Scalable Synthesis of Chiral Rivastigmine Intermediates via Advanced Catalytic Reduction
Introduction to Advanced Chiral Synthesis Technologies
The pharmaceutical industry's demand for high-purity chiral intermediates, particularly for neurodegenerative treatments like Alzheimer's disease, necessitates robust and scalable synthetic methodologies. Patent CN102264672A introduces a transformative catalytic process for the preparation of optically active compounds, specifically focusing on the synthesis of (S)-3-(1-dimethylaminoethyl)phenol, a critical precursor for Rivastigmine. This technology leverages asymmetric transfer hydrogenation to achieve superior stereocontrol without the thermodynamic inefficiencies of prior art methods. By utilizing chiral transition metal catalysts, manufacturers can bypass traditional resolution steps that often cap theoretical yields at 50%. The strategic implementation of this route allows for the direct conversion of readily available ketones into high-value chiral alcohols with exceptional enantiomeric purity.

This methodology represents a significant leap forward in process chemistry, addressing the long-standing challenge of maintaining stereochemical integrity while minimizing operational complexity. The ability to produce commercially useful enantiomeric ratios directly from the catalytic step reduces the burden on downstream purification units. For global supply chains, this translates to a more reliable source of key building blocks, ensuring continuity for the production of life-saving medications. The patent outlines a versatile approach applicable to various substituted hydroxybenzophenones, making it a valuable asset for diverse pharmaceutical portfolios seeking cost-effective and environmentally conscious manufacturing solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing routes for chiral aminoalcohols often rely on classical resolution techniques or non-selective reduction followed by tedious separation processes. These legacy methods are inherently inefficient, frequently suffering from low atom economy and the generation of substantial waste streams due to the discard of the unwanted enantiomer. Furthermore, conventional catalytic hydrogenation typically requires high-pressure equipment and rigorous safety protocols to handle molecular hydrogen, which increases both capital expenditure and operational risk. In many existing protocols, the presence of free hydroxyl groups necessitates complex protection and deprotection sequences to prevent catalyst poisoning or side reactions, adding multiple steps that erode overall yield and increase production time. These thermodynamic and economic inefficiencies create bottlenecks in large-scale production, making it difficult to meet the growing global demand for affordable therapeutics.
The Novel Approach
The innovative process described in the patent utilizes asymmetric transfer hydrogenation to overcome these historical barriers, offering a streamlined pathway to high-purity intermediates. By employing a chiral Ruthenium-based catalyst system, the reaction achieves high conversion rates and exceptional stereoselectivity under mild conditions, typically using formic acid and triethylamine as a safe hydrogen source. A distinct advantage of this novel approach is its chemoselectivity, which allows for the reduction of the ketone functionality in the presence of a free phenolic hydroxyl group without the need for protecting groups. This simplification drastically reduces the number of synthetic steps and the consumption of auxiliary reagents. The result is a more sustainable and economically viable process that delivers the desired chiral alcohol with an enantiomeric excess often exceeding 96% directly from the reactor, setting a new standard for efficiency in fine chemical manufacturing.
Mechanistic Insights into Ru-Catalyzed Asymmetric Transfer Hydrogenation
The core of this technological breakthrough lies in the sophisticated mechanism of the chiral transition metal catalyst, specifically the Ruthenium-TsDPEN complex. This catalyst operates through a concerted outer-sphere mechanism where the hydride is transferred from the metal center to the carbonyl carbon while a proton is simultaneously delivered from the ligand nitrogen to the carbonyl oxygen. This bifunctional activation ensures a highly organized transition state that dictates the stereochemical outcome of the reduction. The specific geometry of the chiral diamine ligand creates a steric environment that favors the formation of one enantiomer over the other, effectively discriminating between the prochiral faces of the ketone substrate. Understanding this mechanistic nuance is crucial for R&D teams aiming to optimize reaction parameters such as temperature and solvent composition to maximize turnover numbers.
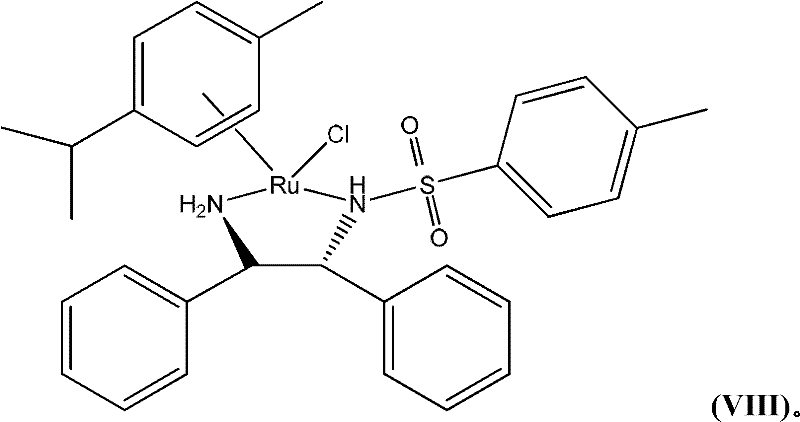
Impurity control is intrinsically linked to the stability and selectivity of this catalytic system. The patent highlights that the catalyst maintains activity even in the presence of potentially coordinating functional groups like phenols, which typically deactivate standard hydrogenation catalysts. This robustness minimizes the formation of over-reduced byproducts or racemic material that could complicate downstream purification. The reaction conditions, typically maintained around 70°C in alcoholic solvents, are optimized to balance reaction rate with catalyst longevity. By carefully controlling the ratio of hydrogen donor to substrate and the catalyst loading, manufacturers can suppress side reactions and ensure that the impurity profile remains within strict regulatory limits. This level of control is essential for producing pharmaceutical intermediates that require consistent quality batch after batch.
How to Synthesize (S)-3-(1-dimethylaminoethyl)phenol Efficiently
The synthesis of this critical Rivastigmine intermediate begins with the asymmetric reduction of 1-(3-hydroxyphenyl)ethanone, followed by a sequence of activation and substitution steps to install the amine functionality. The initial reduction step is the most critical, establishing the chiral center with high fidelity using the Ru-catalyst system described previously. Once the chiral alcohol is obtained, it undergoes activation, typically via mesylation, to convert the hydroxyl group into a suitable leaving group for nucleophilic attack. This is followed by displacement with dimethylamine and subsequent hydrolysis to reveal the final phenolic structure. The detailed standardized synthetic steps for executing this transformation with optimal yield and purity are provided in the guide below.
- Perform asymmetric catalytic reduction of 1-(3-hydroxyphenyl)ethanone using a chiral Ruthenium-TsDPEN catalyst and formic acid/triethylamine as the hydrogen source.
- Activate the resulting chiral alcohol via double mesylation using methanesulfonic anhydride to create a leaving group for nucleophilic attack.
- Execute nucleophilic substitution with dimethylamine followed by hydrolysis to regenerate the phenolic hydroxyl group and yield the final chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology offers profound strategic benefits beyond mere technical superiority. The elimination of high-pressure hydrogenation equipment and the reduction in synthetic steps directly correlate to lower capital intensity and reduced operational overhead. By avoiding the use of hazardous high-pressure gases and minimizing the number of unit operations, facilities can achieve significant cost reductions in manufacturing while enhancing workplace safety. The high selectivity of the process means less raw material is wasted on unwanted isomers, leading to better atom economy and lower costs for starting materials. These efficiencies compound to create a more resilient supply chain capable of responding to market fluctuations without compromising on margin or delivery timelines.
- Cost Reduction in Manufacturing: The streamlined nature of this process eliminates the need for expensive protecting group chemistry and reduces solvent consumption across fewer reaction steps. By operating at atmospheric pressure with liquid hydrogen donors, the process avoids the high energy costs and specialized infrastructure associated with gaseous hydrogen handling. The high catalyst turnover allows for the use of precious metals at very low loadings, further driving down the cost of goods sold. These factors combine to offer substantial cost savings that can be passed down the supply chain or reinvested into further process optimization.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system ensures consistent performance across different batches, reducing the risk of production delays caused by failed reactions or off-spec material. The use of common, commercially available solvents and reagents mitigates the risk of supply disruptions for exotic chemicals. Furthermore, the scalability of the transfer hydrogenation method allows for seamless transition from pilot scale to full commercial production, ensuring that supply can be ramped up quickly to meet surging demand for Alzheimer's therapeutics without lengthy requalification periods.
- Scalability and Environmental Compliance: This green chemistry approach significantly reduces the environmental footprint of the manufacturing process by minimizing waste generation and energy consumption. The absence of heavy metal waste streams typical of stoichiometric reductions simplifies wastewater treatment and disposal compliance. The process is inherently safer and more sustainable, aligning with modern corporate sustainability goals and regulatory expectations for greener pharmaceutical manufacturing. This environmental stewardship not only reduces liability but also enhances the brand value of the final pharmaceutical product in an increasingly eco-conscious market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric transfer hydrogenation technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing frameworks. The clarity provided here aims to remove ambiguity regarding catalyst performance, safety profiles, and downstream processing requirements.
Q: Why is asymmetric transfer hydrogenation preferred over high-pressure hydrogenation for this intermediate?
A: Transfer hydrogenation eliminates the need for specialized high-pressure hydrogenation equipment, significantly reducing capital expenditure and safety risks associated with handling hydrogen gas at scale. Furthermore, the use of formic acid/triethylamine azeotropes allows for milder reaction conditions that preserve sensitive functional groups without requiring extensive protection strategies.
Q: How does the process handle the free phenolic hydroxyl group during reduction?
A: The patented Ruthenium-TsDPEN catalyst system exhibits high chemoselectivity, allowing for the direct reduction of the ketone moiety in the presence of a free phenolic hydroxyl group. This avoids the costly and wasteful steps of protecting and deprotecting the phenol, streamlining the overall synthetic route and improving atom economy.
Q: What enantiomeric excess can be achieved with this catalytic method?
A: The process initially yields products with high enantiomeric excess, often exceeding 96% directly from the reaction. Through optimized crystallization techniques described in the patent, the enantiomeric purity can be further upgraded to greater than 99.5%, meeting the stringent requirements for active pharmaceutical ingredient (API) intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivastigmine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and production of effective Alzheimer's treatments. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic processes described in patent CN102264672A can be successfully implemented at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (S)-3-(1-dimethylaminoethyl)phenol meets the highest standards required for API synthesis. Our commitment to excellence ensures that your supply chain remains uninterrupted and compliant with global regulatory bodies.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's efficiency and profitability. Let us be your partner in delivering high-performance pharmaceutical intermediates to the market faster and more economically.
