Advanced Synthesis of Cefmetazole Sodium for High-Purity Pharmaceutical Applications
The pharmaceutical industry constantly seeks robust synthetic routes for critical antibiotics that balance high purity with economic viability. Patent CN102718780A introduces a refined preparation method for Cefmetazole Sodium, a potent second-generation cephalosporin known for its stability against beta-lactamases produced by Gram-negative bacilli. This molecule, chemically defined as (6R, 7S)-7-[[2-(cyanomethylthio)acetyl]amino]-7-methoxy-3-[(1-methyltetrazol-5-yl)thiomethyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid sodium salt, is essential for treating respiratory, urinary, and biliary tract infections. The structural complexity of this compound demands precise control over stereochemistry and functional group integrity during synthesis.
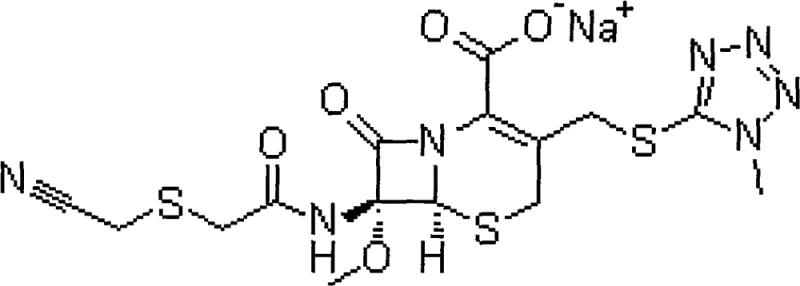
As a reliable cefmetazole sodium supplier, understanding the nuances of its synthesis is paramount. The patented process addresses historical challenges in cephalosporin manufacturing, specifically targeting the issues of low yield, poor color, and difficult purification that have plagued earlier methodologies. By integrating a unique amine salt intermediate strategy and ion exchange technology, this route offers a compelling solution for manufacturers aiming to optimize their production lines for high-purity cefmetazole sodium while maintaining strict regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art technologies for synthesizing Cefmetazole Sodium have faced significant hurdles that impact both product quality and operational efficiency. For instance, earlier patents such as CN200910014972.5 relied on the use of highly basic sodium hydroxide for neutralization. This aggressive chemical environment frequently resulted in the formation of excessive impurities, leading to a final product with lower content specifications and a darker, less desirable color profile. Furthermore, alternative methods disclosed in patents like CN201010116167.6 utilized iron trichloride for deprotection, which, while effective, often yielded products with purity levels hovering around only 92% and overall yields restricted to the 58-62% range. These limitations necessitate extensive downstream processing to meet pharmacopeial standards.
Another conventional approach involved the use of macroporous adsorbent resins for purification, as seen in CN201010100852.X. While effective at removing certain contaminants, this technique introduced substantial operational complexity. The requirement for large volumes of solvents to wash out the adsorbed product significantly increased production costs and environmental waste. Additionally, the multi-stage nature of adsorption and desorption often led to a reduction in total recovery, negatively impacting the overall economics of cost reduction in pharmaceutical intermediates manufacturing. These legacy methods struggle to provide the consistent quality and efficiency required by modern supply chains.
The Novel Approach
The methodology outlined in CN102718780A represents a strategic shift towards process intensification and purity enhancement. Instead of attempting to crystallize the free acid directly from water—a process known to be difficult and prone to trapping impurities—this novel approach converts the cefmetazole acid into a stable organic amine salt. By selecting specific organic amines such as dicyclohexylamine, the process leverages solubility differences to precipitate the product in a highly purified form. This crystallization step effectively acts as a purification barrier, removing soluble impurities that would otherwise persist in the final API.
Furthermore, the transition from the amine salt to the final sodium salt is achieved through a sophisticated ion exchange mechanism rather than direct neutralization with strong bases. This gentle conversion preserves the integrity of the sensitive beta-lactam ring while ensuring the complete removal of the organic amine. The result is a streamlined workflow that eliminates the need for complex macroporous resin washing stages. This innovation not only simplifies the operational procedure but also drastically improves the gross weight yield to over 72% and pushes product gas purity to exceed 99.5%, setting a new benchmark for commercial scale-up of complex antibiotics.
Mechanistic Insights into Lewis Acid Deprotection and Ion Exchange
The core of this synthesis lies in the precise manipulation of the cephalosporin nucleus. The process begins with the acylation of 7-Methoxycephalosporanic acid (7-MAC) using cyanomethylthioacetyl chloride, generated in situ from phosphorus pentachloride and potassium cyanomethylthioacetate. This reaction must be conducted at low temperatures, typically between -40°C and -30°C, to prevent degradation of the beta-lactam ring. Following acylation, the benzhydryl protecting group is removed using a Lewis acid system comprising aluminum chloride and anisole. This step is critical; the aluminum chloride coordinates with the ester oxygen, facilitating cleavage under mild conditions that avoid the harsh acidity of traditional mineral acids.
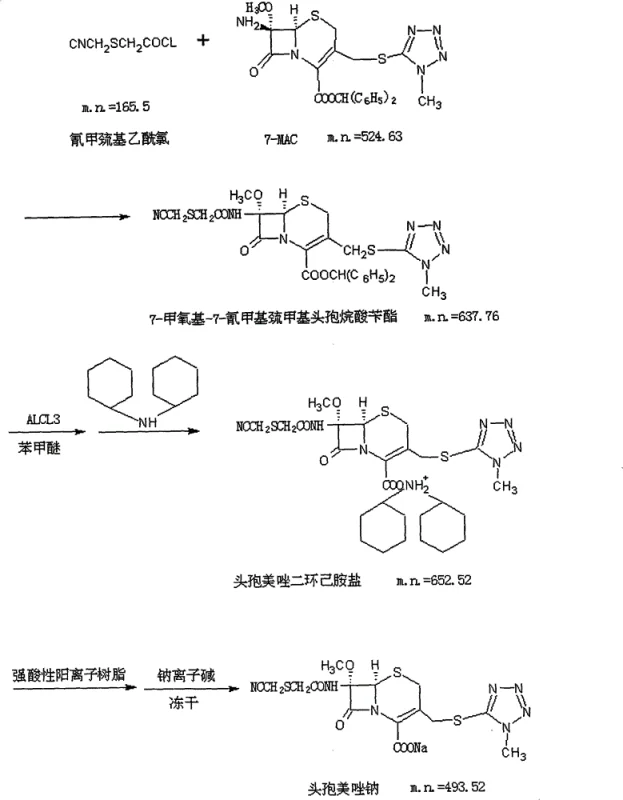
Following deprotection, the mechanism diverges significantly from traditional routes. The resulting cefmetazole acid is treated with an organic amine to form an insoluble amine salt. This precipitation is not merely a isolation step but a thermodynamic purification event where the crystal lattice excludes impurities. The final transformation involves dissolving this amine salt and passing it through a strongly acidic cation exchange resin. The resin selectively binds the protonated organic amine cations, releasing protons that are subsequently neutralized by a sodium base (such as sodium bicarbonate) to form the sodium salt. This ion-exchange mechanism ensures that no residual organic amines remain in the final product, a common failure point in other synthesis routes.
Impurity control is inherently built into this mechanistic pathway. The use of anisole as a scavenger during the Lewis acid deprotection prevents alkylation side reactions on the electron-rich heterocycles. Moreover, the crystallization of the amine salt removes colored byproducts and polymeric impurities that often arise during the acylation and deprotection phases. By avoiding the use of strong alkalis like sodium hydroxide until the very final pH adjustment, the process minimizes the risk of beta-lactam ring opening, which is a primary degradation pathway for cephalosporins. This meticulous control over reaction conditions and intermediate states is what allows for the achievement of such high purity levels.
How to Synthesize Cefmetazole Sodium Efficiently
Implementing this synthesis route requires careful attention to temperature control and stoichiometry, particularly during the acylation and deprotection phases. The patent details specific ratios, such as maintaining a mass ratio of phosphorus pentachloride to potassium cyanomethylthioacetate between 1:1.0 and 1:1.5, to ensure complete activation of the side chain without excess reagent carryover. The subsequent formation of the amine salt requires precise pH adjustment to the 6.0-9.0 range to maximize precipitation efficiency. For a detailed breakdown of the operational parameters, reagents, and specific workup procedures, please refer to the standardized synthesis guide below.
- Prepare cefmetazole benzhydryl ester by reacting 7-MAC with cyanomethylthioacetyl chloride in dichloromethane using an organic base.
- Deprotect the benzhydryl ester using aluminum chloride in anisole to form cefmetazole acid, then convert to a stable amine salt using dicyclohexylamine or similar organic amines.
- Dissolve the amine salt, pass through a strong acidic cation exchange resin to remove the amine, adjust pH with sodium base, and freeze-dry to obtain sterile Cefmetazole Sodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible operational benefits that extend beyond simple yield metrics. The elimination of complex purification stages, such as macroporous resin washing, significantly reduces solvent consumption and waste disposal costs. This streamlining of the manufacturing process leads to a more predictable production schedule, reducing the risk of delays caused by lengthy downstream processing. Consequently, this enhances the reliability of supply for critical antibiotic intermediates, ensuring that downstream formulation teams receive materials on time.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the simplification of unit operations. By replacing multi-step resin adsorption and washing protocols with a direct crystallization and ion exchange sequence, the method drastically reduces the volume of organic solvents required. Furthermore, the avoidance of expensive heavy metal catalysts or complex scavenging agents lowers the raw material bill. The higher overall yield means less starting material is wasted per kilogram of final product, driving down the cost of goods sold (COGS) substantially without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the amine salt intermediate provides a stable checkpoint in the manufacturing workflow. Unlike free acids which may be hygroscopic or unstable, the amine salt can be dried and stored with minimal degradation, offering flexibility in production scheduling. This stability reduces the pressure on immediate downstream processing, allowing for batch consolidation and more efficient use of freeze-drying capacity. Such operational flexibility is crucial for maintaining continuous supply lines in the face of fluctuating market demand.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in solvent usage and the elimination of heavy metal residues align with increasingly stringent green chemistry regulations. The process generates less hazardous waste, simplifying effluent treatment and reducing the environmental footprint of the facility. The scalability is further supported by the use of standard equipment such as filtration units and ion exchange columns, which are easily scaled from pilot to commercial production without requiring specialized or exotic reactor configurations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Cefmetazole Sodium using this advanced methodology. These insights are derived directly from the technical specifications and comparative data provided in the patent literature, offering clarity on how this process differentiates itself from legacy methods. Understanding these details is vital for technical teams evaluating potential suppliers.
Q: How does the new method improve purity compared to traditional sodium hydroxide neutralization?
A: Traditional methods using strong bases like sodium hydroxide often lead to excessive impurities and dark product color. This patented method utilizes organic amine salt formation followed by ion exchange, which acts as an additional purification step, significantly reducing impurities and ensuring a white, high-purity final product.
Q: What are the advantages of using ion exchange resin over macroporous adsorption resins?
A: Macroporous adsorption resins require extensive solvent washing, which complicates post-treatment and increases costs. The use of strong acidic cation exchange resin in this process simplifies the workflow by directly exchanging the organic amine cation for sodium, eliminating the need for complex solvent washouts and improving overall recovery rates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It avoids difficult crystallization steps associated with free acid forms and utilizes standard unit operations like filtration and freeze-drying, making it highly adaptable for commercial scale-up from pilot plants to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefmetazole Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into practical, commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity promised by CN102718780A are realized in every batch. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze the complex impurity profiles of cephalosporins, guaranteeing that our Cefmetazole Sodium meets the highest global pharmacopeial standards.
We invite pharmaceutical partners to collaborate with us to leverage this superior synthesis route for their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain security.
