Advanced Manufacturing of Cefmetazole Sodium: A Cost-Effective Route for Global Antibiotic Supply Chains
Advanced Manufacturing of Cefmetazole Sodium: A Cost-Effective Route for Global Antibiotic Supply Chains
The global demand for second-generation cephalosporins remains robust, driven by the persistent need for effective treatments against Gram-negative bacterial infections. At the forefront of this therapeutic class is Cefmetazole Sodium, a potent agent known for its stability against beta-lactamases produced by organisms such as E. coli and Proteus mirabilis. However, the commercial viability of this API often hinges on the efficiency of its synthetic route. A pivotal advancement in this domain is detailed in patent CN101787040B, which outlines a novel preparation method that significantly enhances both yield and purity while mitigating environmental impact. This technical insight report analyzes the strategic advantages of this methodology for R&D directors and supply chain leaders seeking a reliable cefmetazole sodium supplier. By shifting away from harsh alkaline hydrolysis towards a Lewis acid-mediated deprotection strategy, manufacturers can achieve product purity levels of 92% with yields between 58% and 62%. 
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefmetazole derivatives has been plagued by significant downstream processing challenges, particularly during the deprotection phase. Traditional routes, such as those disclosed in earlier literature involving tosyl chloride activation or direct alkaline hydrolysis, often suffer from severe drawbacks that compromise commercial feasibility. For instance, methods utilizing strong bases like sodium hydroxide for removing protecting groups frequently induce degradation of the sensitive beta-lactam ring, leading to a proliferation of polymeric impurities and open-ring byproducts. Furthermore, these conventional processes often result in products with suboptimal physical characteristics, such as dark coloration and low assay content, necessitating costly and time-consuming purification steps like repeated recrystallization or chromatography. The reliance on such aggressive chemical conditions not only diminishes the overall mass balance but also generates substantial hazardous waste streams, creating a bottleneck for cost reduction in antibiotic manufacturing and complicating regulatory compliance regarding residual solvents and heavy metals.
The Novel Approach
In stark contrast, the methodology presented in patent CN101787040B introduces a refined synthetic pathway that elegantly circumvents these historical pitfalls through the strategic application of Lewis acid chemistry. The core innovation lies in the replacement of strong alkaline deprotection with an iron trichloride (FeCl3) mediated cleavage of the benzyl ester intermediate. This approach allows for the gentle removal of the protecting group under controlled acidic conditions, thereby preserving the integrity of the 7-methoxy-cephem nucleus which is notoriously susceptible to base-catalyzed hydrolysis. Additionally, the process incorporates a highly efficient side-chain coupling step using phosphorus pentachloride (PCl5) activation at cryogenic temperatures ranging from -100°C to -10°C, ensuring high stereochemical fidelity. By integrating these specific reaction parameters, the novel route achieves a streamlined workflow that minimizes impurity formation from the outset, ultimately delivering a high-purity intermediate that requires less intensive finishing, thus offering substantial cost savings and operational simplicity for large-scale producers.
Mechanistic Insights into FeCl3-Catalyzed Deprotection and Side-Chain Coupling
The chemical elegance of this synthesis is best understood by examining the mechanistic nuances of the side-chain preparation and the subsequent deprotection. The process initiates with the formation of the critical cyanomethylthioacetic acid side chain, a reaction that must be tightly controlled to prevent polymerization or over-alkylation. As illustrated in the reaction scheme, mercaptoacetic acid reacts with chloroacetonitrile in an aqueous sodium hydroxide medium, where precise pH control and temperature maintenance between 8°C and 12°C are paramount to maximizing the nucleophilic attack of the thiolate anode on the nitrile-bearing carbon. 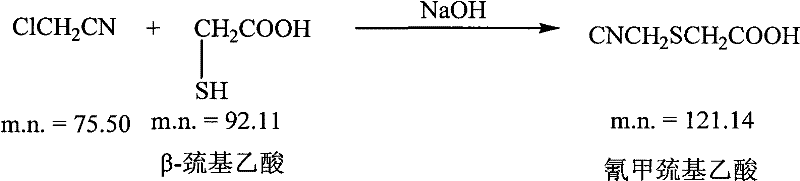
Following the isolation of the side chain, the subsequent acylation of the 7-MAC (7-methoxy-7-aminocephalosporanic acid) nucleus requires rigorous exclusion of moisture and thermal management to prevent epimerization at the C-7 position. The use of PCl5 generates a reactive acid chloride in situ, which then couples with the amine of the cephem core. However, the true breakthrough occurs in the final deprotection step shown below, where the benzyl ester is cleaved. Unlike traditional hydrogenolysis which requires expensive palladium catalysts and poses safety risks with hydrogen gas, or saponification which risks ring opening, the FeCl3 method utilizes the hard Lewis acid character of iron to coordinate with the carbonyl oxygen, facilitating cleavage without disturbing the beta-lactam bond. 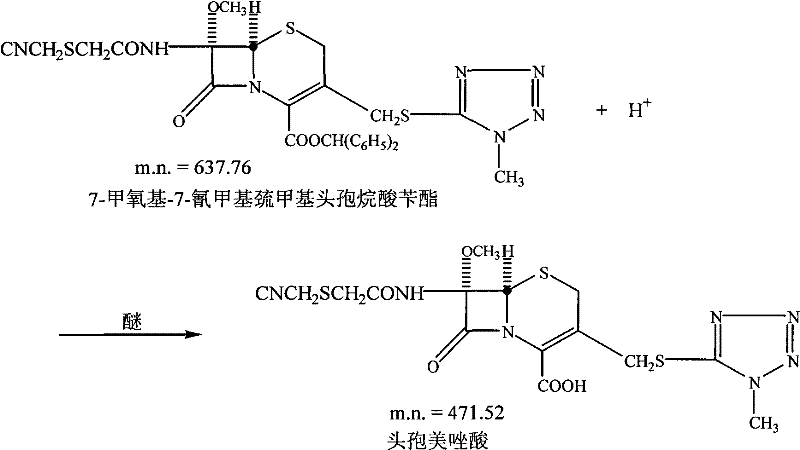
This mechanism effectively suppresses the formation of delta-2 isomers and other regio-isomeric impurities that typically plague cephalosporin synthesis. The patent specifies the use of ether as a co-solvent for the FeCl3, which likely aids in solubilizing the iron complex while maintaining a non-nucleophilic environment. Furthermore, the workup procedure involves a sophisticated phase separation strategy where the product is extracted into an aqueous bicarbonate layer, leaving organic-soluble impurities behind in the dichloromethane phase. This liquid-liquid extraction acts as a built-in purification step, significantly enhancing the impurity profile before the final crystallization. The ability to seed the crystallization of cefmetazole acid directly from the aqueous phase ensures consistent particle size distribution and polymorphic form, which are critical quality attributes for downstream formulation into injectable dosage forms.
How to Synthesize Cefmetazole Sodium Efficiently
The implementation of this synthetic route requires strict adherence to the operational parameters defined in the patent to ensure reproducibility and safety. The process is divided into four distinct unit operations: side-chain synthesis, activated ester formation, Lewis acid deprotection, and salt formation. Each step demands precise stoichiometric control, particularly the ratio of phosphorus pentachloride to the side chain acid, which is maintained at approximately 1ml to 2.5g to ensure complete activation without excess reagent carryover. The cryogenic conditions during acylation are non-negotiable for maintaining the 7-alpha-methoxy configuration, which is essential for the antibiotic's resistance to beta-lactamases. Detailed standardized operating procedures for temperature ramping, phase separation times, and crystallization seeding protocols are essential for technology transfer.
- Preparation of cyanomethylthioacetic acid side chain by reacting mercaptoacetic acid with chloroacetonitrile in alkaline aqueous solution followed by acidification and extraction.
- Activation of the side chain using phosphorus pentachloride (PCl5) and subsequent acylation of 7-MAC at low temperatures (-100°C to -10°C) to form the benzyl ester intermediate.
- Deprotection of the benzyl ester using iron trichloride (FeCl3) in ether/dichloromethane to yield cefmetazole acid, avoiding strong alkaline hydrolysis.
- Conversion of cefmetazole acid to the final sodium salt using sodium bicarbonate in water followed by freeze-drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits beyond mere technical superiority. The primary advantage lies in the significant simplification of the supply chain for raw materials; the process relies on commodity chemicals such as mercaptoacetic acid, chloroacetonitrile, and iron trichloride, all of which are readily available in the global market with stable pricing dynamics. This reduces the risk of supply disruption compared to routes requiring specialized enzymes or rare metal catalysts. Moreover, the elimination of expensive noble metal catalysts like palladium, which are traditionally used for benzyl deprotection, removes a major cost driver and eliminates the need for complex metal scavenging steps to meet stringent ICH Q3D guidelines for elemental impurities. This inherent process safety and material availability contribute to a more resilient supply chain capable of withstanding market volatility.
- Cost Reduction in Manufacturing: The economic model of this synthesis is bolstered by the high overall yield of 58% to 62%, which is competitive for a multi-step cephalosporin synthesis. By avoiding the formation of dark-colored impurities associated with alkaline hydrolysis, the process reduces the need for extensive decolorization treatments using large quantities of activated carbon, thereby lowering material costs and waste disposal fees. Furthermore, the use of FeCl3 is economically favorable compared to enzymatic methods which require costly biocatalysts and strict buffer systems. The streamlined workup, involving direct phase separation and crystallization, minimizes solvent consumption and energy usage associated with distillation and drying, leading to a lower cost of goods sold (COGS) per kilogram of active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this chemical route ensures consistent batch-to-batch quality, which is critical for maintaining regulatory filings and avoiding production stoppages. The process tolerance, evidenced by the specific pH and temperature ranges provided in the patent, suggests a wide operating window that is forgiving of minor variations in industrial reactors. This reliability reduces the lead time for high-purity cefmetazole sodium batches, as there is less likelihood of batch failure or the need for reprocessing. Additionally, the final freeze-drying step for salt formation is a standard unit operation in most API facilities, meaning that existing infrastructure can be utilized without significant capital expenditure on new equipment, further accelerating time-to-market for generic manufacturers.
- Scalability and Environmental Compliance: The patent explicitly highlights the environmental friendliness of the method, a key factor for modern sustainable manufacturing. The avoidance of strong alkalis reduces the load on wastewater treatment plants, as the effluent is less corrosive and easier to neutralize. The solvent system, primarily consisting of dichloromethane, ethyl acetate, and ether, allows for efficient recovery and recycling through standard distillation trains, aligning with green chemistry principles of solvent reuse. The scalability is proven by the successful transition from laboratory to pilot scales described in the embodiments, demonstrating that the exothermic nature of the PCl5 activation and the FeCl3 deprotection can be safely managed in large vessels. This makes the commercial scale-up of complex cephalosporin intermediates feasible for contract development and manufacturing organizations (CDMOs) looking to expand their antibiotic portfolio.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of cefmetazole sodium using this specific patented route. These insights are derived directly from the experimental data and comparative analysis provided in patent CN101787040B, offering clarity on why this method represents a superior choice for industrial synthesis. Understanding these nuances helps stakeholders make informed decisions regarding technology licensing or procurement strategies.
Q: Why is the FeCl3 deprotection method superior to traditional alkaline hydrolysis for Cefmetazole?
A: Traditional methods using strong alkali like sodium hydroxide often lead to excessive impurities, low content, and dark product color due to beta-lactam ring instability. The patented FeCl3 method operates under milder conditions, preserving the core structure and achieving 92% purity.
Q: What is the expected yield and purity of this synthesis route?
A: According to patent CN101787040B, this optimized process delivers a total yield ranging from 58% to 62% with a product purity reaching 92%, making it highly suitable for industrial-scale antibiotic production.
Q: Is this process scalable for commercial manufacturing?
A: Yes, the patent explicitly states the method is environmentally friendly, low-cost, and applicable to industrial production. The use of standard reagents like PCl5 and FeCl3 facilitates easy scale-up compared to complex enzymatic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefmetazole Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust manufacturing capabilities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate temperature controls and phase separation techniques required for cefmetazole synthesis are executed with precision. Our state-of-the-art facilities are equipped with cryogenic reactors capable of maintaining the -100°C conditions necessary for the acylation step, alongside rigorous QC labs that enforce stringent purity specifications to guarantee every batch meets the 92% purity benchmark outlined in the patent. We are committed to delivering high-purity cefmetazole sodium that supports the global fight against bacterial infections while adhering to the highest standards of quality and safety.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthetic route for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this FeCl3-mediated process can optimize your COGS. We encourage potential clients to contact us directly to obtain specific COA data from our recent pilot batches and to discuss route feasibility assessments for your upcoming projects. Let us be your trusted partner in securing a stable, high-quality supply of this critical antibiotic intermediate.
