Advanced Catalytic Hydrogenation Route for High-Purity Rivastigmine Intermediate Manufacturing
Advanced Catalytic Hydrogenation Route for High-Purity Rivastigmine Intermediate Manufacturing
The pharmaceutical landscape for neurodegenerative disorders continues to evolve, with Rivastigmine remaining a cornerstone therapy for moderate Alzheimer's disease. Central to the efficient manufacturing of this carbamate acetylcholinesterase inhibitor is the availability of its key precursor, 3-(1-dimethylaminoethyl)phenol. Recent intellectual property developments, specifically patent CN101270058B, have unveiled a transformative synthetic methodology that addresses long-standing inefficiencies in producing this critical intermediate. This patent discloses a robust catalytic hydrogenation process that replaces archaic reduction techniques, delivering exceptional purity and yield profiles essential for modern GMP manufacturing. By leveraging this technology, manufacturers can significantly enhance the quality of the supply chain for Alzheimer's therapeutics, ensuring that the final API meets stringent regulatory standards while optimizing production economics.
The Limitations of Conventional Methods vs. The Novel Approach
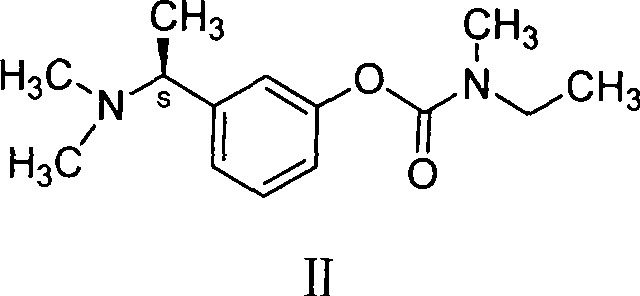
The Limitations of Conventional Methods
Historically, the synthesis of 3-(1-dimethylaminoethyl)phenol has relied on reductive amination protocols utilizing formaldehyde and formic acid systems, often referred to as Eschweiler-Clarke type conditions. As documented in legacy literature such as J. Chem. Soc., 1929, this approach involves reacting m-hydroxyphenylacetamide in a formic acid medium. However, this conventional pathway is plagued by severe operational and qualitative drawbacks that hinder large-scale efficiency. The reaction environment is highly corrosive and generates substantial amounts of byproducts, resulting in a crude product content that typically fails to exceed 80%. Furthermore, the isolation of the product is notoriously difficult due to the high solubility of the amine salts in the acidic medium, leading to significant yield losses during extraction, often capping overall recovery at merely 60%. Perhaps most critically for downstream processing, the product obtained via this route is frequently an impure brown oil rather than a crystalline solid, complicating purification and necessitating energy-intensive distillation or chromatography steps to meet pharmaceutical grade specifications.
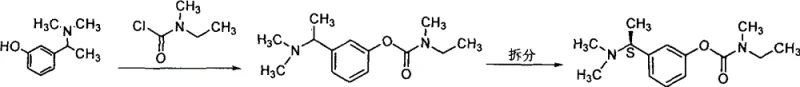
The Novel Approach
In stark contrast to the cumbersome formic acid reduction, the novel approach detailed in CN101270058B introduces a clean catalytic hydrogenation strategy that fundamentally alters the reaction landscape. This method utilizes m-hydroxyphenylacetamide and paraformaldehyde as starting materials, reacting them under a hydrogen atmosphere in the presence of a heterogeneous catalyst. By shifting from a stoichiometric chemical reduction to a catalytic process, the invention effectively eliminates the generation of formic acid waste streams and the associated extraction nightmares. The result is a process capable of achieving molar yields approaching 100%, with the product isolating as a high-purity, off-white to white solid. This dramatic improvement in physical form—from brown oil to crystalline solid—simplifies the post-reaction workup to mere filtration and crystallization, removing the need for complex separations. The operational simplicity, combined with the ability to run the reaction at mild temperatures and pressures, positions this hydrogenation route as the superior choice for industrial scale-up.
Mechanistic Insights into Pd/C and Raney Ni Catalyzed Reductive Amination
The core of this technological advancement lies in the mechanism of catalytic reductive amination, where the carbonyl group of the in-situ generated imine intermediate is reduced by molecular hydrogen on the surface of a metal catalyst. In this specific transformation, m-hydroxyphenylacetamide reacts with paraformaldehyde to form an iminium ion or Schiff base intermediate, which is then rapidly hydrogenated. The use of noble metal catalysts like 10% Palladium on Carbon (Pd/C) or base metal catalysts like Raney Nickel provides active sites for hydrogen dissociation and subsequent transfer to the organic substrate. The patent data indicates that the reaction proceeds efficiently at relatively low hydrogen pressures, preferably between 1 to 4 atmospheres, and mild temperatures ranging from 15°C to 30°C. This mildness is crucial for preserving the integrity of the phenolic moiety and preventing over-reduction or side reactions that often plague harsher chemical reductions. The catalyst facilitates the addition of hydrogen across the carbon-nitrogen double bond with high stereoselectivity and chemoselectivity, ensuring that the aromatic ring and other functional groups remain untouched.
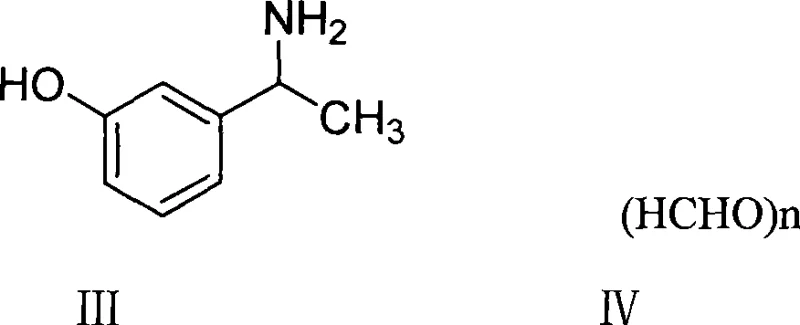
Impurity control in this hydrogenation process is inherently superior due to the cleanliness of the catalytic cycle. Unlike the formic acid method, which introduces carbonaceous impurities and difficult-to-remove salts, the hydrogenation byproduct is essentially non-existent or easily removable water. The patent examples demonstrate that HPLC purity can consistently reach levels between 98.5% and 99.2% directly after crystallization, without the need for extensive chromatographic purification. The choice of solvent plays a pivotal role in this purity profile; lower alcohols such as methanol and ethanol are preferred as they provide excellent solubility for the reactants while allowing the product to crystallize out upon concentration. The removal of the catalyst via simple filtration post-reaction ensures that no heavy metal residues contaminate the final product, provided standard washing protocols are followed. This mechanistic clarity allows for precise process control, making it easier to validate the manufacturing process for regulatory submissions.
How to Synthesize 3-(1-Dimethylaminoethyl)phenol Efficiently
Implementing this synthesis requires careful attention to the hydrogenation parameters to maximize safety and yield. The process begins with the suspension of the catalyst in the alcoholic solvent, followed by the addition of the amide and paraformaldehyde sources. It is critical to ensure that the reactor atmosphere is thoroughly purged with inert gas before introducing hydrogen to prevent the formation of explosive mixtures. Once the reaction conditions of 1-4 atm pressure and ambient temperature are established, the mixture is stirred until TLC or HPLC analysis confirms the complete consumption of the starting m-hydroxyphenylacetamide. The detailed standardized operating procedures, including specific catalyst loading ratios and crystallization cooling rates, are outlined in the technical guide below.
- Charge a hydrogenation reactor with m-hydroxyphenylacetamide, paraformaldehyde, and a suitable alcohol solvent such as methanol or ethanol.
- Add the heterogeneous catalyst, specifically 10% Pd/C or Raney Nickel, and replace the atmosphere with hydrogen gas.
- Maintain reaction temperature between 15-30°C and hydrogen pressure at 1-4 atm until conversion is complete, followed by filtration and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this hydrogenation-based synthesis represents a significant opportunity for cost optimization and risk mitigation. The elimination of formic acid not only reduces raw material costs but also drastically lowers the expense associated with waste treatment and environmental compliance. Traditional methods generate acidic wastewater that requires neutralization and specialized disposal, whereas the new catalytic route produces a much cleaner effluent profile. Furthermore, the dramatic increase in yield from approximately 60% to nearly quantitative levels means that less raw material is required to produce the same amount of finished intermediate, directly improving the cost of goods sold (COGS). The ability to isolate the product as a solid rather than an oil also reduces packaging and storage complexities, enhancing overall logistics efficiency.
- Cost Reduction in Manufacturing: The transition to catalytic hydrogenation removes the dependency on stoichiometric amounts of formic acid, a reagent that contributes significantly to both material costs and downstream processing expenses. By achieving near-theoretical yields, the process minimizes raw material waste, ensuring that every kilogram of m-hydroxyphenylacetamide is converted into valuable product. Additionally, the simplified workup procedure, which relies on filtration and crystallization rather than complex extractions, reduces labor hours and solvent consumption, leading to substantial operational savings.
- Enhanced Supply Chain Reliability: The robustness of this hydrogenation method ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed purification steps. The use of common, commercially available solvents like methanol and ethanol, along with standard heterogeneous catalysts, mitigates the risk of supply shortages for specialized reagents. This reliability is crucial for maintaining continuous production schedules for high-demand pharmaceutical intermediates like Rivastigmine precursors.
- Scalability and Environmental Compliance: The process operates under mild conditions (low pressure and temperature), making it highly scalable from pilot plants to multi-ton commercial reactors without requiring exotic high-pressure equipment. The reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, facilitating easier permitting and long-term sustainability for manufacturing sites. This green chemistry approach future-proofs the supply chain against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What are the primary advantages of the hydrogenation method over the traditional formic acid reduction?
A: The hydrogenation method eliminates the use of formic acid, which traditionally causes difficult extractions and generates significant byproducts. This new route achieves near-quantitative yields (>99%) and produces a white solid with purity exceeding 98%, compared to the brown, low-purity oil obtained via the conventional Eschweiler-Clarke type reduction.
Q: Which catalysts are most effective for this reductive amination process?
A: The patent specifies that metal or metal-carbon catalysts are highly effective, with a preference for 10% or 5% Palladium on Carbon (Pd/C) and Raney Nickel. These catalysts facilitate the reaction under mild conditions (15-30°C) and low hydrogen pressures (1-4 atm).
Q: How does this process impact the scalability of Rivastigmine production?
A: By utilizing standard hydrogenation equipment and common solvents like methanol or ethanol, the process is inherently scalable for industrial production. The simplicity of the work-up—filtration followed by concentration and crystallization—removes complex purification bottlenecks associated with older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(1-Dimethylaminoethyl)phenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of Alzheimer's medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of multinational pharmaceutical partners. We are committed to delivering 3-(1-dimethylaminoethyl)phenol with stringent purity specifications, utilizing advanced analytical capabilities in our rigorous QC labs to verify that every batch exceeds the 98% purity threshold required for API synthesis. Our facility is equipped to handle the specific hydrogenation requirements of this process safely and efficiently.
We invite you to collaborate with us to leverage this superior synthetic route for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's economic and operational performance.
