Advanced Solid-Phase Synthesis of Eptifibatide Acetate for Commercial Scale-Up
Advanced Solid-Phase Synthesis of Eptifibatide Acetate for Commercial Scale-Up
The pharmaceutical landscape for antithrombotic agents continues to evolve, with Eptifibatide acetate standing out as a critical synthetic cyclic heptapeptide. Based on the structural insights from barbourin found in snake venom, this potent platelet glycoprotein IIb/IIIa receptor antagonist offers rapid onset and reversible inhibition, making it indispensable for acute coronary syndromes. The preparation method detailed in patent CN102584944A introduces a transformative approach to synthesizing this complex molecule, addressing long-standing challenges in solid-phase peptide synthesis (SPPS). By shifting from traditional stepwise amino acid addition to a strategic fragment condensation methodology, this innovation directly targets the reduction of difficult-to-remove impurities. For R&D directors and procurement specialists alike, understanding this shift is vital for securing a reliable supply of high-purity API intermediates that meet stringent regulatory standards.
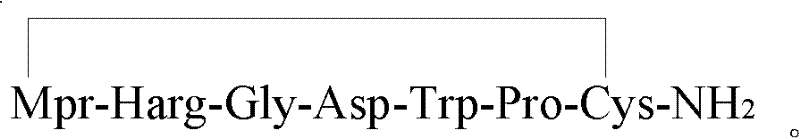
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional solid-phase synthesis of Eptifibatide often relies on the sequential inoculation of individual Fmoc-protected amino acids onto a resin backbone. While conceptually straightforward, this linear approach encounters significant chemical hurdles when dealing with specific sequences like Glycine (Gly) and Homoarginine (Harg). The inherent characteristics of Glycine, lacking a side chain, combined with the steric and electronic properties of the Harg structure, create a hotspot for synthesis errors. Specifically, the conventional process is prone to generating deletion sequences such as [-1Harg]-Eptifibatide and insertion impurities like [+1Gly]-Eptifibatide. These structurally similar byproducts possess polarities nearly identical to the target molecule, rendering standard purification techniques inefficient and drastically reducing the overall yield of the crude product. Consequently, manufacturers face escalated costs due to excessive solvent consumption and column loading during the purification phase, alongside the risk of failing to meet purity specifications.
The Novel Approach
The innovative strategy outlined in the patent circumvents these pitfalls by employing pre-synthesized protected amino acid fragments rather than single residue coupling. By defining Fragment X as Mpr(R2)-Harg(R3) and Fragment Y as Gly-Asp(OtBu), the synthesis effectively bypasses the problematic junctions where impurities typically form. Coupling these larger units reduces the total number of reaction cycles on the solid support, thereby minimizing the cumulative risk of racemization and incomplete reactions. This fragment-based condensation not only streamlines the workflow but also fundamentally alters the impurity profile of the crude peptide. The result is a crude product with significantly higher purity, which translates directly into reduced burden on downstream processing. For supply chain managers, this means a more predictable production timeline and a substantial reduction in the waste associated with failed batches or extensive rework.
Mechanistic Insights into Fragment-Based Solid-Phase Coupling
The core of this enhanced synthesis lies in the precise selection of coupling reagents and resin supports to facilitate the attachment of these complex fragments. The protocol utilizes carbodiimide-based activation, specifically favoring N,N-Diisopropylcarbodiimide (DIC) in conjunction with 1-hydroxybenzotriazole (HOBt). This combination is critical for activating the carboxyl groups of the protected fragments without inducing significant racemization at the chiral centers, a common concern in peptide chemistry. The reaction is conducted on amino resins with a substitution value ranging from 0.3 to 1.5 mmol/g, with Rink Amide MBHA resin identified as the preferred matrix. This resin choice is not arbitrary; its specific polymeric structure provides superior swelling in organic solvents like DMF and DCM, ensuring that the bulky fragments can diffuse freely to the reactive sites within the resin beads. 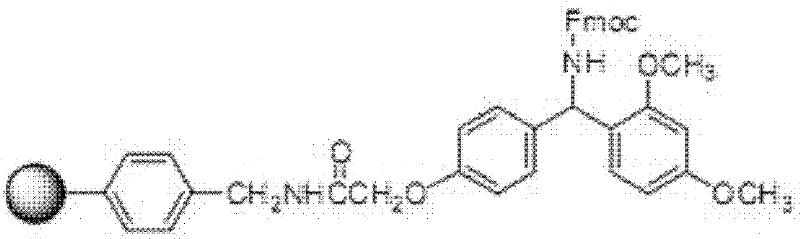
Following the assembly of the peptide chain on the solid support, the cleavage and cyclization steps are equally pivotal for maintaining product integrity. The acidolysis step employs a cocktail of trifluoroacetic acid (TFA), 1,2-Ethanedithiol (EDT), and water, typically in a ratio of 90:5:5. The EDT acts as a scavenger to prevent the alkylation of sensitive residues like Tryptophan and Methionine by carbocations generated during cleavage. Once the linear peptide is liberated, the formation of the critical disulfide bond between the Mercaptopropionic acid (Mpr) and Cysteine (Cys) residues is achieved through controlled oxidation. Using iodine titration in a dilute acetic acid solution allows for the precise formation of the intramolecular S-S bridge. This oxidative cyclization is monitored carefully to avoid over-oxidation to sulfonic acids, ensuring that the final cyclic heptapeptide retains its biological activity and structural conformation required for GPIIb/IIIa receptor binding.
How to Synthesize Eptifibatide Acetate Efficiently
The synthesis of Eptifibatide acetate via this fragment condensation route represents a robust pathway for manufacturing high-quality pharmaceutical intermediates. The process begins with the preparation of the specific resin-bound intermediates, followed by the sequential coupling of the defined fragments X and Y. Detailed operational parameters, including solvent volumes, reaction times, and temperature controls, are critical for reproducibility. The subsequent cleavage, oxidation, and purification steps require strict adherence to the specified gradients and reagent concentrations to achieve the reported purity levels of greater than 99.5%. For technical teams looking to implement this methodology, the following guide outlines the standardized operational framework derived from the patent data.
- Prepare Eptifibatide resin by sequentially coupling protected fragments X (Mpr-Harg) and Y (Gly-Asp) onto amino resin using DIC/HOBt activation.
- Perform acidolysis using a TFA/EDT/Water mixture to cleave the peptide from the resin and remove side-chain protecting groups simultaneously.
- Oxidize the linear peptide crude product using iodine titration to form the critical disulfide bond, followed by HPLC purification and salt exchange.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this advanced synthesis route offers profound implications for the economic and logistical aspects of Eptifibatide production. From a procurement perspective, the shift towards fragment-based coupling mitigates the risks associated with low-yielding steps that traditionally plague linear peptide synthesis. By reducing the formation of hard-to-separate impurities, the process inherently lowers the consumption of expensive chromatography media and organic solvents. This efficiency gain is not merely a laboratory metric; it scales directly into significant cost reductions in large-scale manufacturing environments. Furthermore, the simplified impurity profile reduces the complexity of quality control testing, allowing for faster batch release times and improved inventory turnover rates for finished goods.
- Cost Reduction in Manufacturing: The elimination of difficult purification steps drives down operational expenditures significantly. Traditional methods often require multiple rounds of preparative HPLC to remove deletion sequences, which consumes vast amounts of acetonitrile and column life. By preventing the formation of these impurities at the source through fragment coupling, the new method drastically reduces the load on purification infrastructure. This leads to a leaner manufacturing process where resources are focused on value-added production rather than waste remediation, ultimately enhancing the margin potential for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the variability of complex chemical syntheses. The robust nature of the fragment condensation method ensures higher consistency between batches, reducing the likelihood of production failures that can disrupt supply schedules. Additionally, the use of stable, protected fragments allows for better inventory management of starting materials. Suppliers can stock these key intermediates with longer shelf lives compared to activated single amino acids, providing a buffer against raw material shortages and enabling more responsive fulfillment of customer orders.
- Scalability and Environmental Compliance: Scaling peptide synthesis from grams to kilograms often exposes hidden inefficiencies, particularly regarding solvent waste. This optimized protocol, with its reduced cycle count and higher crude purity, is inherently more scalable. The decreased reliance on extensive chromatographic purification translates to a smaller environmental footprint, aligning with modern green chemistry initiatives. For multinational corporations with strict sustainability mandates, partnering with a supplier utilizing this efficient technology demonstrates a commitment to reducing industrial waste while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Eptifibatide acetate using this novel methodology. These insights are derived directly from the patent specifications and are intended to clarify the advantages of this specific synthetic route for stakeholders evaluating potential suppliers. Understanding these nuances is essential for making informed decisions about long-term sourcing strategies and technical partnerships.
Q: How does the fragment condensation method improve Eptifibatide purity?
A: By coupling pre-formed fragments like Mpr-Harg and Gly-Asp instead of single amino acids, the method avoids the formation of deletion sequences such as [-1Harg] and insertion impurities like [+1Gly], significantly simplifying downstream purification.
Q: What resin is preferred for this solid-phase synthesis?
A: While various resins like Rink Amide and Sieber resin are compatible, Rink Amide MBHA resin is specifically preferred in this protocol due to its optimal swelling properties and stability during the extended coupling cycles required for complex peptides.
Q: What is the critical step for forming the cyclic structure?
A: The formation of the disulfide bridge between the mercaptopropionic acid (Mpr) and Cysteine (Cys) residues is critical. This is achieved through controlled oxidation using iodine titration in an acetic acid solution to ensure correct cyclization without over-oxidation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eptifibatide Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the fragment condensation method are fully realized in our manufacturing facilities. We operate with stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Eptifibatide acetate meets the highest global pharmacopeial standards. Our commitment to technical precision ensures that the complex disulfide bridging and impurity profiles are managed with absolute consistency.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis platform, we can offer competitive pricing structures without compromising on the critical quality attributes of the peptide. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can secure your supply chain and drive value for your organization.
