Scalable Synthesis of Polypeptide Drugs Using Novel Soluble Solid-Phase Carriers
The pharmaceutical industry is constantly seeking more efficient methods for the large-scale production of complex polypeptide therapeutics, and patent CN116284238A introduces a groundbreaking approach to this challenge. This technology utilizes a novel non-classical solid-phase synthesis carrier, specifically designed to overcome the inherent limitations of traditional resin-based synthesis. By employing a carrier structure based on bis[4-(octadecyloxy)phenyl]methylamine, the process enables a unique "soluble-reactive, insoluble-purifiable" mechanism that transforms the economics of peptide manufacturing. This innovation is particularly relevant for the production of high-value drugs like Eptifibatide, offering a pathway to significantly lower production costs and environmental impact while maintaining high purity standards.
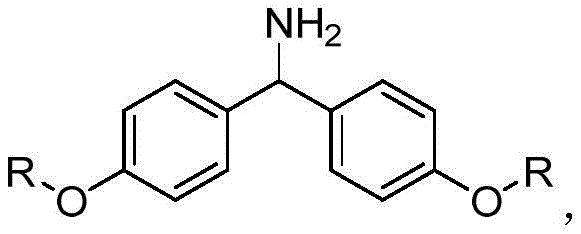
For R&D directors and process chemists, understanding the structural basis of this carrier is crucial. The carrier features long alkyl chains (n-octadecyl) attached to a central diphenylmethane amine scaffold. This specific architecture imparts amphiphilic properties that are key to the process success: the carrier dissolves readily in common organic reaction media such as dichloromethane or DMF, allowing the peptide coupling reactions to proceed in a homogeneous phase. However, upon the addition of anti-solvents like n-hexane or methyl tert-butyl ether, the carrier-peptide conjugate precipitates quantitatively. This solubility switch eliminates the diffusion limitations often encountered with cross-linked polystyrene resins, ensuring higher coupling efficiencies and easier monitoring of reaction progress.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional Solid Phase Peptide Synthesis (SPPS), while revolutionary since Merrifield's inception, suffers from significant scalability bottlenecks that hinder cost-effective commercial manufacturing. The primary issue is the physical behavior of standard resins, which swell dramatically—often expanding 5 to 10 times their original volume—upon exposure to reaction solvents. This swelling necessitates the use of disproportionately large reactors to accommodate the expanded gel phase, leading to inefficient use of capital equipment and floor space. Furthermore, the heterogeneous nature of the reaction requires a substantial excess of amino acids and coupling reagents to drive the equilibrium forward, as diffusion into the resin beads is slow and often incomplete. This not only inflates raw material costs but also generates significant chemical waste that must be managed.
Another critical drawback of conventional methods is the immense consumption of organic solvents required for washing the resin between coupling and deprotection steps. To remove excess reagents and by-products from the swollen resin matrix, vast volumes of DMF or DCM are pumped through the reactor, creating a heavy environmental burden and increasing disposal costs. Additionally, traditional SPPS often requires specialized reactors equipped with bottom filtration plates or sintered discs to retain the resin while allowing liquids to pass. These specialized vessels are more expensive to procure and maintain than standard stirred tank reactors, and they introduce potential points of failure, such as clogging, which can disrupt production schedules and compromise batch consistency.
The Novel Approach
The novel approach described in the patent fundamentally reimagines the physical state of the synthesis support, transitioning from a static, swollen gel to a dynamic, soluble species. By utilizing the non-classical carrier, the coupling reactions occur in a true homogeneous solution, mimicking the kinetics of liquid-phase synthesis while retaining the purification benefits of solid-phase methods. This homogeneity ensures that every molecule of the carrier is equally accessible to the activated amino acids, drastically reducing the need for the massive reagent excesses typical of resin-based protocols. The reaction kinetics are faster and more predictable, allowing for tighter control over the synthesis timeline and impurity profile.
Purification in this new system is achieved through a simple precipitation step rather than iterative filtration and washing. After the coupling or deprotection reaction is complete, the reaction mixture is concentrated and poured into a poor solvent, causing the peptide-carrier conjugate to crash out of solution as a solid. This solid can then be easily collected via filtration or centrifugation. This "precipitation purification" strategy effectively removes soluble impurities, excess reagents, and urea by-products in a single operation, slashing solvent consumption by an estimated 70%-80% compared to traditional washing protocols. The result is a streamlined process that combines the best aspects of both liquid and solid-phase synthesis.
Mechanistic Insights into Soluble Solid-Phase Peptide Synthesis
The core mechanism driving this technology is the reversible solubility of the HZ-WSP-M carrier, which is dictated by the interaction between the long alkyl chains and the solvent environment. In benign solvents like dichloromethane (DCM) or dimethylformamide (DMF), the lipophilic octadecyl chains solvate effectively, keeping the entire macromolecular construct in solution. This allows the amine functionality on the carrier to react freely with Fmoc-protected amino acids activated by condensing agents such as DIC/HOBt. The reaction proceeds under mild conditions, typically between 35-40°C, which minimizes the risk of racemization—a common concern in peptide synthesis. The homogeneous nature of the reaction ensures that the local concentration of reactants around the reactive site is uniform, preventing the formation of deletion sequences that often plague heterogeneous syntheses.
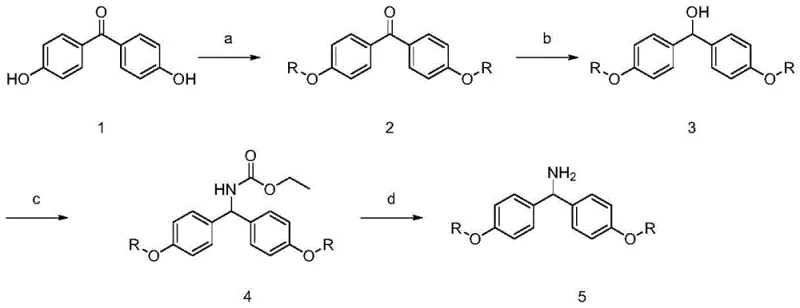
Impurity control is inherently built into the precipitation mechanism. When the reaction mixture is introduced to an anti-solvent like n-hexane or methyl tert-butyl ether, the solubility parameter of the medium changes drastically. The peptide-carrier conjugate, being a large amphiphilic molecule, becomes insoluble and aggregates into a solid phase. Crucially, small molecule impurities, unreacted amino acids, and urea by-products remain soluble in this new solvent mixture and are removed in the supernatant. This selective precipitation acts as a powerful purification step at every cycle of the synthesis. Furthermore, because the intermediate is isolated as a discrete solid after each step, it becomes possible to perform quality control checks, such as HPLC or MS analysis, on the growing peptide chain. This level of process analytical technology (PAT) is difficult to achieve with traditional resins where the peptide remains tethered and hidden within the bead matrix.
How to Synthesize Eptifibatide Efficiently
The synthesis of Eptifibatide using this novel carrier follows a logical sequence of coupling, deprotection, and final cleavage, leveraging the unique solubility properties of the HZ-WSP-M support. The process begins with the activation of Fmoc-protected amino acids using standard coupling reagents, followed by reaction with the soluble carrier. After each coupling, the product is precipitated and filtered, removing excess reagents before the next Fmoc deprotection step using diethylamine in DCM. This cycle is repeated until the full linear sequence is assembled. The detailed operational parameters, including specific molar ratios and temperature controls, are critical for maximizing yield and minimizing epimerization. For a comprehensive guide on the specific execution of these steps, please refer to the standardized protocol below.
- Perform amino acid coupling reaction in a benign solvent medium using the non-classical solid-phase carrier and Fmoc-amino acid with a condensing reagent.
- Concentrate, precipitate, and filter the reaction mixture to isolate the Fmoc-peptide carrier intermediate.
- Repeat coupling and deprotection cycles until the full peptide sequence is assembled, then cleave the final product using a cutting solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this non-classical solid-phase synthesis technology offers tangible economic and logistical benefits that directly impact the bottom line. The most significant advantage is the drastic reduction in raw material costs, driven by the improved atom economy of the homogeneous coupling reactions. Unlike traditional resin methods that often require a 3-to-5-fold excess of expensive protected amino acids to ensure completion, this method operates efficiently with much lower stoichiometric ratios, typically around 1.2 equivalents. This optimization translates directly into lower cost of goods sold (COGS) for high-value peptide intermediates. Additionally, the ability to use standard stainless steel reactors instead of specialized glass-lined or filtered vessels reduces capital expenditure (CAPEX) barriers for scaling up production.
- Cost Reduction in Manufacturing: The economic model of this process is superior due to the simultaneous reduction in both material and utility costs. By eliminating the need for massive solvent washes, the facility saves significantly on solvent purchase, recovery, and disposal fees. The patent data indicates that organic solvent usage can be reduced by 70%-80%, which is a massive operational saving in large-scale manufacturing. Furthermore, the reduced requirement for excess amino acids and condensing agents lowers the direct material cost by approximately 50%-60%. These efficiencies compound to deliver an overall production cost reduction estimated between 50% and 70%, making the final API or intermediate much more competitive in the global market.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the simplification of the equipment requirements enhances reliability and flexibility. Since the process does not rely on fragile resin beads or specialized filtration hardware, the risk of batch failure due to mechanical issues is minimized. The use of common industrial solvents like DCM and Hexane ensures that raw material sourcing is stable and not subject to the supply constraints of specialty resins. Moreover, the shorter cycle times associated with homogeneous reactions allow for faster throughput, enabling the manufacturing site to respond more agilely to fluctuations in demand. This agility is crucial for maintaining continuous supply of critical medications like Eptifibatide.
- Scalability and Environmental Compliance: Scaling peptide synthesis is notoriously difficult due to heat transfer and mixing issues in swollen resin beds. This new technology bypasses those physical limitations, allowing for straightforward scale-up from pilot to commercial tonnage using standard mixing protocols. The environmental footprint is also substantially smaller, aligning with modern green chemistry mandates. The reduction in solvent waste by 70%-80% significantly lowers the burden on wastewater treatment facilities and reduces the carbon footprint of the manufacturing process. This compliance with stringent environmental regulations future-proofs the supply chain against tightening ecological laws and potential carbon taxes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel polypeptide synthesis technology. These answers are derived directly from the technical specifications and comparative data provided in the patent literature, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: How does this new carrier solve the swelling issue of traditional resins?
A: Traditional insoluble resins swell 5-10 times their volume, requiring massive reactors. This novel carrier remains soluble during reaction (homogeneous phase) and only precipitates during workup, drastically reducing reactor volume requirements by 30%-50%.
Q: Does this method require specialized solid-phase reactors?
A: No. Unlike traditional SPPS which needs reactors with sieve plates for filtration, this method uses standard reaction kettles because the purification is achieved via precipitation and centrifugation/filtration of the dissolved carrier.
Q: What are the environmental benefits regarding solvent usage?
A: The process significantly reduces organic solvent consumption for washing. Since the carrier precipitates out of solution, extensive washing of swollen resin is unnecessary, reducing solvent usage by approximately 70%-80%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eptifibatide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthesis technologies like the non-classical solid-phase carrier system for producing high-quality polypeptide intermediates. As a leading CDMO partner, we possess the technical expertise to adapt and optimize such innovative routes for commercial production. Our facilities are equipped to handle complex synthetic pathways, and we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, ensuring that every batch of Eptifibatide or related peptide intermediate adheres to the highest global pharmacopeial standards.
We invite pharmaceutical companies and biotech firms to collaborate with us to leverage these cost-saving and efficiency-boosting technologies. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific peptide portfolio. We encourage you to reach out to request specific COA data and route feasibility assessments for your target molecules. Let us help you navigate the complexities of peptide manufacturing with a reliable partner dedicated to quality, efficiency, and sustainable growth.
