Advanced Purification Technology for High-Purity Cefprozil Manufacturing and Commercial Scale-Up
Advanced Purification Technology for High-Purity Cefprozil Manufacturing and Commercial Scale-Up
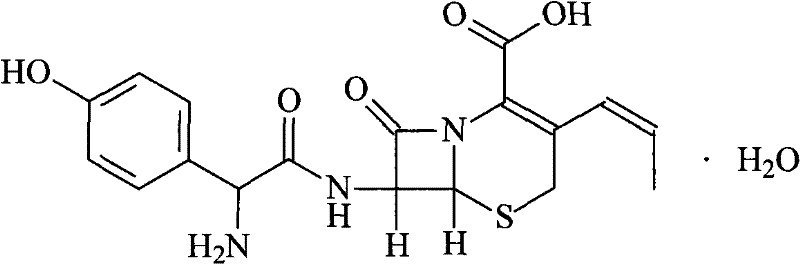
The global demand for second-generation cephalosporins continues to rise, driven by the persistent need for effective broad-spectrum antibiotics capable of combating resistant bacterial strains. At the forefront of this therapeutic class is Cefprozil, a potent agent known for its exceptional activity against both Gram-positive and Gram-negative bacteria. However, the commercial viability of this critical Active Pharmaceutical Ingredient (API) has historically been constrained by the limitations of traditional synthetic routes, which often yield crude products laden with impurities that are difficult to remove. A groundbreaking approach detailed in patent CN101880289A offers a transformative solution to these challenges, introducing a sophisticated purification methodology that leverages macroporous polymeric adsorption technology. This innovation shifts the paradigm from complex synthesis optimization to efficient downstream processing, enabling manufacturers to achieve pharmaceutical-grade purity levels exceeding 99.5% with remarkable recovery rates. By integrating this advanced purification protocol, producers can secure a consistent supply of high-quality Cefprozil, ensuring compliance with stringent international pharmacopoeia standards while optimizing overall production economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Cefprozil has relied heavily on semi-synthetic routes utilizing key intermediates such as GCLE (7-amino-3-chloro-3-cephem-4-carboxylic acid p-methoxybenzyl ester) or GCLH. While these pathways provide the necessary carbon skeleton for the cephalosporin nucleus, they are fraught with significant technical and economic inefficiencies that hinder large-scale optimization. The primary bottleneck lies in the purification stage; conventional recrystallization techniques often fail to adequately remove structurally similar impurities and polymeric by-products generated during the coupling reactions. Consequently, the final crude product frequently exhibits suboptimal purity profiles, necessitating multiple, yield-depleting purification cycles that drive up manufacturing costs. Furthermore, the reliance on expensive starting materials like GCLE, combined with the use of hazardous solvents and complex reaction conditions, results in a process with a substantial environmental footprint and limited scalability. These factors collectively create a fragile supply chain where minor deviations in raw material quality can lead to batch failures, jeopardizing the continuity of supply for downstream formulation manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the novel purification technique disclosed in the patent data introduces a robust, resin-based separation strategy that fundamentally redefines the efficiency of Cefprozil processing. This approach bypasses the need for aggressive chemical modifications or exhaustive solvent exchanges by utilizing the selective adsorption properties of macroporous resins such as Hz816 or Amberlite XAD-4. The process begins by converting the crude Cefprozil into a soluble salt form through precise pH adjustment using alkaline solutions, ensuring complete dissolution and accessibility of the active molecules to the adsorbent matrix. Once loaded onto the resin column, the Cefprozil is selectively retained while non-polar impurities and reaction by-products are washed away, effectively decoupling the purification efficiency from the variability of the upstream synthesis. Subsequent elution with optimized solvent systems, such as ethanol-water mixtures, allows for the recovery of the antibiotic in a highly concentrated and purified state. This streamlined workflow not only simplifies the operational complexity but also dramatically enhances the overall yield, with experimental data demonstrating recovery rates consistently above 90%, thereby setting a new benchmark for cost-effective antibiotic manufacturing.
Mechanistic Insights into Macroporous Resin Adsorption and pH-Controlled Crystallization
The efficacy of this purification system is rooted in the intricate interplay between the physicochemical properties of the Cefprozil molecule and the surface characteristics of the macroporous adsorbent. Cefprozil, characterized by its zwitterionic nature and multiple hydrogen-bonding sites, interacts strongly with the hydrophobic polystyrene backbone of the resin through van der Waals forces and pi-pi stacking interactions when in its salt form. The initial dissolution step, where the pH is adjusted to a range of 7 to 10 using bases like sodium hydroxide or potassium hydroxide, is critical as it ionizes the carboxylic acid group, increasing the molecule's polarity and solubility in the aqueous phase while maintaining its affinity for the resin's porous network. As the solution percolates through the column, the large surface area of the macroporous resin facilitates rapid mass transfer, allowing the Cefprozil ions to penetrate the pores and bind securely, effectively filtering out larger molecular weight impurities and colored bodies that do not fit within the resin's pore structure or lack the necessary affinity. This selective retention mechanism acts as a molecular sieve, ensuring that only the target compound is carried forward into the elution phase, thereby laying the foundation for the ultra-high purity observed in the final product.
Following adsorption, the controlled release of the active ingredient is achieved through a carefully calibrated elution and crystallization sequence that preserves the integrity of the sensitive beta-lactam ring. The use of low-carbon alcohol-water mixtures, particularly ethanol-water at a 1:1 volume ratio, serves to disrupt the hydrophobic interactions between the drug and the resin, facilitating efficient desorption without degrading the molecule. The subsequent addition of activated carbon plays a pivotal role in removing trace organic impurities and endotoxins, further polishing the solution before the final precipitation step. By slowly introducing an acidic solution to lower the pH to between 2 and 5, the solubility of the Cefprozil is drastically reduced, inducing nucleation and crystal growth under thermodynamically favorable conditions. This pH-swing crystallization ensures the formation of uniform, high-density crystals that are easy to filter and wash, minimizing solvent retention and maximizing the drying efficiency. The result is a chemically stable, high-purity monohydrate form of Cefprozil that meets the rigorous specifications required for parenteral and oral dosage forms.
How to Synthesize Cefprozil Efficiently
The implementation of this purification protocol requires a systematic approach to unit operations, focusing on precise control of pH, flow rates, and solvent compositions to maximize throughput and product quality. The process is designed to be adaptable to various scales of production, from pilot plant trials to multi-ton commercial batches, making it an ideal candidate for technology transfer and process intensification initiatives. Operators must ensure that the crude feedstock is fully solubilized prior to loading to prevent column channeling, and that the elution profile is monitored closely to collect only the high-purity fractions. For a comprehensive understanding of the specific operational parameters, including residence times, column dimensions, and washing volumes, please refer to the detailed technical guidelines provided below.
- Dissolve crude Cefprozil in an alkaline solution or organic solvent with base to adjust pH to 7-10, forming a clear saline solution.
- Pass the solution through a column packed with macroporous polymeric adsorbent (e.g., Hz816 or Amberlite XAD-4) to capture the active compound.
- Elute the captured Cefprozil salt with a solvent-water mixture, treat with activated carbon, acidify to pH 2-5 to precipitate pure crystals, and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resin-based purification technology represents a strategic opportunity to mitigate risk and enhance the financial performance of antibiotic production lines. By shifting the burden of quality control from the synthesis stage to a highly efficient purification step, manufacturers can utilize lower-grade crude intermediates without compromising the final API specification, thereby unlocking significant cost savings in raw material sourcing. The simplicity of the unit operations involved—primarily dissolution, adsorption, and crystallization—reduces the dependency on specialized equipment and highly skilled labor, leading to lower operational expenditures and a more resilient manufacturing infrastructure. Furthermore, the high recovery yields associated with this method mean that less raw material is wasted, directly contributing to a more sustainable and economically viable production model that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic separations and the reduction in solvent consumption inherent in this resin-based process lead to a substantial decrease in overall production costs. By utilizing widely available and regenerable macroporous resins, facilities can avoid the recurring expense of disposable silica gels or expensive preparative HPLC columns, resulting in a leaner cost structure. Additionally, the high yield efficiency ensures that the maximum amount of valuable product is recovered from each batch, minimizing the cost per kilogram of the final API and improving profit margins in a competitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of this purification method significantly enhances supply chain stability by reducing the sensitivity of the process to variations in crude feedstock quality. Since the resin effectively scrubs impurities, suppliers are less vulnerable to disruptions caused by off-spec intermediate batches, ensuring a continuous and reliable flow of finished goods to customers. The use of common industrial solvents and standard reactor equipment further simplifies logistics and inventory management, allowing for faster turnaround times and the ability to respond swiftly to fluctuations in market demand without the need for extensive requalification of processes.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this technology offers distinct advantages by minimizing the generation of hazardous waste and reducing the overall solvent load discharged from the facility. The closed-loop nature of the adsorption-elution cycle allows for the recovery and recycling of solvents, aligning with increasingly strict environmental regulations and corporate sustainability goals. Moreover, the straightforward scale-up characteristics of column chromatography and crystallization mean that production capacity can be expanded linearly with minimal engineering hurdles, supporting long-term growth strategies and the ability to secure large-volume contracts with confidence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Cefprozil purification technology. These insights are derived directly from the patent specifications and practical application data, providing a clear framework for evaluating the feasibility of adopting this method within existing manufacturing frameworks. Understanding these details is crucial for technical teams assessing process compatibility and for commercial teams negotiating supply agreements based on quality and cost metrics.
Q: What is the primary advantage of using macroporous resin for Cefprozil purification?
A: The primary advantage is the ability to achieve exceptionally high purity (up to 99.9%) and high recovery yields (over 90%) while eliminating complex chromatographic steps, significantly reducing production costs and environmental waste compared to traditional GCLE routes.
Q: Which types of macroporous resins are most effective for this process?
A: Polystyrene-based adsorption resins such as Hz816 and Amberlite XAD-4 have demonstrated superior performance in adsorbing Cefprozil salts, offering optimal binding capacity and ease of elution with alcohol-water mixtures.
Q: How does this purification method impact the stability of the beta-lactam ring?
A: By carefully controlling the pH during dissolution (pH 7-10) and precipitation (pH 2-5) and avoiding harsh thermal conditions, the process minimizes hydrolysis of the sensitive beta-lactam structure, ensuring the biological activity of the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefprozil Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature are faithfully reproduced in our state-of-the-art manufacturing facilities. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of Cefprozil meets or exceeds global pharmacopoeial standards, providing our partners with the assurance they need to move forward with their drug development programs.
We invite you to collaborate with us to leverage this cutting-edge purification technology for your specific supply chain needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, as well as obtain specific COA data and route feasibility assessments that demonstrate the tangible benefits of our manufacturing capabilities. Let us help you secure a stable, high-quality supply of Cefprozil that drives your business forward.
