Advanced Synthesis of 20-Keto Steroid Intermediates via TEMPO Catalysis for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic pathways for critical hormonal intermediates, particularly those serving as precursors for corticosteroids like cortisone and hydrocortisone. A recent technological breakthrough detailed in patent CN114315946A introduces a highly efficient preparation method for steroid intermediates, specifically targeting the synthesis of 20-keto steroid compounds. This innovation addresses long-standing challenges in the field by replacing hazardous and costly reagents with a greener, catalytic approach. The core of this advancement lies in a two-step sequence: first, the selective oxidation of a 20-hydroxymethyl group to a carboxylic acid using a TEMPO catalyst system, and second, an oxidative decarboxylation mediated by a Schiff base metal complex. For R&D directors and process chemists, this represents a significant leap forward in controlling impurity profiles and reaction conditions, moving away from the harsh environments of the past toward a more sustainable and controllable manufacturing paradigm that ensures consistent quality for downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 20-keto steroids from their 20-hydroxymethyl precursors has been plagued by significant operational and environmental drawbacks. The traditional route, as illustrated in the prior art, typically involves the use of N-chlorosuccinimide (NCS) in conjunction with dimethyl sulfide (DMS) to generate an aldehyde intermediate, followed by a rearrangement step. 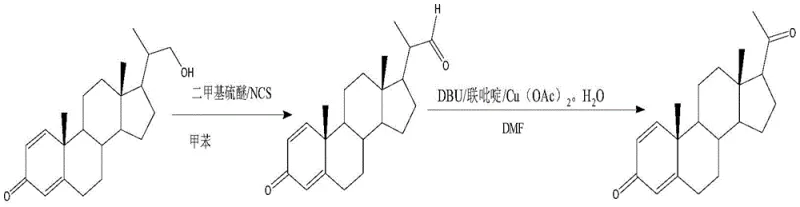 This legacy method suffers from severe disadvantages, primarily due to the use of dimethyl sulfide, a reagent notorious for its extremely unpleasant odor and high toxicity, which complicates waste treatment and poses health risks to plant personnel. Furthermore, these reactions often require rigorous low-temperature control to manage exotherms and selectivity, making the process difficult to scale safely. Alternative methods utilizing Dess-Martin periodinane offer better selectivity but are prohibitively expensive for large-scale production and generate substantial organic waste, rendering them economically unviable for commercial manufacturing of high-volume API intermediates.
This legacy method suffers from severe disadvantages, primarily due to the use of dimethyl sulfide, a reagent notorious for its extremely unpleasant odor and high toxicity, which complicates waste treatment and poses health risks to plant personnel. Furthermore, these reactions often require rigorous low-temperature control to manage exotherms and selectivity, making the process difficult to scale safely. Alternative methods utilizing Dess-Martin periodinane offer better selectivity but are prohibitively expensive for large-scale production and generate substantial organic waste, rendering them economically unviable for commercial manufacturing of high-volume API intermediates.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent employs a sophisticated yet practical catalytic system that fundamentally alters the reaction landscape. The new route utilizes a TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) catalyst combined with inexpensive hypochlorite salts and a mild alkali reagent to effect the initial oxidation. 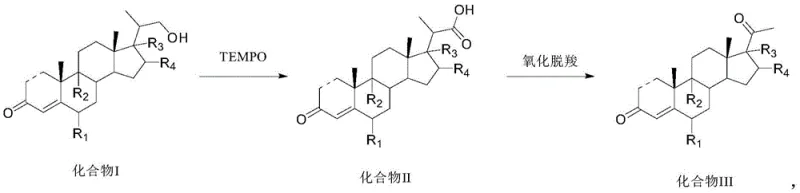 This is followed by a second step where the resulting carboxylic acid undergoes oxidative decarboxylation using a transition metal Schiff base complex and a periodate oxidant. This approach completely eliminates the need for malodorous DMS and expensive stoichiometric oxidants. The reaction conditions are remarkably mild, operating between 0°C and 30°C, which drastically reduces energy consumption for cooling and heating. By shifting to this catalytic paradigm, manufacturers can achieve superior reaction control, higher throughput, and a significantly cleaner process that aligns with modern green chemistry principles while maintaining the structural integrity of the sensitive steroid backbone.
This is followed by a second step where the resulting carboxylic acid undergoes oxidative decarboxylation using a transition metal Schiff base complex and a periodate oxidant. This approach completely eliminates the need for malodorous DMS and expensive stoichiometric oxidants. The reaction conditions are remarkably mild, operating between 0°C and 30°C, which drastically reduces energy consumption for cooling and heating. By shifting to this catalytic paradigm, manufacturers can achieve superior reaction control, higher throughput, and a significantly cleaner process that aligns with modern green chemistry principles while maintaining the structural integrity of the sensitive steroid backbone.
Mechanistic Insights into TEMPO-Catalyzed Oxidation and Decarboxylation
The success of this synthetic strategy hinges on the precise mechanistic control offered by the TEMPO catalytic cycle in the first step. TEMPO acts as a stable nitroxyl radical that mediates the oxidation of the primary alcohol at the C20 position to the corresponding carboxylic acid via an aldehyde intermediate, without over-oxidizing other sensitive functionalities on the steroid ring system. The presence of a bromide source, such as potassium bromide, facilitates the generation of the active oxoammonium species in situ when coupled with hypochlorite. This catalytic turnover is highly efficient, requiring only minute amounts of the TEMPO catalyst (molar ratios as low as 0.005:1), which minimizes metal-free organic impurities. The alkaline environment provided by bicarbonates ensures that the acidic byproducts are neutralized, preventing acid-catalyzed degradation of the steroid skeleton, a common issue in traditional acidic oxidation methods. This selectivity is crucial for maintaining the high HPLC purity observed in the examples, often exceeding 99%.
The second step involves a complex oxidative decarboxylation mechanism driven by the Schiff base metal complex. Transition metals such as Ruthenium, Manganese, or Iron, coordinated within a Schiff base ligand framework, activate the periodate oxidant to facilitate the removal of the carboxyl group as carbon dioxide. This radical-mediated process effectively shortens the side chain, converting the C21 carboxylic acid into the desired C20 ketone functionality. The choice of the metal center and the ligand environment allows for fine-tuning of the redox potential, ensuring that the decarboxylation proceeds rapidly at near-ambient temperatures (20-30°C). The use of organic bases like imidazole or DBU further assists in stabilizing intermediates and driving the equilibrium towards the ketone product. This mechanistic elegance results in a process that is not only chemically efficient but also robust against variations in raw material quality, providing a reliable pathway for generating high-purity intermediates essential for potent hormonal drugs.
How to Synthesize 20-Keto Steroid Intermediates Efficiently
Implementing this advanced synthesis requires careful attention to reagent addition rates and temperature control to maximize the benefits of the catalytic systems. The process begins with the dissolution of the 20-hydroxymethyl steroid precursor in a chlorinated or ester solvent, followed by the sequential addition of the bromide promoter, alkali buffer, and TEMPO catalyst. The oxidation is initiated by the controlled dosing of hypochlorite at low temperatures to manage the exotherm, ensuring the formation of the carboxylic acid intermediate with minimal side products. Once isolated, this intermediate is subjected to the decarboxylation conditions in a biphasic or mixed solvent system containing water and an organic co-solvent like THF. The detailed standardized operating procedures, including specific molar ratios, stirring speeds, and quenching protocols required to replicate the high yields reported in the patent, are outlined below.
- Oxidize the 20-hydroxymethyl steroid precursor (Compound I) using a TEMPO catalyst, hypochlorite, and an alkali reagent in a solvent like dichloromethane at 0-10°C to form the carboxylic acid intermediate (Compound II).
- Subject the resulting carboxylic acid (Compound II) to oxidative decarboxylation using a transition metal Schiff base complex (e.g., Ruthenium or Manganese), a periodate oxidant, and an organic base in a THF/water mixture at 20-30°C.
- Isolate the final 20-keto steroid product (Compound III) through standard workup procedures including filtration, extraction, and concentration, achieving high purity suitable for downstream hormone synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers transformative economic and logistical benefits that extend far beyond simple yield improvements. The elimination of dimethyl sulfide removes a major bottleneck related to environmental compliance and odor control, which often necessitates specialized scrubbing equipment and costly waste disposal contracts. By switching to hypochlorite and TEMPO, facilities can leverage widely available, commodity-grade chemicals that are significantly cheaper and easier to source globally than specialized reagents like Dess-Martin periodinane. This shift simplifies the supply chain, reducing the risk of production stoppages due to reagent shortages. Furthermore, the mild reaction temperatures reduce the load on plant cooling infrastructure, leading to lower utility costs and allowing existing reactors to be utilized more efficiently without requiring expensive retrofits for cryogenic capabilities.
- Cost Reduction in Manufacturing: The replacement of expensive stoichiometric oxidants with catalytic systems drives down the raw material cost per kilogram of the final intermediate substantially. The use of cheap hypochlorite and recoverable metal catalysts instead of precious metal reagents or high-cost organic oxidants means that the variable cost of production is drastically lowered. Additionally, the high purity of the crude product reduces the burden on downstream purification steps, saving solvents and time during crystallization or chromatography, which translates directly into improved margins for the final API.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified because the key reagents—TEMPO, hypochlorites, and simple metal salts—are bulk commodities with stable global supply chains, unlike niche fine chemicals that may have single-source suppliers. This diversification of the supply base mitigates the risk of geopolitical disruptions or vendor-specific quality issues. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in reagent quality, ensuring consistent batch-to-batch performance and reliable delivery schedules to downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The process is inherently safer and more scalable due to the absence of toxic, volatile sulfur compounds and the operation at near-ambient temperatures. This ease of scale-up allows manufacturers to increase batch sizes confidently, moving from pilot plant to commercial tonnage without encountering the heat transfer limitations typical of exothermic oxidations. From an environmental perspective, the reduction in hazardous waste and the use of less toxic reagents streamline regulatory approvals and reduce the carbon footprint of the manufacturing site, aligning with corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this steroid synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios.
Q: How does this new TEMPO-based route improve upon traditional NCS/DMS methods?
A: The traditional method relies on N-chlorosuccinimide (NCS) and dimethyl sulfide (DMS), which generates significant malodorous waste and requires strict low-temperature control. The new TEMPO/hypochlorite route operates at milder temperatures (0-10°C), eliminates the toxic DMS odor issue, and utilizes cheaper, more abundant oxidants, significantly improving the environmental profile and operational safety.
Q: What represents the key cost-saving advantage in the second oxidation step?
A: Conventional routes often employ expensive stoichiometric oxidants like Dess-Martin periodinane, which drastically increases raw material costs and generates heavy organic waste. This patent utilizes a catalytic system involving Schiff base metal complexes and periodate salts, which are far more cost-effective and allow for easier purification, thereby reducing the overall cost of goods sold (COGS) for the intermediate.
Q: Is this process scalable for industrial production of corticosteroids?
A: Yes, the process is specifically designed for scalability. By avoiding cryogenic conditions and hazardous reagents, and by demonstrating high yields (over 90% in examples) and high HPLC purity (up to 99.8%), the method addresses the critical bottlenecks of heat management and impurity control that typically hinder the scale-up of steroid oxidations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Steroid Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the TEMPO-catalyzed route described in CN114315946A and is fully prepared to implement this technology for our partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of steroid intermediate we produce adheres to the highest international standards required for hormone synthesis.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your needs, obtain specific COA data from our recent pilot runs, and review our comprehensive route feasibility assessments. Let us help you optimize your supply chain and reduce your manufacturing costs with our proven expertise in steroid chemistry.
