Scalable Green Electrosynthesis of 2-Acyloxy Aniline Derivatives for Commercial API Production
Scalable Green Electrosynthesis of 2-Acyloxy Aniline Derivatives for Commercial API Production
The pharmaceutical industry is constantly seeking sustainable and cost-effective pathways for synthesizing complex intermediates, particularly those serving as backbones for kinase inhibitors and antiviral agents. A significant breakthrough in this domain is documented in patent CN111394747B, which discloses a novel green electrochemical synthesis method for 2-acyloxy aniline derivatives. This technology represents a paradigm shift from traditional stoichiometric oxidation to catalytic electrosynthesis, addressing critical pain points regarding waste generation and reagent costs. By leveraging electricity as a clean oxidant, this process eliminates the need for hazardous chemical oxidants and expensive noble metal additives often required in conventional C-H activation protocols. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition: a streamlined, environmentally benign route to high-value scaffolds used in drugs like Hitinib and Rilpivirine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-acyloxyaniline derivatives has relied heavily on classical acylation reactions using o-hydroxyphenyl amine compounds, a route severely constrained by the limited availability and high cost of diverse substituted o-hydroxyanilines. In recent decades, transition metal-catalyzed directed C-H activation emerged as a powerful alternative, yet early iterations of this technology were fraught with inefficiencies. For instance, seminal work by Padala et al. utilized ruthenium catalysis but necessitated the addition of silver hexafluoroantimonate (AgSbF6) as an additive and ammonium persulfate ((NH4)2S2O8) as a stoichiometric oxidant. Similarly, Sarkar et al. reported methods requiring silver carbonate (Ag2CO3) alongside ruthenium catalysts. These conventional approaches suffer from substantial drawbacks: the reliance on stoichiometric amounts of expensive silver salts drastically inflates raw material costs, while the use of strong chemical oxidants generates significant amounts of inorganic waste, complicating downstream purification and environmental compliance. Furthermore, these reactions often demand harsh conditions, such as temperatures reaching 100°C and extended reaction times up to 24 hours, which negatively impacts energy consumption and throughput.
The Novel Approach
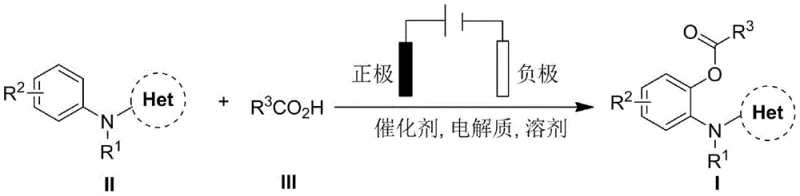
The electrochemical methodology described in patent CN111394747B effectively circumvents these legacy issues by replacing chemical oxidants with electrons. In this innovative system, aniline compounds and carboxylic acids undergo oxidative coupling in an undivided electrolytic cell under the influence of a constant current. The process operates under significantly milder conditions, typically at temperatures between 25°C and 75°C, and completes within just 4 hours, representing a six-fold improvement in time efficiency compared to prior art. Crucially, the only byproduct of this redox neutral transformation is hydrogen gas, rendering the process inherently greener and safer. By removing the requirement for external oxidants and silver additives, the method simplifies the reaction mixture, reduces the burden on waste treatment facilities, and lowers the overall cost of goods sold (COGS). This approach aligns perfectly with modern green chemistry principles, offering a robust platform for the sustainable manufacturing of complex pharmaceutical intermediates.
Mechanistic Insights into Electrochemical C-H Activation
The core of this technology lies in the anodic oxidation mechanism facilitated by a ruthenium or rhodium catalyst, such as [Ru(p-cymene)Cl2]2. In the proposed catalytic cycle, the metal center coordinates with the nitrogen-containing heterocyclic directing group (Het) on the aniline substrate, forming a stable metallacycle intermediate. Upon application of anodic potential, the metal center undergoes oxidation, increasing its electrophilicity and facilitating the activation of the ortho-C-H bond. Simultaneously, the carboxylic acid substrate interacts with the activated metal complex, leading to the formation of a metal-acyl species. The subsequent reductive elimination step releases the desired 2-acyloxy aniline product and regenerates the active catalyst species at the electrode surface. This electrochemical regeneration of the high-valent metal oxidant is the key differentiator, as it bypasses the need for chemical oxidants to turn over the catalytic cycle. The use of a reticulated vitreous carbon (RVC) anode provides a high surface area for efficient electron transfer, ensuring consistent reaction kinetics throughout the batch.
From an impurity control perspective, this mechanism offers distinct advantages over traditional oxidative couplings. The absence of aggressive chemical oxidants like persulfates minimizes the risk of over-oxidation side reactions, which often lead to complex impurity profiles that are difficult to separate. The selectivity is driven primarily by the chelation of the directing group, ensuring that functionalization occurs exclusively at the ortho-position relative to the amine nitrogen. Furthermore, the mild reaction conditions prevent thermal degradation of sensitive functional groups on the aromatic rings. The use of tetrabutylammonium tetrafluoroborate (nBu4NBF4) as the supporting electrolyte ensures high conductivity without introducing nucleophilic species that could interfere with the acylation step. This high degree of chemoselectivity translates directly to higher crude purity, reducing the load on downstream purification processes such as column chromatography or recrystallization, which is a critical factor for commercial viability.
How to Synthesize N-pyrimidinyl-2-benzoyloxyaniline Efficiently
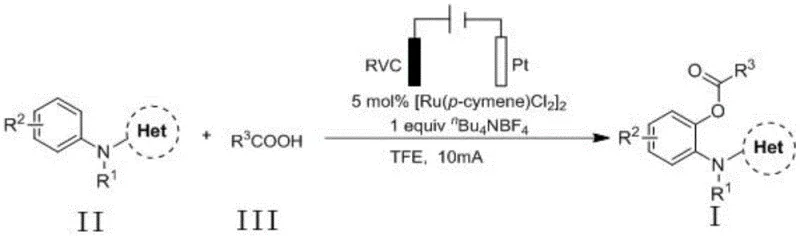
To implement this synthesis in a laboratory or pilot setting, strict adherence to the optimized electrochemical parameters is essential for maximizing yield and reproducibility. The patent outlines a standardized protocol where the aniline substrate and carboxylic acid are dissolved in trifluoroethanol (TFE), a solvent chosen for its ability to stabilize charged intermediates and solubilize the electrolyte. The reaction mixture is subjected to constant current electrolysis using a specific electrode configuration that balances conductivity and stability. Detailed below is the procedural framework derived from the patent examples, which serves as a baseline for process optimization.
- Dissolve the aniline substrate, carboxylic acid coupling partner, Ruthenium catalyst (e.g., [Ru(p-cymene)Cl2]2), and electrolyte (e.g., nBu4NBF4) in trifluoroethanol solvent within an undivided electrolytic cell.
- Insert a reticulated vitreous carbon (RVC) anode and a platinum cathode into the reaction mixture, ensuring proper electrode spacing and connection to a constant current power supply.
- Apply a constant current of 10 mA at 75°C under nitrogen protection for 4 hours, then purify the resulting 2-acyloxy aniline derivative via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this electrochemical methodology offers tangible strategic benefits beyond mere technical novelty. The primary driver for adoption is the significant reduction in raw material costs associated with the elimination of stoichiometric oxidants and silver additives. In traditional C-H activation processes, silver salts can account for a disproportionate share of the material cost; removing them entirely creates immediate margin expansion. Additionally, the simplification of the workup procedure—due to the absence of inorganic oxidant residues—reduces solvent consumption and processing time, further driving down operational expenditures. This cost structure makes the final API intermediate more competitive in a price-sensitive global market.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior because it substitutes expensive chemical reagents with electricity, a utility that is generally cheaper and more stable in price than specialty chemicals. By eliminating the need for silver hexafluoroantimonate and ammonium persulfate, the direct material cost per kilogram of product is drastically lowered. Moreover, the catalyst loading is low (1 to 20 mol%), and the potential for catalyst recovery in flow electrochemical cells suggests even greater long-term savings. The reduction in waste disposal costs is another critical factor; handling and treating heavy metal waste from silver salts is expensive and regulated, whereas the only byproduct here is hydrogen gas, which can be safely vented or captured.
- Enhanced Supply Chain Reliability: Relying on stoichiometric oxidants introduces supply chain volatility, as these reagents are often subject to market fluctuations and regulatory scrutiny regarding their transport and storage. In contrast, the inputs for this electrochemical method—anilines, carboxylic acids, and electricity—are commodity-grade materials with robust, multi-source supply chains. This diversification of input risk ensures greater continuity of supply for downstream API manufacturers. The operational simplicity of the undivided cell setup also means that production is less susceptible to disruptions caused by the shortage of specialized reactor hardware, as standard electrochemical equipment is widely available from multiple vendors globally.
- Scalability and Environmental Compliance: Scaling electrochemical reactions has historically been a challenge, but the use of undivided cells with robust electrodes like RVC and platinum simplifies engineering requirements for commercial scale-up. The process operates at ambient pressure and moderate temperatures, removing the need for high-pressure autoclaves or cryogenic cooling systems, which lowers capital expenditure (CAPEX) for new production lines. From an environmental standpoint, the E-factor (mass of waste per mass of product) is significantly improved due to the atom economy of using electrons as reagents. This aligns with increasingly stringent environmental regulations in major pharmaceutical manufacturing hubs, reducing the risk of production shutdowns due to compliance issues and enhancing the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These answers are derived directly from the experimental data and claims within patent CN111394747B, providing a factual basis for evaluating the technology's fit for your specific production needs. Understanding these nuances is vital for R&D teams planning tech transfer and for procurement teams assessing vendor capabilities.
Q: What are the primary advantages of this electrochemical method over traditional transition metal catalysis?
A: Unlike traditional methods requiring stoichiometric oxidants like ammonium persulfate or silver salts which generate significant waste, this electrochemical approach uses electricity as the terminal oxidant, producing only hydrogen gas as a byproduct and eliminating the need for expensive silver additives.
Q: Can this synthesis protocol be scaled for industrial manufacturing of API intermediates?
A: Yes, the process utilizes an undivided cell setup with commercially available electrodes (RVC/Pt) and operates at moderate temperatures (25-75°C), making it highly amenable to scale-up without the safety hazards associated with large quantities of chemical oxidants.
Q: What is the substrate scope for the carboxylic acid component in this reaction?
A: The method demonstrates broad compatibility with various carboxylic acids, including benzoic acid derivatives with electron-donating or withdrawing groups, heteroaromatic acids like thiophene-2-carboxylic acid, and aliphatic acids like acetic acid, yielding products with good to excellent efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acyloxy Aniline Derivatives Supplier
The electrochemical synthesis of 2-acyloxy aniline derivatives represents a cutting-edge solution for the efficient production of high-value pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this technology and have integrated advanced electrochemical capabilities into our CDMO infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle the unique analytical requirements of electrochemically synthesized compounds, guaranteeing that every batch meets the highest international standards.
We invite you to collaborate with us to leverage this green chemistry innovation for your pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us today to request specific COA data for our catalog of 2-acyloxy aniline derivatives or to discuss route feasibility assessments for your custom synthesis projects. Let us help you build a more sustainable and cost-effective supply chain.
