Advanced Synthesis of 3-(Alpha-Methoxy)-Methylenebenzofuran-2(3H)-One for Scalable Agrochemical Production
The global demand for high-efficiency strobilurin fungicides, particularly Azoxystrobin, has necessitated the development of robust and cost-effective synthetic routes for key intermediates like 3-(α-methoxy)-methylenebenzofuran-2(3H)-one. Patent CN102199137A introduces a transformative methodology that addresses the critical bottlenecks of traditional synthesis, offering a pathway characterized by superior atom economy and operational simplicity. This innovation leverages a two-step sequence involving base-catalyzed formylation followed by selective methoxylation, utilizing widely accessible reagents such as alkali metal alkoxides and formate esters. For R&D directors and process chemists, this represents a significant leap forward in impurity profile control, as the avoidance of acetic anhydride minimizes the formation of difficult-to-remove acetate byproducts. Furthermore, the mild reaction conditions, ranging from -10°C to 60°C, ensure enhanced safety profiles and reduced energy consumption during commercial scale-up. By shifting away from complex orthoformate chemistry, this patent provides a foundational technology for reliable agrochemical intermediate supplier networks seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical benzofuran derivative relied heavily on the condensation of benzofuran-2(3H)-one with trimethyl orthoformate in the presence of acetic anhydride, a process fraught with significant economic and environmental inefficiencies. As illustrated in the conventional reaction pathway below, acetic anhydride serves a dual role as both a catalyst and a dehydrating agent to drive the equilibrium, yet this results in the stoichiometric generation of large quantities of methyl acetate as a waste byproduct.
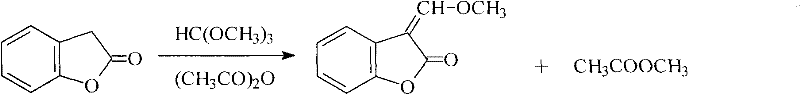
This reliance on acetic anhydride not only inflates raw material costs due to the consumption of a stoichiometric amount of reagent but also complicates the downstream purification process, requiring extensive distillation or washing steps to remove the volatile ester side products. Moreover, the reaction kinetics in the traditional method are often sluggish, necessitating prolonged heating periods that degrade thermal stability and lead to lower overall yields, thereby increasing the cost per kilogram of the final active pharmaceutical ingredient. The accumulation of acetate impurities can also interfere with subsequent coupling reactions in the Azoxystrobin synthesis tree, potentially compromising the purity specifications required by regulatory bodies for agricultural applications. Consequently, procurement managers have long identified this legacy route as a major source of supply chain volatility and margin erosion in the production of strobilurin fungicides.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a direct formylation strategy employing methyl formate or ethyl formate catalyzed by strong bases such as sodium hydride or sodium methoxide, effectively bypassing the need for acetic anhydride entirely. This strategic shift allows for a much cleaner reaction profile where the primary byproduct is simply the corresponding alcohol from the formate ester, which is far easier to manage and remove than methyl acetate. The process operates under milder thermal conditions, typically initiating at sub-zero temperatures to control exotherms before warming to ambient or slightly elevated temperatures, which preserves the integrity of the sensitive lactone ring structure. By decoupling the formylation and methoxylation steps, chemists gain precise control over the reaction stoichiometry, ensuring that the intermediate 3-formylbenzofuran-2(3H)-one is generated with high fidelity before proceeding to the final etherification. This modularity not only enhances the robustness of the manufacturing process but also provides flexibility in sourcing raw materials, as various alkoxide catalysts can be substituted based on availability and cost without compromising the reaction outcome.
Mechanistic Insights into Base-Catalyzed Formylation and Methoxylation
The core of this technological advancement lies in the efficient generation of the enolate ion from benzofuran-2(3H)-one using strong non-nucleophilic or alkoxide bases, which then attacks the carbonyl carbon of the formate ester to install the crucial aldehyde functionality. In the first stage, the base abstracts the acidic proton at the C3 position of the lactone ring, creating a resonance-stabilized nucleophile that reacts rapidly with methyl formate to form the 3-formyl intermediate. This step is critical because it avoids the equilibrium limitations seen in orthoformate reactions, driving the conversion to completion through the irreversible formation of the alkoxide leaving group. Following isolation or in-situ processing, the second stage involves the nucleophilic attack of the enol form of the aldehyde intermediate on dimethyl sulfate, facilitated by a weaker base like potassium carbonate. This methoxylation step proceeds via an SN2 mechanism, where the methoxy group is transferred to the alpha-carbon, establishing the conjugated exocyclic double bond essential for the biological activity of the final fungicide.
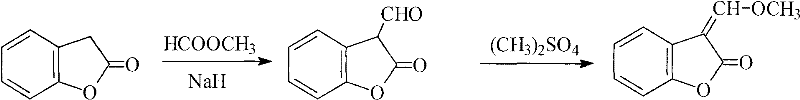
From an impurity control perspective, this mechanism offers distinct advantages by eliminating the acidic conditions and high temperatures that often promote ring-opening hydrolysis of the lactone moiety. The use of anhydrous DMF as a solvent ensures that the reactive enolate species remains stable and soluble, preventing premature precipitation or side reactions that could lead to polymeric tars. Furthermore, the separation of the formylation and methoxylation steps allows for intermediate quality checks, ensuring that any unreacted starting material is removed before the introduction of the genotoxic alert reagent, dimethyl sulfate. This level of process control is paramount for meeting the stringent purity standards required for high-purity agrochemical intermediate manufacturing, as it minimizes the risk of carryover impurities that could affect crop safety or environmental toxicity profiles. The result is a crystalline product with a sharp melting point range of 100-103°C, indicative of high chemical homogeneity and suitability for direct use in downstream coupling reactions.
How to Synthesize 3-(Alpha-Methoxy)-Methylenebenzofuran-2(3H)-One Efficiently
The implementation of this synthesis route requires careful attention to moisture control and temperature management to maximize yield and safety. The process begins with the preparation of a dry reaction vessel charged with anhydrous DMF, into which the lactone and formate ester are introduced under an inert atmosphere.
- Perform formylation of benzofuran-2(3H)-one using methyl formate and an alkoxide catalyst (e.g., sodium methoxide) in anhydrous DMF at -10 to 45°C.
- Hydrolyze the reaction mixture with ice water, adjust pH to 4, and extract the intermediate 3-formylbenzofuran-2(3H)-one.
- React the isolated intermediate with dimethyl sulfate and potassium carbonate in DMF at 0-60°C to obtain the final methoxylated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into tangible operational efficiencies and risk mitigation strategies. The substitution of expensive and specialized reagents like trimethyl orthoformate with commodity chemicals such as methyl formate and sodium methoxide drastically simplifies the raw material sourcing landscape, reducing dependency on niche suppliers and enhancing supply continuity. Additionally, the elimination of acetic anhydride removes a significant waste stream, lowering the costs associated with solvent recovery and hazardous waste disposal, which is increasingly critical in regions with strict environmental regulations. The higher yields reported in the patent examples, consistently reaching above 80%, mean that less starting material is required to produce the same volume of finished goods, effectively increasing the throughput of existing reactor capacity without capital expenditure. These factors combine to create a more resilient and cost-competitive supply chain for the production of Azoxystrobin and related strobilurin analogues.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of stoichiometric acetic anhydride with catalytic amounts of inexpensive alkoxides, which fundamentally alters the cost structure of the synthesis. By avoiding the generation of large volumes of methyl acetate byproduct, the facility saves significantly on energy costs related to distillation and solvent recycling, while also reducing the load on wastewater treatment plants. The use of dimethyl sulfate, while requiring careful handling, is a highly efficient methylating agent that offers superior atom economy compared to alternative methylation strategies, further driving down the variable cost per unit. Moreover, the ability to operate at near-ambient temperatures for significant portions of the reaction reduces the utility burden associated with heating and cooling, contributing to a lower overall carbon footprint and operational expense.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key reagents, including benzofuran-2(3H)-one, methyl formate, and dimethyl sulfate, are produced on a massive global scale for various industrial applications, ensuring consistent availability and price stability. Unlike specialized catalysts or proprietary reagents that may face supply disruptions, these commodity chemicals can be sourced from multiple qualified vendors, mitigating the risk of single-source dependency. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, allowing for greater flexibility in vendor selection and inventory management. This reliability is crucial for maintaining uninterrupted production schedules for downstream fungicide formulations, ensuring that agricultural markets are supplied consistently throughout the growing season.
- Scalability and Environmental Compliance: The process design inherently supports commercial scale-up of complex agrochemical intermediates due to its linear workflow and manageable exothermic profile, which can be easily controlled in large-scale reactors using standard jacketed vessels. The reduction in hazardous waste generation aligns with modern green chemistry principles, facilitating easier permitting and compliance with increasingly stringent environmental protection laws in major manufacturing hubs. The simplified workup procedure, involving straightforward aqueous extraction and crystallization, reduces the complexity of the isolation phase, allowing for faster batch turnover times and higher asset utilization. This scalability ensures that manufacturers can respond rapidly to fluctuations in market demand for Azoxystrobin without the need for complex process re-engineering or significant infrastructure investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology.
Q: What are the primary advantages of the alkoxide-catalyzed route over the traditional orthoformate method?
A: The alkoxide-catalyzed route eliminates the need for expensive trimethyl orthoformate and acetic anhydride, significantly reducing raw material costs and minimizing the generation of methyl acetate byproducts, which simplifies downstream purification.
Q: What is the typical yield range for this synthesis method?
A: According to patent data, the optimized process achieves yields ranging from 82% to 90%, demonstrating high efficiency compared to older methodologies that often suffered from prolonged reaction times and lower conversion rates.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common industrial solvents like DMF and readily available reagents such as methyl formate and dimethyl sulfate, making it highly scalable with manageable exothermic profiles and simplified waste treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(Alpha-Methoxy)-Methylenebenzofuran-2(3H)-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy and safety of modern crop protection solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 3-(α-methoxy)-methylenebenzofuran-2(3H)-one meets the exacting standards required for the synthesis of premium fungicides. Our commitment to quality assurance extends beyond mere compliance, as we actively collaborate with clients to refine process parameters for optimal yield and impurity control.
We invite global partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this alkoxide-catalyzed method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific production requirements, ensuring a partnership built on transparency, technical excellence, and mutual growth.
