Advanced Synthetic Route for Cefmetazole Sodium: Enhancing Purity and Scalability for Global Pharma Markets
Advanced Synthetic Route for Cefmetazole Sodium: Enhancing Purity and Scalability for Global Pharma Markets
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antibiotics, particularly second-generation cephalosporins like Cefmetazole Sodium. Patent CN101550151B introduces a transformative synthetic methodology that addresses long-standing inefficiencies in the production of this vital antimicrobial agent. This technology leverages a streamlined acylation strategy using p-toluenesulfonyl chloride activation to couple a specialized side chain with a cephem nucleus, resulting in superior yields and purity profiles. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for broad-spectrum antibiotics used in treating respiratory, urinary, and soft tissue infections. The structural complexity of Cefmetazole Sodium, characterized by its 7-alpha-methoxy group and tetrazole thiomethyl side chain, demands precise chemical control to ensure efficacy and safety.
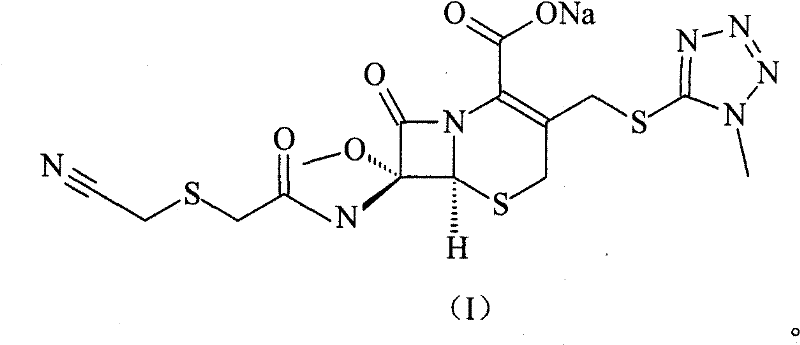
As a reliable cefmetazole sodium supplier, understanding the nuances of this synthesis is paramount for ensuring consistent quality. The patent details a method that bypasses the cumbersome multi-step protections and deprotections often associated with cephalosporin chemistry. Instead, it utilizes a direct coupling approach that minimizes exposure of the sensitive beta-lactam ring to harsh conditions. This not only enhances the overall throughput but also significantly lowers the impurity burden, a critical factor for regulatory compliance in global markets. The ability to produce high-purity intermediates efficiently is a key differentiator in the competitive landscape of generic antibiotic manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefmetazole Sodium has been plagued by low efficiency and complex operational procedures. Prior art, such as methods disclosed in academic journals, often described a four-step reaction sequence starting from 7-ACA. These traditional routes typically suffered from cumulative yield losses, with total recovery rates hovering around 57%. Such low efficiency translates directly into higher production costs and increased waste generation, posing significant challenges for cost reduction in cephalosporin manufacturing. Furthermore, multi-step syntheses increase the risk of introducing diverse impurities at each stage, complicating the purification process and potentially compromising the safety profile of the final API. The reliance on multiple isolation steps also extends the production cycle time, impacting supply chain responsiveness.
The Novel Approach
In stark contrast, the novel approach detailed in the patent simplifies the synthesis into a more direct and efficient pathway. By reacting 7-beta-amino-7-alpha-methoxy-3-(1-methyl-1H-tetrazole-5-sulfidomethyl)-3-cephem-4-benzyl carboxylate directly with cyanomethylthio acetic acid sodium in the presence of Tosyl chloride, the process achieves a remarkable convergence of steps. This one-pot style activation and coupling strategy eliminates the need for isolating unstable intermediates, thereby preserving the integrity of the cephem nucleus. The visual representation of this streamlined reaction pathway highlights the elegance of the design, where the side chain is activated in situ and immediately consumed by the nucleophilic amine.
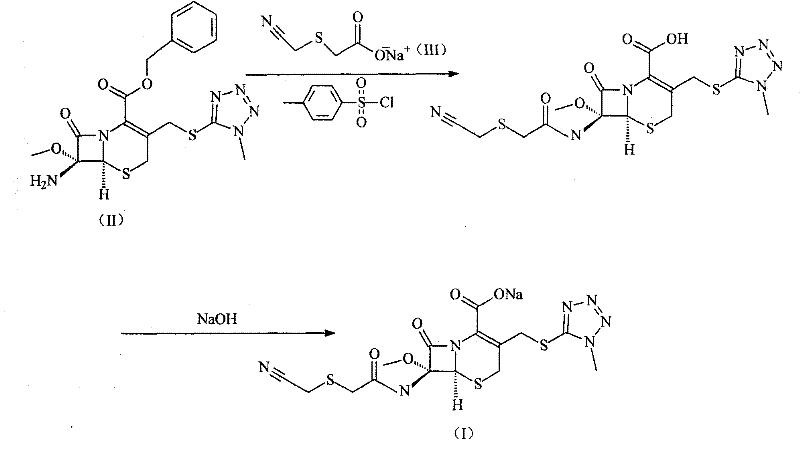
This methodological shift allows for yields exceeding 90%, as demonstrated in the experimental embodiments, representing a substantial improvement over historical benchmarks. The use of common organic solvents like DMF or toluene further enhances the practicality of this route for commercial scale-up of complex pharmaceutical intermediates. By reducing the number of unit operations, manufacturers can achieve faster turnaround times and lower energy consumption, aligning with modern green chemistry principles while maintaining economic viability.
Mechanistic Insights into Tosyl Chloride Mediated Acylation
The core of this synthetic innovation lies in the mechanistic efficiency of the Tosyl chloride mediated acylation. In the initial phase, cyanomethylthio acetic acid sodium acts as the nucleophile precursor, which is activated by p-toluenesulfonyl chloride to form a highly reactive mixed anhydride or sulfonyl ester species. This activation step is conducted at controlled low temperatures, typically between 5°C and 10°C, to prevent the decomposition of the activating agent and the acid substrate. The subsequent addition of the cephem amine, specifically the 7-beta-amino group, triggers a nucleophilic attack on the activated carbonyl carbon. This stereoselective acylation is crucial, as it must proceed without epimerizing the chiral centers at the C6 and C7 positions of the beta-lactam ring.
Impurity control is inherently built into this mechanism through strict temperature regulation and pH management. The beta-lactam ring is notoriously susceptible to hydrolysis under acidic or basic extremes; therefore, the process maintains a delicate balance. After the coupling reaction, the mixture is acidified to generate the free acid form of Cefmetazole, followed by a careful neutralization with sodium hydroxide to adjust the pH to the 7.0-8.0 range. This specific pH window ensures the formation of the stable sodium salt while minimizing the formation of ring-opened byproducts or polymeric impurities. The final lyophilization step removes residual solvents and water, yielding a product with a purity profile that meets stringent pharmacopoeial standards, essential for any high-purity API intermediate.
How to Synthesize Cefmetazole Sodium Efficiently
Implementing this synthesis requires precise adherence to the reaction parameters outlined in the patent to maximize yield and safety. The process begins with the dissolution of the side chain acid salt in a polar aprotic solvent, followed by the controlled addition of the activating agent. The subsequent coupling with the cephem nucleus must be monitored closely to ensure complete conversion before proceeding to the workup phase. Detailed standard operating procedures regarding stoichiometry, addition rates, and temperature ramps are critical for reproducibility on a manufacturing scale. For a comprehensive guide on the specific operational steps and safety precautions, please refer to the technical protocol below.
- Dissolve cyanomethylthio acetic acid sodium salt in a polar aprotic solvent such as DMF or DMAc and cool the mixture to between 0°C and 20°C.
- Add p-toluenesulfonyl chloride (Tosyl chloride) to activate the acid, followed by the addition of the 7-beta-amino cephem derivative and triethylamine.
- After reaction completion and acidification, adjust the pH to 7.0-8.0 using sodium hydroxide, wash with ethyl acetate, and lyophilize to obtain the final sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits. The drastic simplification of the reaction sequence directly correlates to reduced operational expenditures, as fewer reactors and less labor are required per batch. The elimination of intermediate isolation steps not only saves time but also reduces the volume of solvents and reagents consumed, leading to substantial cost savings in raw material procurement. Furthermore, the high yield achieved minimizes the amount of starting material needed to produce a fixed quantity of API, effectively lowering the cost of goods sold (COGS) and improving margin potential in a competitive generic market.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive coupling reagents often used in peptide-like bond formations, substituting them with cost-effective Tosyl chloride. By avoiding the isolation of unstable intermediates, the process reduces material loss associated with transfer and purification steps. This efficiency translates into a leaner manufacturing model where resource utilization is optimized, allowing for more competitive pricing strategies without compromising on quality standards.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including 7-ACA derivatives and Tosyl chloride, are commodity chemicals with robust global supply chains. This availability reduces the risk of supply disruptions that can occur with specialized or proprietary reagents. Additionally, the shortened production cycle time means that manufacturers can respond more agilely to fluctuations in market demand, ensuring a steady flow of product to downstream formulation partners and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The use of standard solvents like DMF and toluene facilitates easy scale-up from pilot plants to multi-ton commercial production without requiring exotic equipment. The simplified workup procedure, involving pH adjustment and extraction, generates less hazardous waste compared to multi-step chromatographic purifications. This aligns with increasingly strict environmental regulations, reducing the burden on waste treatment facilities and supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Cefmetazole Sodium using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of the process for industry stakeholders. Understanding these details is crucial for making informed decisions about technology transfer and vendor selection.
Q: What are the primary advantages of this synthetic route over traditional methods?
A: This method significantly reduces the number of reaction steps compared to prior art, which often required four distinct steps with a total recovery of only around 57%. By utilizing a direct acylation strategy with Tosyl chloride activation, the process achieves yields exceeding 90% while maintaining high product purity, thereby reducing waste and processing time.
Q: How does the process ensure the stability of the beta-lactam ring during synthesis?
A: The protocol strictly controls reaction temperatures, maintaining the initial activation and coupling phases between 5°C and 10°C. This low-temperature environment is critical for preventing the hydrolysis or degradation of the sensitive beta-lactam core found in the 7-beta-amino-7-alpha-methoxy cephem intermediate, ensuring the structural integrity of the final antibiotic.
Q: Is this manufacturing process suitable for large-scale commercial production?
A: Yes, the process is highly amenable to scale-up due to the use of common industrial solvents like DMF and toluene, and readily available reagents such as Tosyl chloride. The workup procedure involves standard pH adjustment and liquid-liquid extraction, avoiding complex chromatographic purifications that often bottleneck large-scale API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefmetazole Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for essential antibiotics like Cefmetazole Sodium. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch against global pharmacopoeial standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pharmaceutical projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and reliability in your supply chain.
