Advanced Manufacturing of Cefprozil via Novel Ester Intermediates for Global Supply Chains
Introduction to Patent CN101798312B: A Paradigm Shift in Cephalosporin Manufacturing
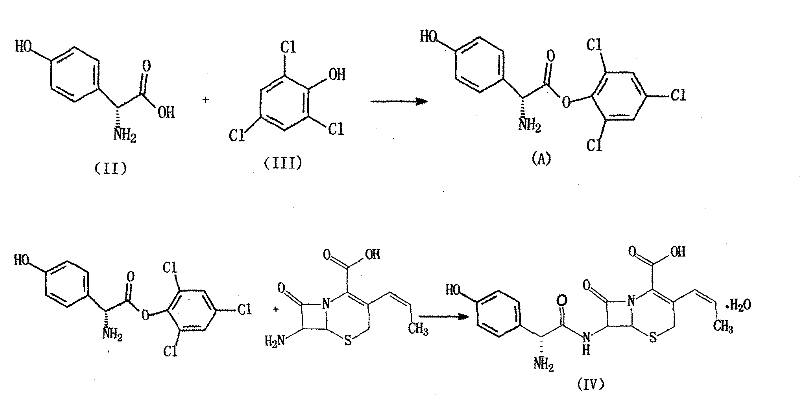
The pharmaceutical landscape for second-generation cephalosporins is constantly evolving, driven by the need for higher purity and more sustainable manufacturing processes. Patent CN101798312B introduces a groundbreaking variation route for the preparation of the Cefprozil compound, addressing critical bottlenecks found in legacy synthetic pathways. This technology fundamentally re-engineers the construction of the Cefprozil molecule by introducing a novel activated ester intermediate derived from D-pHPG and 2,4,6-trichlorophenol. Unlike conventional methods that struggle with complex reaction steps and low yields, this invention streamlines the transformation of 7-phenacetylamino-3-chloromethyl cephalosporanic acid anisyl acetate (GCLE) into the final active pharmaceutical ingredient. The strategic elimination of activating reagents such as sodium iodide not only simplifies the post-treatment workflow but also drastically reduces the generation of hazardous waste streams. For global supply chain leaders, this represents a significant opportunity to secure a more reliable source of high-purity antibiotic intermediates while mitigating the environmental risks associated with traditional heavy metal or halide-based catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cefprozil has been plagued by inefficiencies that drive up costs and compromise product quality. Prior art, such as USP NO.4694079, discloses methods yielding DMF solvates with yields as low as 65%, necessitating extensive recycling and purification efforts that erode profit margins. Furthermore, patents like USP NO.5869648 and NO.6136967 rely heavily on adsorption chromatography to separate isomers, a technique that is notoriously difficult to implement on a multi-ton scale due to solvent consumption and column capacity limitations. Chinese patent CN1694888A further highlights the industry's struggle with high pollution levels and significant isomer content, which directly impacts the safety profile and regulatory approval potential of the final drug substance. These conventional routes often involve harsh activation conditions that lead to the degradation of the sensitive beta-lactam ring, resulting in a broad impurity profile that requires costly downstream processing to meet stringent pharmacopoeial standards.
The Novel Approach
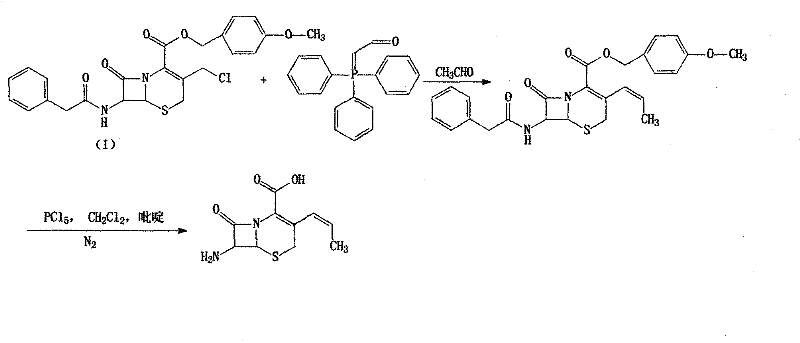
In stark contrast, the methodology outlined in CN101798312B offers a robust solution by decoupling the side-chain activation from the final coupling step. The core innovation lies in the pre-formation of a stable D-pHPG 2,4,6-trichlorophenyl ester intermediate. This activated ester serves as a superior acylating agent that reacts selectively with the 7-amino group of the cephalosporin nucleus under mild conditions. By shifting the activation burden to the side chain precursor, the process avoids the harsh conditions typically required to activate the amino acid directly in the presence of the fragile beta-lactam core. Additionally, the initial modification of the GCLE starting material utilizes a modified Wittig reaction that proceeds efficiently at 60°C without the need for sodium iodide promoters. This holistic redesign of the synthetic tree results in a process that is not only chemically elegant but also operationally simpler, facilitating easier production amplification and consistent batch-to-batch reproducibility.
Mechanistic Insights into Activated Ester Coupling and Wittig Olefination
The chemical elegance of this route is best understood through the lens of reaction kinetics and thermodynamic stability. The first critical transformation involves the conversion of the 3-chloromethyl group on the GCLE scaffold into a propenyl moiety. Traditionally, this requires the conversion of the chloride to a more reactive iodide using sodium iodide in acetone, followed by reaction with triphenylphosphine. However, this patent demonstrates that direct reaction with triphenylphosphine in a biphasic system of chloroform and water at elevated temperatures (60°C) generates the phosphonium salt in situ. Subsequent treatment with base and acetaldehyde effects the Wittig olefination with high Z-selectivity. This mechanistic bypass eliminates the stoichiometric waste associated with halide exchange and removes a unit operation from the process flow, thereby reducing the overall cycle time and solvent inventory.
Furthermore, the coupling mechanism utilizing the D-pHPG 2,4,6-trichlorophenyl ester is pivotal for controlling the stereochemical integrity of the final product. The electron-withdrawing nature of the three chlorine atoms on the phenol ring significantly enhances the electrophilicity of the carbonyl carbon in the ester intermediate. This heightened reactivity allows the nucleophilic attack by the 7-amino group of the cephalosporin nucleus to proceed rapidly at lower temperatures (5-10°C), minimizing the risk of epimerization at the chiral alpha-carbon of the side chain. The use of triethylamine as a proton scavenger ensures that the reaction medium remains sufficiently basic to facilitate amine nucleophilicity without inducing beta-lactam ring opening. The result is a reaction profile that favors the formation of the desired trans-isomer while suppressing the formation of cis-isomers and other degradation products, leading to a crude product with exceptionally high purity that requires minimal recrystallization.
How to Synthesize Cefprozil Efficiently
The implementation of this novel synthetic route requires precise control over reaction parameters to maximize the benefits of the activated ester chemistry. The process is divided into three distinct operational phases: the olefination of the cephalosporin nucleus, the deprotection of the 7-amino group, and the final acylation with the novel side-chain ester. Each step has been optimized to balance reaction rate with product stability, ensuring that the sensitive beta-lactam structure is preserved throughout the synthesis. Operators must pay particular attention to temperature gradients during the exothermic deprotection step and pH control during the final isolation to ensure optimal crystal formation. The detailed standardized operating procedures for executing this high-efficiency synthesis are provided in the technical guide below.
- Perform Wittig reaction on GCLE with triphenylphosphine and acetaldehyde to form the propenyl side chain without sodium iodide activation.
- Execute deprotection using phosphorus pentachloride and phenol to generate the 7-amino cephalosporanic acid core (APRA).
- Couple APRA with the pre-formed D-pHPG 2,4,6-trichlorophenyl ester intermediate under mild pH control to yield high-purity Cefprozil.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic advantages beyond mere chemical yield. The primary value driver is the substantial simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced supply security. By eliminating the need for chromatographic purification and replacing it with crystallization-based isolation, the process becomes inherently more scalable and less prone to equipment bottlenecks. This shift allows manufacturers to utilize standard stainless steel reactors rather than specialized chromatography columns, significantly lowering the barrier to entry for contract manufacturing organizations and increasing the pool of potential suppliers for this critical antibiotic intermediate.
- Cost Reduction in Manufacturing: The economic impact of this route is profound, primarily driven by the removal of expensive and hazardous reagents. The elimination of sodium iodide, a costly activator in traditional Wittig reactions, removes a significant line item from the bill of materials. Furthermore, the avoidance of chromatography reduces solvent consumption by orders of magnitude, lowering both raw material costs and waste disposal fees. The higher yields reported in the patent examples, exceeding 90% in key steps compared to the 65% baseline of older methods, mean that less starting material is required to produce the same amount of final product, effectively stretching the purchasing power of every kilogram of GCLE acquired.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthetic routes; the simpler the process, the fewer the points of failure. This novel route relies on readily available commodity chemicals such as triphenylphosphine, acetaldehyde, and 2,4,6-trichlorophenol, which are produced globally in massive volumes. By decoupling the side-chain synthesis from the core coupling, manufacturers can stockpile the stable trichlorophenyl ester intermediate, creating a buffer against supply shocks for the more sensitive cephalosporin nucleus. This modularity ensures that production schedules can be maintained even if there are temporary disruptions in the supply of one specific precursor, providing a robust safety net for long-term supply agreements.
- Scalability and Environmental Compliance: As regulatory pressure regarding pharmaceutical waste intensifies, the environmental profile of a synthesis route becomes a key qualification criterion. This process generates significantly less hazardous waste by avoiding heavy metal catalysts and minimizing halogenated solvent usage through efficient recycling protocols inherent in the crystallization steps. The simplified workup procedures, which rely on pH adjustment and filtration rather than complex extractions or column chromatography, reduce the volume of effluent requiring treatment. This 'green chemistry' advantage not only lowers compliance costs but also future-proofs the supply chain against tightening environmental regulations, ensuring uninterrupted production capabilities in jurisdictions with strict emission standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cefprozil synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning tech transfer and for procurement teams negotiating supply contracts based on process efficiency.
Q: How does the novel trichlorophenyl ester intermediate improve Cefprozil purity?
A: The use of D-pHPG 2,4,6-trichlorophenyl ester creates a highly reactive yet stable acylating agent. This specificity minimizes side reactions and racemization during the coupling step, significantly reducing isomer content compared to traditional activation methods.
Q: What are the cost advantages of eliminating sodium iodide in the Wittig step?
A: Traditional routes often require sodium iodide to activate the chloromethyl group, adding reagent costs and complicating waste treatment. This patented route utilizes direct reaction with triphenylphosphine at elevated temperatures, removing the need for expensive halide exchange catalysts and simplifying downstream purification.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability. It replaces difficult chromatographic separations with straightforward crystallization and filtration steps, achieving yields over 90% in key stages, which is critical for cost-effective metric-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefprozil Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to monitor every critical quality attribute, from isomer ratios to residual solvent levels. Our commitment to excellence ensures that every batch of Cefprozil intermediate we deliver meets the exacting standards required by global regulatory bodies.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and security for your antibiotic production programs.
