Revolutionizing Pharmaceutical Intermediate Production via Green Electrochemical C-H Activation
Revolutionizing Pharmaceutical Intermediate Production via Green Electrochemical C-H Activation
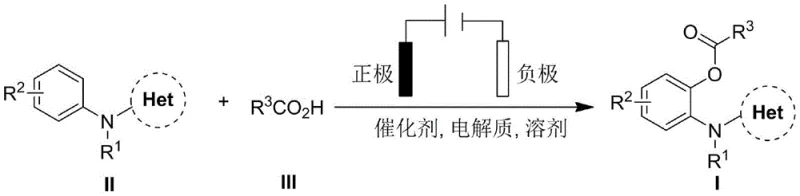
The landscape of pharmaceutical intermediate manufacturing is undergoing a paradigm shift towards sustainable and atom-economical processes, driven by the urgent need to reduce environmental footprints and operational costs. Patent CN111394747A introduces a groundbreaking green electrochemical synthesis method for 2-acyloxy aniline derivatives, a critical structural motif found in numerous high-value bioactive compounds such as Osimertinib and Rilpivirine. This technology leverages the power of anodic oxidation to drive C-H functionalization, effectively replacing hazardous chemical oxidants with clean electrical energy. By utilizing an undivided electrolytic cell setup, the process achieves high chemical selectivity while generating hydrogen gas as the sole byproduct, marking a significant departure from traditional waste-intensive synthetic routes. For R&D directors and procurement specialists, this innovation represents a tangible opportunity to streamline supply chains and enhance the sustainability profile of complex API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-acyloxy aniline derivatives has relied heavily on the acylation of o-hydroxyaniline precursors, a pathway severely constrained by the limited availability and high cost of substituted o-hydroxyanilines. Alternatively, transition metal-catalyzed C-H activation strategies have emerged as powerful tools; however, existing protocols often suffer from significant drawbacks that hinder their industrial adoption. Conventional ruthenium-catalyzed methods typically require stoichiometric amounts of expensive silver additives, such as AgSbF6, and strong chemical oxidants like ammonium persulfate or silver carbonate to regenerate the active catalytic species. These reagents not only escalate raw material costs but also generate substantial quantities of heavy metal waste and acidic byproducts, complicating downstream purification and wastewater treatment. Furthermore, these reactions frequently necessitate elevated temperatures exceeding 100°C and prolonged reaction times up to 24 hours, leading to higher energy consumption and potential thermal degradation of sensitive functional groups.
The Novel Approach
In stark contrast, the electrochemical methodology disclosed in the patent offers a streamlined and environmentally benign alternative that directly addresses the inefficiencies of prior art. By employing electricity as the terminal oxidant, this novel approach completely eliminates the need for external chemical oxidants and noble metal additives, thereby drastically simplifying the reaction mixture and reducing the E-factor of the process. The system operates under milder conditions, typically around 75°C, and achieves completion within just 4 hours, demonstrating superior reaction kinetics and energy efficiency. The use of a simple undivided cell configuration with readily available electrode materials, such as reticulated vitreous carbon and platinum, ensures that the process is not only chemically elegant but also practically feasible for scale-up. This shift from chemical to electrochemical oxidation fundamentally alters the economic and environmental calculus of producing these valuable pharmaceutical building blocks.
Mechanistic Insights into Ru-Catalyzed Electrochemical Oxidative Coupling
The core of this transformative technology lies in the synergistic interplay between ruthenium catalysis and anodic oxidation within an electrochemical cell. The mechanism initiates with the coordination of the ruthenium catalyst to the nitrogen-containing directing group on the aniline substrate, forming a stable cyclometalated intermediate that directs the activation of the ortho-C-H bond. Unlike traditional thermal cycles that rely on chemical oxidants to re-oxidize the reduced metal center, this system utilizes the anode to facilitate the electron transfer required for catalyst regeneration. The carboxylic acid substrate undergoes deprotonation in the presence of the electrolyte, and the resulting carboxylate species participates in the coupling event at the activated ortho-position. This electrochemically driven cycle ensures a continuous turnover of the catalyst without the accumulation of reduced metal species or oxidant-derived waste, maintaining high catalytic efficiency throughout the reaction duration.
From an impurity control perspective, the high selectivity of this electrochemical protocol is attributed to the precise control over the oxidation potential applied to the system. By tuning the constant current, the reaction avoids over-oxidation of the substrate or the product, which is a common issue when using strong chemical oxidants like persulfates. The absence of aggressive oxidizing agents minimizes side reactions such as polymerization or decomposition of sensitive heterocyclic moieties, resulting in a cleaner crude reaction profile. Additionally, the generation of hydrogen gas at the cathode serves as a driving force that helps shift the equilibrium forward without introducing gaseous pollutants. This inherent cleanliness reduces the burden on purification steps, allowing for simpler workup procedures like direct column chromatography, which is crucial for maintaining high throughput in a manufacturing environment.
How to Synthesize 2-Acyloxy Aniline Derivatives Efficiently
Implementing this electrochemical synthesis route requires careful attention to reactor configuration and parameter optimization to ensure reproducibility and safety. The standard protocol involves dissolving the aniline derivative, carboxylic acid, ruthenium catalyst, and supporting electrolyte in a fluorinated alcohol solvent, which provides the necessary conductivity and solubility for the ionic species involved. The reaction is conducted in an undivided cell under an inert atmosphere to prevent interference from oxygen, with specific electrode materials chosen to maximize surface area and stability. While the patent details specific batch conditions, the underlying principles are highly adaptable to continuous flow systems, which are increasingly preferred for commercial electrochemical synthesis due to their superior mass transfer and heat management capabilities.
- Dissolve the aniline substrate, carboxylic acid coupling partner, ruthenium catalyst, and electrolyte in trifluoroethanol solvent within an undivided electrolytic cell.
- Insert a reticulated vitreous carbon (RVC) anode and a platinum cathode into the reaction mixture under a nitrogen atmosphere.
- Apply a constant current of 10mA at 75°C for 4 hours, then isolate the product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates into immediate and long-term strategic benefits that extend beyond mere technical novelty. The elimination of stoichiometric silver salts and hazardous oxidants significantly lowers the bill of materials, removing some of the most volatile and expensive components from the supply chain. Furthermore, the simplified waste profile reduces the costs associated with hazardous waste disposal and environmental compliance, which are becoming increasingly stringent regulatory burdens for chemical manufacturers. The robustness of the method across a wide range of substrates ensures supply continuity, as it allows for the flexible sourcing of diverse carboxylic acid and aniline starting materials without needing to re-optimize the entire process for each new derivative.
- Cost Reduction in Manufacturing: The removal of expensive silver additives and chemical oxidants leads to a substantial decrease in raw material costs, while the milder reaction conditions reduce energy expenditure associated with heating and cooling. The simplified workup procedure minimizes solvent usage and labor time required for purification, contributing to an overall leaner manufacturing process that enhances profit margins without compromising quality.
- Enhanced Supply Chain Reliability: By relying on electricity rather than specialized chemical reagents that may face supply shortages, the process mitigates risks associated with raw material availability. The use of common electrode materials and standard solvents ensures that the supply chain remains resilient against market fluctuations, providing a stable foundation for long-term production planning and contract fulfillment.
- Scalability and Environmental Compliance: The undivided cell design is inherently scalable and well-suited for adaptation into continuous flow reactors, facilitating the transition from laboratory benchtop to multi-ton commercial production. The generation of hydrogen as the only byproduct aligns perfectly with green chemistry principles, easing regulatory approval processes and enhancing the corporate sustainability profile for partners seeking eco-friendly suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, aiming to clarify the practical implications for industrial application. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of this electrochemical method over traditional C-H activation?
A: Unlike traditional methods requiring stoichiometric silver oxidants and harsh conditions, this electrochemical approach uses electricity as the terminal oxidant, generating only hydrogen gas as a byproduct and eliminating heavy metal waste.
Q: Is this process scalable for commercial API intermediate production?
A: Yes, the use of undivided cells and standard electrode materials like RVC and platinum suggests high scalability, particularly when adapted to continuous flow electrochemical reactors for large-scale manufacturing.
Q: What types of substrates are compatible with this synthesis route?
A: The method demonstrates broad substrate scope, tolerating various substituted anilines with pyrimidine or pyridine directing groups and diverse carboxylic acids, including aromatic, heteroaromatic, and aliphatic acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acyloxy Aniline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-acyloxy aniline derivatives delivered meets the highest standards required for API synthesis. We are committed to leveraging advanced technologies like the one described in CN111394747A to provide our clients with superior quality and reliability.
We invite you to collaborate with us to explore how this green electrochemical route can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your target molecules. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can support your supply chain goals with efficient, sustainable, and cost-effective manufacturing solutions.
