Industrial Scale-Up of Beta-Thymidine: A Stereospecific Route for HIV Drug Intermediates
Introduction to Advanced Beta-Thymidine Manufacturing
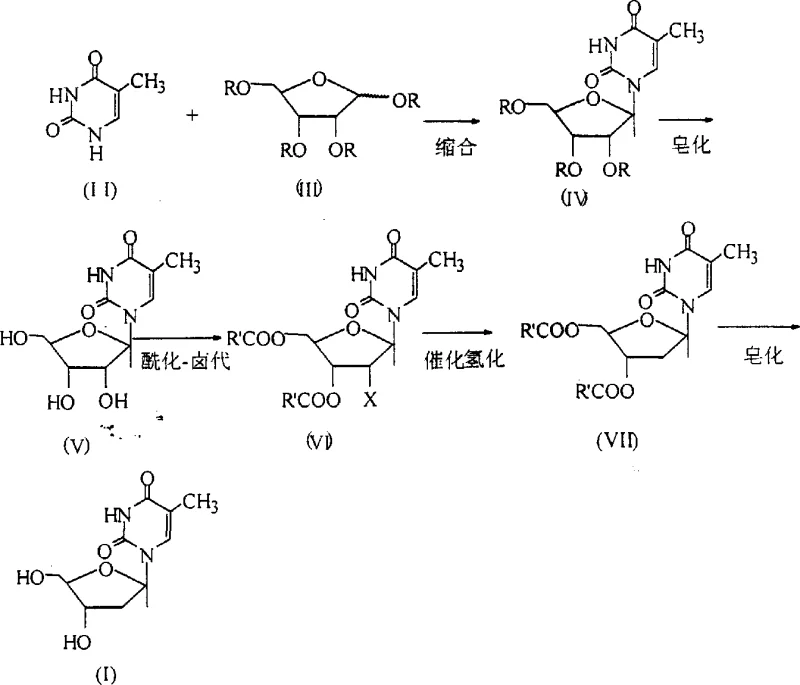
The pharmaceutical industry's demand for high-quality antiretroviral intermediates continues to drive innovation in synthetic methodology, particularly for critical compounds like beta-thymidine, the key precursor for Zidovudine (AZT). Patent CN1055293C discloses a robust, five-step chemical route designed specifically for industrial scalability, addressing long-standing challenges in stereoselectivity and cost efficiency. This technology represents a significant leap forward from earlier methods that relied on toxic mercury salts or produced complex mixtures of alpha and beta anomers requiring tedious separation. By integrating stereospecific glycosylation with efficient catalytic hydrodehalogenation, this process ensures a consistent supply of high-purity material suitable for GMP manufacturing environments. For procurement leaders and R&D directors, understanding the nuances of this pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deoxythymidine has been plagued by significant technical hurdles that impact both yield and environmental safety. Traditional approaches often utilized mercury salts to catalyze the condensation between thymine and ribose derivatives, a practice that introduces severe heavy metal contamination risks and complicates waste disposal protocols. Furthermore, many prior art methods lacked stereocontrol, resulting in the co-formation of alpha-isomers alongside the desired beta-product. This lack of stereospecificity necessitated complex downstream purification processes, such as repeated crystallizations or column chromatography, which drastically reduced overall throughput and increased production costs. The reliance on expensive precious metal catalysts for deoxygenation steps further exacerbated the economic burden, making cost reduction in pharmaceutical intermediate manufacturing a persistent challenge for producers.
The Novel Approach
The methodology outlined in the referenced patent overcomes these deficiencies through a cleverly designed sequence that prioritizes atom economy and operational simplicity. By employing a silylation protection strategy followed by Lewis acid-catalyzed condensation, the process achieves exceptional stereoselectivity, yielding the beta-isomer with a content exceeding 99 percent without the need for mercury. The route incorporates a telescoped design where key intermediates, such as the triacetyl-ribouridine and the halo-deoxy species, are carried forward without rigorous purification, significantly streamlining the workflow. Additionally, the substitution of expensive palladium catalysts with Raney-Ni for the hydrodehalogenation step demonstrates a commitment to cost-effective processing. This novel approach not only enhances the commercial scale-up of complex nucleoside analogs but also aligns with modern green chemistry principles by reducing hazardous waste generation.
Mechanistic Insights into Stereospecific Glycosylation and Hydrodehalogenation
The core of this synthesis lies in the precise control of the glycosidic bond formation and the subsequent selective removal of the 2-prime substituent. The initial condensation involves the reaction of silylated thymine with tetraacetyl-D-ribose in the presence of Lewis acids like tin tetrachloride. This catalytic environment promotes the formation of the oxocarbenium ion intermediate, which is attacked by the nucleobase from the beta-face due to steric and electronic influences exerted by the protecting groups and the catalyst coordination. Following condensation, the acetyl groups are removed via saponification to expose the hydroxyl functionalities necessary for the subsequent activation. The critical acylation-halogenation step converts the 2-prime hydroxyl into a good leaving group (typically a bromide), setting the stage for the reductive removal. Finally, the catalytic hydrogenation utilizes Raney-Ni to cleave the carbon-halogen bond, effectively installing the hydrogen atom at the 2-prime position while preserving the sensitive glycosidic linkage and the base moiety.
Impurity control is meticulously managed throughout this cascade, particularly during the final purification stage. Unlike conventional routes that might struggle to separate structurally similar byproducts like 5-methyluridine, this process leverages the unique complexation properties of boron compounds. During the final saponification and crystallization, the addition of boric acid or borax facilitates the selective precipitation of the target beta-thymidine. This mechanism likely involves the formation of transient borate esters with cis-diol containing impurities or the modulation of solubility profiles, allowing the pure product to crystallize out while impurities remain in the mother liquor. This results in a final product with HPLC purity levels consistently above 99.3 percent, meeting the rigorous specifications required for antiviral API synthesis without the need for resource-intensive chromatographic separation.
How to Synthesize Beta-Thymidine Efficiently
Executing this synthesis requires careful attention to reaction parameters, particularly temperature control during the exothermic condensation and halogenation steps. The process begins with the in-situ silylation of thymine, followed by the addition of the sugar donor and Lewis acid catalyst at controlled low temperatures to maximize beta-selectivity. Subsequent steps involve basic hydrolysis and reaction with acyl halides to generate the 2-prime halo intermediate, which is then subjected to hydrogenation. The detailed standardized operating procedures, including specific molar ratios, solvent choices, and workup protocols necessary to replicate these high yields in a pilot or production plant, are critical for successful technology transfer.
- Condensation of protected thymine with tetraacetyl-D-ribose using Lewis acid catalysts to form the beta-glycosidic bond with high stereoselectivity.
- Saponification of the triacetyl intermediate followed by acylation-halogenation to introduce the 2-prime leaving group.
- Catalytic hydrogenation using Raney-Ni for dehalogenation, followed by final saponification and boron-assisted crystallization for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain managers and procurement officers, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical feasibility. The elimination of mercury salts removes a major regulatory and environmental liability, simplifying the permitting process for manufacturing facilities and reducing the costs associated with hazardous waste treatment. Furthermore, the ability to telescope multiple steps without isolating intermediates significantly reduces the consumption of solvents and the requirement for large-scale drying and filtration equipment. This streamlined workflow translates directly into shorter cycle times and improved asset utilization, enhancing the overall responsiveness of the supply chain to market fluctuations. By optimizing the catalyst selection and purification strategy, manufacturers can achieve substantial cost savings while maintaining the high quality standards demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with Raney-Ni for the dehalogenation step represents a direct reduction in raw material expenses, as nickel-based catalysts are significantly more economical than palladium alternatives. Additionally, the avoidance of resin adsorption-desorption purification in favor of crystallization eliminates the recurring cost of chromatography resins and the associated solvent recovery overhead. The telescoping of reactions means fewer unit operations, which lowers energy consumption and labor costs per kilogram of finished product. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality of the high-purity pharmaceutical intermediate delivered to the customer.
- Enhanced Supply Chain Reliability: The robustness of this chemical route ensures consistent batch-to-batch reproducibility, a critical factor for maintaining uninterrupted supply lines for AIDS medication production. The use of readily available starting materials like thymine and tetraacetyl-D-ribose mitigates the risk of raw material shortages that can plague more exotic synthetic pathways. Moreover, the mild reaction conditions, particularly the ambient temperature and pressure hydrogenation, reduce the likelihood of equipment failure or safety incidents that could halt production. This operational stability provides procurement teams with greater confidence in long-term supply agreements and capacity planning.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial tonnage is facilitated by the absence of highly toxic reagents and the use of standard chemical engineering unit operations. The wastewater generated is easier to treat due to the lack of heavy metals, aligning with increasingly strict environmental regulations in key manufacturing regions. The high stereoselectivity minimizes the generation of isomeric waste, improving the overall mass balance of the process. This environmental compatibility not only future-proofs the manufacturing site against regulatory changes but also appeals to corporate sustainability goals, making the supply chain more resilient and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of beta-thymidine using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and quality assurances associated with this route. Understanding these details helps stakeholders make informed decisions regarding vendor qualification and process validation.
Q: How does this process ensure high stereospecificity for the beta-isomer?
A: The process utilizes a specific silylation protection strategy combined with Lewis acid catalysis (such as tin halides) during the condensation step. This controls the anomeric configuration, achieving a beta-isomer content of greater than 99 percent, effectively eliminating the need for difficult alpha/beta separation.
Q: What are the advantages of using Raney-Ni in the dehalogenation step?
A: Unlike precious metal catalysts like Palladium on Carbon, Raney-Ni offers a significantly lower cost profile while maintaining high activity under mild conditions (normal temperature and pressure). This reduces the overall catalyst cost and simplifies the equipment requirements for the hydrogenation unit.
Q: How is the final purity of beta-thymidine achieved without resin chromatography?
A: The method replaces traditional resin adsorption-desorption purification with a water-boron compound preferential crystallization technique. By adding boric acid or borax during the recrystallization phase, impurities such as 5-methyluridine are effectively removed, yielding product purity exceeding 99.3 percent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Thymidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against viral diseases. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering beta-thymidine that meets stringent purity specifications, utilizing rigorous QC labs to verify every batch against international pharmacopoeia standards. Our facility is equipped to handle the specific requirements of nucleoside chemistry, including moisture control and specialized hydrogenation capabilities, guaranteeing a product that supports your downstream API synthesis without deviation.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages inherent in this mercury-free, stereospecific process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in the production of life-saving pharmaceutical ingredients.
