasibility assessments that can drive your project forward with speed and confidence. Let us be your partner in turning complex chemical challenges into commercial successes.
