Revolutionizing Ergothioneine Production: Advanced EanB Mutant Technology for Commercial Scale-Up
The global demand for high-purity antioxidants in the pharmaceutical and nutraceutical sectors has driven intense research into efficient synthesis pathways for ergothioneine. Patent CN114667346B introduces a groundbreaking advancement in this field by disclosing a novel oxidative sulfurase mutant, EanB, derived from Chlorobium limicola. This technology addresses the critical bottlenecks of traditional production methods by enabling a highly specific, anaerobic biosynthetic route. The core innovation lies in the enzyme's ability to catalyze the formation of a carbon-sulfur bond at the epsilon position of N(α)-trimethylhistidine (TMH) without the need for oxygen, a distinct advantage over aerobic pathways that often suffer from oxidative side reactions. This patent represents a significant leap forward for manufacturers seeking reliable ergothioneine suppliers who can guarantee consistent quality and supply continuity.
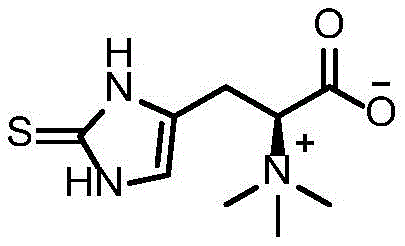
Ergothioneine, chemically known as 2-mercaptohistidine trimethyl inner salt, is a unique sulfur-containing amino acid with potent cytoprotective properties. Its market value is underpinned by its stability and efficacy in combating oxidative stress in various disease models, including neurodegenerative conditions. The structural integrity of the final product is paramount for its biological activity, and the enzymatic precision offered by the EanB mutant ensures the correct stereochemistry and purity required for high-end applications. By leveraging this patented biocatalytic approach, producers can bypass the variability inherent in natural extraction processes, offering a more robust solution for cost reduction in nutritional ingredients manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of ergothioneine has relied heavily on the submerged fermentation of edible mushrooms or chemical synthesis. While effective to a degree, mushroom fermentation presents substantial logistical and economic challenges that hinder scalability. The mycelial growth of fungi is inherently slow, typically requiring fermentation cycles of 7 to 10 days to achieve sufficient biomass accumulation. Furthermore, the target molecule accumulates primarily within the fungal mycelium, necessitating rigorous and costly downstream processing steps such as cell disruption, filtration, and complex extraction protocols to recover the product. These factors collectively result in prolonged lead times and elevated production costs, making it difficult to meet the surging global demand with a reliable agrochemical intermediate or pharma ingredient supply chain.
The Novel Approach
In stark contrast, the methodology described in CN114667346B utilizes a recombinant microbial system, specifically employing Escherichia coli as a host for the expression of the engineered EanB enzyme. This shift from fungal fermentation to bacterial biocatalysis fundamentally alters the production timeline and efficiency. The engineered strains can be cultivated rapidly in standard fermentation media, and the enzymatic conversion of the substrate TMH to ergothioneine can be achieved within a mere 24 hours under optimized conditions. This drastic reduction in process time, coupled with the elimination of complex mycelial extraction steps, streamlines the entire manufacturing workflow. For procurement managers, this translates to a more agile supply chain capable of responding quickly to market fluctuations while maintaining stringent purity specifications.
Mechanistic Insights into EanB-Catalyzed Anaerobic Sulfuration
The core of this technological breakthrough is the protein engineering of the EanB oxidative sulfurase. The patent details the identification of specific amino acid residues that, when mutated, significantly enhance the enzyme's catalytic turnover. Through error-prone PCR and site-directed mutagenesis, variants such as EanB-888 were developed, featuring mutations at positions 75 (Isoleucine to Arginine) and 369 (Glutamate to Proline). These structural modifications optimize the active site geometry, facilitating a more efficient nucleophilic attack during the sulfur transfer process. The mechanism proceeds under anaerobic conditions, where the enzyme catalyzes the direct insertion of sulfur from a donor like sodium thiosulfate into the imidazole ring of the histidine derivative. This specificity minimizes the formation of oxidative byproducts, ensuring a cleaner reaction profile that is easier to purify.
From an impurity control perspective, the high specificity of the EanB mutant is a critical advantage for R&D directors focused on regulatory compliance. Traditional chemical synthesis often generates a complex spectrum of regioisomers and side products that require extensive chromatographic purification, leading to yield losses. The biocatalytic route, however, leverages the innate stereoselectivity of the enzyme to produce the L-ergothioneine isomer exclusively. The patent data indicates that the EanB-888 mutant exhibits relative enzyme activity up to 480% compared to the wild type, demonstrating that the protein engineering efforts have successfully removed kinetic barriers. This high activity allows for lower enzyme loading or shorter reaction times, directly impacting the cost of goods sold and enhancing the overall process economics without compromising the chemical identity of the final API intermediate.
How to Synthesize Ergothioneine Efficiently
The implementation of this synthesis route involves a straightforward yet highly controlled bioprocess. It begins with the construction of a recombinant plasmid carrying the optimized EanB gene sequence, which is then transformed into a competent E. coli host strain. Following fermentation and induction, the resulting biomass serves as the biocatalyst. The reaction is conducted in a buffered aqueous system containing the substrate TMH and a sulfur source. Detailed standardized synthetic steps are provided in the guide below to ensure reproducibility and optimal yield.
- Construct recombinant expression vectors containing the mutated EanB gene (e.g., SEQ ID NO: 4) and transform into host cells like E. coli BL21(DE3).
- Cultivate the engineered strains in optimized fermentation media (e.g., TB medium) to induce high-level expression of the EanB mutant enzyme.
- Perform the biocatalytic reaction by mixing the enzyme source with N(α)-trimethylhistidine (TMH) and sodium thiosulfate under anaerobic conditions at pH 8.0.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the bottom line and supply continuity, the transition to this enzymatic technology offers compelling strategic benefits. The primary driver for adoption is the potential for substantial cost savings derived from process intensification. By replacing week-long fungal fermentation cycles with a rapid 24-hour enzymatic conversion, manufacturers can significantly increase facility throughput without expanding physical infrastructure. This efficiency gain reduces the capital expenditure per unit of production and lowers utility consumption, contributing to a more sustainable and cost-effective manufacturing model. Additionally, the use of E. coli, a well-characterized and industrially robust host, mitigates the risks associated with specialized fungal cultivation, ensuring a more stable and predictable supply of raw materials.
- Cost Reduction in Manufacturing: The enhanced catalytic efficiency of the EanB mutant directly correlates to reduced operational expenses. Because the mutant enzyme displays nearly five-fold higher activity than the wild type, manufacturers can achieve target conversion rates with significantly lower enzyme loading or in shorter timeframes. This efficiency eliminates the need for expensive and time-consuming downstream purification steps associated with mycelial extraction, such as cell wall disruption and solid-liquid separation. Consequently, the overall cost of production is drastically simplified, allowing for more competitive pricing structures in the global market for high-purity ergothioneine.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the decoupling of production from seasonal or biological constraints typical of mushroom farming. The recombinant bacterial system operates independently of agricultural cycles, enabling year-round production with consistent batch-to-batch quality. The rapid reaction kinetics mean that inventory turnover is accelerated, reducing the need for large safety stocks and freeing up working capital. For supply chain heads, this reliability ensures that delivery schedules can be met with greater precision, reducing lead time for high-purity ergothioneine intermediates and strengthening partnerships with downstream formulators.
- Scalability and Environmental Compliance: The scalability of this process is supported by the use of standard fermentation equipment compatible with E. coli cultivation, facilitating seamless scale-up from pilot to commercial volumes. Furthermore, the anaerobic nature of the reaction and the aqueous solvent system align with green chemistry principles, minimizing the generation of hazardous organic waste. The absence of heavy metal catalysts, often required in chemical synthesis, simplifies waste treatment and ensures compliance with stringent environmental regulations. This environmental compatibility not only reduces disposal costs but also enhances the brand value of the final product as a sustainably sourced ingredient.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of the EanB mutant technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What are the key advantages of the EanB mutant over wild-type enzymes?
A: The EanB-888 mutant exhibits significantly enhanced catalytic activity, approximately 4.8 times higher than the wild-type enzyme, allowing for faster conversion rates and reduced enzyme loading requirements.
Q: How does this enzymatic method compare to traditional mushroom fermentation?
A: Unlike traditional mushroom fermentation which requires 7-10 days and complex mycelium extraction, this enzymatic route utilizes fast-growing E. coli hosts and achieves high conversion within 24 hours, drastically simplifying downstream processing.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of standard E. coli expression systems and robust reaction conditions (pH 8.0, 37°C) makes this technology highly scalable for commercial production of high-purity ergothioneine intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ergothioneine Supplier
As the industry moves towards more efficient and sustainable manufacturing practices, NINGBO INNO PHARMCHEM stands ready to support your transition to advanced biocatalytic routes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising lab-scale results of patents like CN114667346B can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ergothioneine meets the highest international standards for pharmaceutical and nutritional applications.
We invite you to engage with our technical procurement team to discuss how this technology can be integrated into your supply chain. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are available to provide specific COA data and comprehensive route feasibility assessments, helping you make informed decisions that drive value and efficiency in your operations.
