Advanced Chiral Catalytic Synthesis of Ergothioneine for Industrial Scale-Up
The global demand for high-purity antioxidants in the cosmetic and pharmaceutical sectors has driven intense research into efficient synthetic routes for ergothioneine. A pivotal development in this field is documented in Chinese Patent CN111574458A, which discloses a robust, chiral catalytic synthesis method that fundamentally alters the manufacturing landscape for this valuable molecule. Unlike traditional pathways that struggle with solubility and purification issues, this innovative approach leverages asymmetric phase transfer catalysis to introduce chirality at a strategic late stage. By utilizing 1,3-dihydroxyacetone as a cost-effective starting material and employing specialized chiral quaternary ammonium salts, the process achieves exceptional enantiomeric control while maintaining intermediates in an organic-soluble state. This technical breakthrough not only addresses the critical need for optical purity but also resolves significant bottlenecks associated with downstream processing, positioning it as a preferred strategy for reliable ergothioneine intermediate supplier networks aiming for commercial excellence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of ergothioneine has been plagued by significant operational challenges that hinder cost reduction in cosmetic active manufacturing. Most prior art methodologies rely on histidine as the starting scaffold, necessitating multi-step reactions conducted predominantly in aqueous media. While water is a green solvent, the high water solubility of the intermediates and the final zwitterionic product creates a nightmare for isolation. Conventional processes require extensive lyophilization or complex ion-exchange chromatography to recover the product, leading to excessive energy consumption and substantial wastewater generation. Furthermore, the chiral center at the alpha-position of the carboxyl group in histidine-derived routes is prone to racemization under the acidic or basic conditions often required for functional group manipulation. This instability frequently results in suboptimal optical purity, forcing manufacturers to implement additional, yield-losing resolution steps to meet the stringent quality standards demanded by the skincare industry.
The Novel Approach
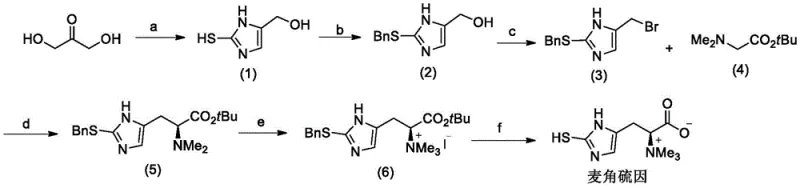
The methodology outlined in CN111574458A presents a paradigm shift by constructing the ergothioneine skeleton from 1,3-dihydroxyacetone rather than histidine. This route ensures that all key intermediates remain soluble in common organic solvents such as dichloromethane, ethyl acetate, and toluene throughout the synthesis. This solubility profile allows for standard unit operations like liquid-liquid extraction and crystallization, drastically simplifying the workup procedures compared to aqueous methods. The core innovation lies in the delayed introduction of chirality via a highly selective alkylation step, avoiding the early-stage racemization risks inherent in histidine chemistry. As illustrated in the reaction scheme below, the process flows smoothly from cyclization to protection, bromination, chiral coupling, methylation, and finally deprotection, offering a streamlined pathway that is inherently more adaptable to industrial scale-up.
Mechanistic Insights into Asymmetric Phase Transfer Catalysis
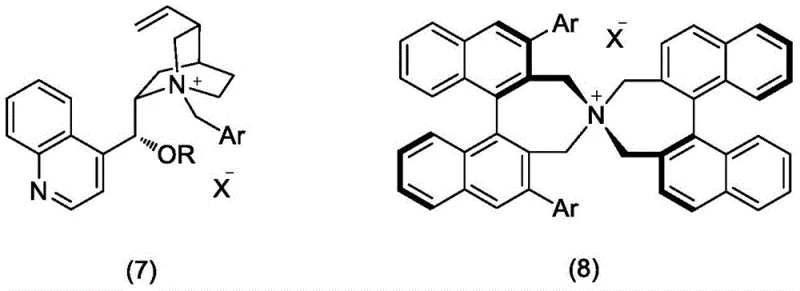
The heart of this synthetic strategy is the enantioselective alkylation step (Reaction d), where the prochiral glycine derivative reacts with the bromo-imidazole intermediate. This transformation is mediated by sophisticated chiral phase transfer catalysts, specifically cinchona alkaloid derivatives (Structure 7) or spiro-bis-naphthalene azepinium salts (Structure 8). In this biphasic system, the chiral cation of the catalyst forms a tight ion pair with the enolate of the glycine Schiff base in the organic phase. The bulky, rigid architecture of the catalyst creates a highly defined chiral environment that shields one face of the enolate, directing the nucleophilic attack on the electrophile exclusively from the opposite face. This precise stereocontrol is what enables the process to achieve enantiomeric excess (ee) values exceeding 98%, as evidenced by the experimental data in the patent.
Beyond stereocontrol, the choice of catalyst and conditions plays a vital role in impurity management. The use of mild bases like sodium hydroxide in conjunction with these catalysts prevents the degradation of the sensitive thioimidazole ring, a common side reaction in harsher alkaline environments. The catalyst structures shown below highlight the steric bulk provided by groups such as the anthracenyl or trifluorophenyl moieties, which are essential for discriminating between the enantiotopic faces of the substrate. By optimizing the catalyst loading to as low as 0.1 mol%, the process maintains high efficiency while minimizing the residual metal or organic impurities that could complicate the final purification, ensuring the production of high-purity ergothioneine suitable for sensitive dermal applications.
How to Synthesize Ergothioneine Efficiently
Implementing this synthesis requires careful attention to the sequential protection and deprotection strategies that define the route. The process begins with the formation of the thioimidazole core, followed by benzyl protection of the sulfur to prevent oxidation and side reactions during the subsequent alkylation. The critical chiral coupling step must be performed at controlled low temperatures (-10 to 30°C) to maximize stereoselectivity. Following the coupling, N-methylation establishes the permanent positive charge characteristic of ergothioneine, and the final hydrogenolysis step removes the benzyl group to reveal the free thiol. For detailed operational parameters, stoichiometry, and specific workup instructions, please refer to the standardized protocol below.
- Cyclization of 1,3-dihydroxyacetone with potassium thiocyanate and ammonium acetate to form the imidazole core.
- Protection of the sulfur atom with a benzyl group followed by bromination of the hydroxyl group.
- Asymmetric alkylation using a chiral phase transfer catalyst to introduce the amino acid side chain with high enantiomeric excess.
- N-methylation to form the quaternary ammonium salt, followed by catalytic hydrogenation to remove the protecting group and yield ergothioneine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from aqueous histidine-based routes to this organic-soluble chiral catalytic method offers profound logistical and economic benefits. The primary advantage lies in the drastic simplification of downstream processing. Because intermediates are organic-soluble, manufacturers can utilize high-throughput centrifuges and standard drying ovens rather than energy-intensive freeze dryers. This shift significantly reduces the utility load per kilogram of product, directly translating to lower manufacturing overheads. Additionally, the avoidance of large volumes of aqueous waste reduces the burden on wastewater treatment facilities, aligning production with increasingly strict environmental compliance regulations and reducing the risk of regulatory shutdowns.
- Cost Reduction in Manufacturing: The elimination of expensive chiral pool starting materials like histidine in favor of commodity chemicals like 1,3-dihydroxyacetone provides a fundamental raw material cost advantage. Furthermore, the high yield and selectivity of the chiral catalytic step minimize the loss of valuable intermediates, improving the overall mass balance of the process. By removing the need for complex chromatographic purification typically required to separate racemates or water-soluble impurities, the process achieves substantial cost savings in both consumables and labor hours.
- Enhanced Supply Chain Reliability: The reliance on robust, non-hazardous reagents and standard organic solvents ensures that the supply chain is less vulnerable to disruptions caused by the scarcity of specialized reagents. The scalability of phase transfer catalysis is well-documented in the fine chemical industry, meaning that scaling from pilot plant to commercial production involves minimal re-engineering. This predictability allows for more accurate lead time forecasting and ensures a consistent supply of high-purity ergothioneine to meet market demand fluctuations.
- Scalability and Environmental Compliance: The process operates under mild thermal conditions, mostly between 0°C and 50°C, which reduces the safety risks associated with exothermic runaways and lowers the energy required for heating or cooling reactors. The organic solvent system allows for efficient solvent recovery and recycling loops, further diminishing the environmental footprint. This green chemistry profile not only supports corporate sustainability goals but also facilitates smoother regulatory approvals in markets with stringent chemical safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on purity, scalability, and operational feasibility for potential partners.
Q: Why is the organic solvent-based route superior to traditional aqueous methods for ergothioneine?
A: Traditional methods often rely on water-soluble intermediates derived from histidine, making isolation and purification energy-intensive due to water removal. This novel route utilizes organic-soluble intermediates, allowing for standard extraction and crystallization, significantly simplifying downstream processing and reducing waste water generation.
Q: What level of optical purity can be achieved with this chiral catalytic method?
A: The patent demonstrates that using specific chiral phase transfer catalysts, such as cinchona alkaloid derivatives or spiro-bis-naphthalene salts, can achieve enantiomeric excess (ee) values of 98% to 99% at the key alkylation step, ensuring high optical purity in the final product without the need for difficult resolution steps.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is designed for industrial adaptability. It employs mild reaction conditions (0-50°C), avoids hazardous reagents where possible, and utilizes robust phase transfer catalysis which is well-established for scale-up. The ability to isolate intermediates as solids or oils in organic solvents further enhances its feasibility for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ergothioneine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this chiral catalytic technology for the global antioxidant market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this patent are fully realized in practical manufacturing. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical integrity of every batch of ergothioneine we produce.
We invite forward-thinking companies to collaborate with us to leverage this efficient synthesis route for their product pipelines. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply of this critical cosmetic active is both economically optimized and technically secure.
