Advanced Manufacturing of Beta-Thymidine: A Technical Breakthrough for Global Pharmaceutical Supply Chains
The global demand for high-quality antiviral intermediates continues to surge, driven by the persistent need for effective treatments against HIV and other retroviral infections. At the forefront of this chemical landscape is beta-thymidine, a critical nucleoside analogue serving as the key intermediate for renowned medications such as zidovudine and stavudine. A pivotal advancement in the manufacturing of this essential compound is detailed in patent CN102086222B, which outlines a robust and economically superior preparation method. This technical disclosure represents a significant leap forward from earlier biosynthetic and chemical synthesis attempts, addressing long-standing issues regarding stereospecificity and cost-efficiency. By leveraging a novel sequence of dehydration, halogenation, and catalytic hydrogenation, the process transforms 5-methyluridine into beta-thymidine with remarkable precision. For R&D directors and procurement strategists, understanding the nuances of this patent is vital, as it offers a pathway to secure a more reliable beta-thymidine supplier capable of meeting stringent pharmaceutical standards without the baggage of legacy production inefficiencies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of beta-thymidine has been plagued by significant technical and economic hurdles that hindered efficient commercial scale-up of complex pharmaceutical intermediates. Early chemical processes often lacked the necessary stereospecificity, resulting in the co-generation of undesirable alpha-isomers alongside the target beta-form. This lack of selectivity not only depressed the overall yield but also necessitated complicated and costly separation technologies to isolate the pure product, thereby driving up the final cost of goods. Furthermore, a widely cited method from 1998, while an improvement, still relied on halo-acylation reactions that utilized propionyl bromide. The use of propionyl bromide introduced severe operational challenges, including intense corrosion of reaction vessels and piping, which escalated maintenance costs and posed safety risks. Additionally, these traditional routes often suffered from moderate yields and required multiple purification stages, increasing labor intensity and extending production lead times, making them less attractive for modern, high-volume manufacturing environments.
The Novel Approach
In stark contrast to these legacy methodologies, the innovative process defined in the patent introduces a streamlined synthetic strategy that fundamentally alters the reaction landscape for cost reduction in pharmaceutical intermediates manufacturing. By initiating the synthesis with a dehydration condensation step using diester carbonates instead of harsh acylating agents, the process establishes a stable 2,2'-anhydro intermediate that inherently favors the formation of the desired beta-configuration. This strategic shift eliminates the need for propionyl bromide, thereby removing the primary source of equipment corrosion and significantly lowering raw material expenses. The subsequent halogenation and catalytic hydrogenation steps are optimized to proceed under milder conditions, ensuring high conversion rates while minimizing the formation of by-products. This approach not only simplifies the workflow by reducing the number of required separation and purification operations to just two but also enhances the overall safety profile of the plant, making it an ideal candidate for sustainable and scalable industrial production.
Mechanistic Insights into Diester Carbonate Dehydration and Catalytic Hydrogenation
The core of this synthetic breakthrough lies in the precise control of stereochemistry through the formation of a rigid anhydro bridge. In the first stage, 5-methyluridine reacts with a diester carbonate, such as diphenyl carbonate or diethyl carbonate, in a polar solvent like DMF or acetonitrile at temperatures ranging from 90°C to 150°C. Under the catalytic influence of sodium hydroxide or potassium hydroxide, a nucleophilic attack occurs, leading to the elimination of water and the formation of the 2,2'-anhydro-5-methyluridine intermediate. This cyclic structure locks the sugar moiety in a specific conformation that prevents the formation of the alpha-anomer during the subsequent ring-opening step. The second stage involves the nucleophilic attack of a halide ion on this anhydro intermediate. By employing a halogenating system such as NaBr-HCl or HBr gas at controlled temperatures between 50°C and 80°C, the anhydro ring is opened selectively at the 2'-position. This regioselective ring opening is crucial as it ensures that the incoming halogen atom occupies the correct spatial orientation, setting the stage for the final reduction to yield exclusively the beta-isomer of thymidine.
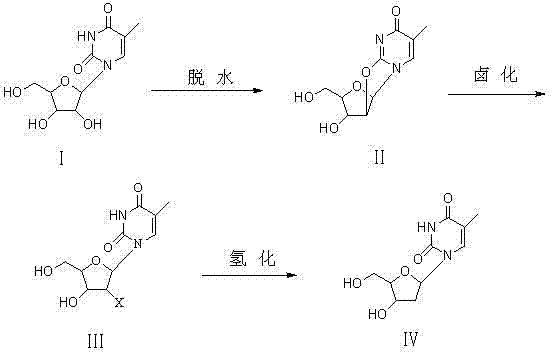
The final transformation involves the catalytic hydrogenation of the 2'-halo thymidine intermediate to produce the target beta-thymidine. This reduction is typically carried out in the presence of a reducing metal catalyst, such as nickel, zinc, or palladium on carbon, under a hydrogen pressure of 0.1 to 0.3 MPa. The mechanism here involves the replacement of the halogen atom at the 2'-position with a hydrogen atom, completing the deoxygenation process required to convert the ribose sugar into deoxyribose. The choice of catalyst and the careful control of temperature (20°C to 60°C) are critical parameters that influence the reaction rate and the purity of the final product. By avoiding harsh reducing conditions that might degrade the nucleobase, this method preserves the integrity of the thymine ring while achieving high-purity beta-thymidine with HPLC values exceeding 99.8%. This mechanistic elegance ensures that the impurity profile remains exceptionally clean, a factor of paramount importance for regulatory compliance in the pharmaceutical sector.
How to Synthesize Beta-Thymidine Efficiently
The synthesis of beta-thymidine via this patented route offers a clear and actionable pathway for laboratories and manufacturing facilities aiming to optimize their production capabilities. The process is distinguished by its operational simplicity and the use of readily available reagents, which collectively contribute to a more robust supply chain. To implement this method effectively, operators must focus on the precise stoichiometry of the diester carbonate and the strict control of reaction temperatures during the dehydration phase to maximize the yield of the anhydro intermediate. Following this, the halogenation step requires careful monitoring of pH and gas flow rates to ensure complete conversion without excessive degradation. The final hydrogenation step benefits from the use of activated catalysts to drive the reaction to completion within a reasonable timeframe. For a comprehensive understanding of the specific operational parameters, solvent ratios, and workup procedures, the detailed standardized synthesis steps are provided in the guide below.
- Dehydration: React 5-methyluridine with diester carbonate (e.g., diphenyl carbonate) at 90-150°C using NaOH/KOH catalyst to form 2,2'-anhydro-5-methyluridine.
- Halogenation: Treat the anhydro intermediate with a halogenating agent (HBr/HCl or NaBr-HCl system) at 50-80°C to obtain 2'-halo thymidine.
- Hydrogenation: Reduce the halide intermediate using a metal catalyst (Ni, Zn, or Pd/C) under hydrogen pressure (0.1-0.3 MPa) at 20-60°C to yield beta-thymidine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages that go beyond mere technical specifications. The elimination of corrosive reagents like propionyl bromide directly impacts the total cost of ownership by extending the lifespan of reactor vessels and reducing the frequency of equipment replacement and maintenance. Furthermore, the simplification of the purification process, which now requires only two separation steps compared to the multiple stages of older methods, significantly reduces labor intensity and solvent consumption. This streamlining of operations leads to substantial cost savings in terms of both utility usage and waste disposal, aligning with modern green chemistry initiatives. Additionally, the high conversion efficiency and yield stability of this process ensure a more predictable production schedule, thereby reducing lead time for high-purity pharmaceutical intermediates and enhancing the overall reliability of the supply chain for downstream API manufacturers.
- Cost Reduction in Manufacturing: The transition away from expensive and corrosive halogenating agents to a diester carbonate-based system fundamentally alters the cost structure of beta-thymidine production. By removing the need for specialized corrosion-resistant materials and reducing the consumption of high-cost reagents, manufacturers can achieve a leaner cost base. Moreover, the high yield of the anhydro intermediate and the subsequent steps minimizes raw material waste, ensuring that a greater proportion of the input 5-methyluridine is converted into saleable product. This efficiency gain allows for competitive pricing strategies without compromising on quality, providing a distinct market advantage in the sourcing of critical antiviral ingredients.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes significantly to supply chain resilience. The use of common, commercially available solvents and catalysts reduces the risk of raw material shortages that can plague specialized chemical supply chains. The process tolerance to slight variations in reaction conditions ensures consistent batch-to-batch quality, which is essential for maintaining long-term contracts with pharmaceutical clients. By stabilizing the production output and minimizing the occurrence of off-spec batches, suppliers can offer more reliable delivery schedules, thus supporting the continuous manufacturing needs of global drug producers who cannot afford interruptions in their API supply.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers a cleaner and more manageable pathway for expansion. The reduction in hazardous waste generation, particularly the avoidance of bromide-heavy waste streams associated with propionyl bromide, simplifies effluent treatment and lowers environmental compliance costs. The process is inherently designed for scale-up, with reaction conditions that are easily transferable from pilot plants to large-scale industrial reactors. This scalability ensures that suppliers can rapidly respond to increases in market demand, securing their position as a dependable partner for growing pharmaceutical projects while adhering to increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial implications of this beta-thymidine synthesis method. These insights are derived directly from the patent data and are intended to clarify the operational benefits for potential partners and technical stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines or for qualifying new suppliers.
Q: How does this new synthesis method improve upon traditional propionyl bromide routes?
A: Traditional methods utilizing propionyl bromide suffer from severe equipment corrosion, high raw material costs, and lower yields. The novel process described in patent CN102086222B replaces this with a diester carbonate dehydration system and a milder halogenation protocol, significantly reducing equipment maintenance costs and improving overall process safety and yield stability.
Q: What is the expected purity profile for beta-thymidine produced via this route?
A: The patented process is designed to achieve exceptional purity levels, specifically targeting an HPLC purity greater than 99.8%. This high level of chemical integrity is critical for downstream applications in antiviral drug manufacturing, ensuring minimal impurity carryover into final API products like zidovudine.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process is explicitly optimized for industrial scale-up. It simplifies the workflow by requiring only two separation and purification steps, drastically reducing labor intensity. The use of common solvents and standard catalytic hydrogenation conditions makes it highly adaptable for large-scale commercial manufacturing ranging from pilot batches to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Thymidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of beta-thymidine in the global fight against viral diseases and are committed to delivering this key intermediate with uncompromising quality and consistency. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of even the largest pharmaceutical contracts. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of beta-thymidine meets the high standards required for API synthesis, including the HPLC >99.8% benchmark highlighted in recent technological advancements. Our facility is equipped to handle the specific dehydration and hydrogenation chemistries required for this process, guaranteeing a supply that is both chemically superior and commercially viable.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how our optimized processes can reduce your overall procurement costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and accelerate your drug development timelines.
