Advanced One-Pot Synthesis of Panipenem: Enhancing Purity and Scalability for Global Supply Chains
Introduction to Advanced Carbapenem Manufacturing
The pharmaceutical landscape for broad-spectrum antibiotics continues to evolve, with carbapenems remaining a cornerstone for treating resistant bacterial infections. Specifically, Panipenem (RS533), chemically known as (5R, 6S)-6-[(1R)-1-hydroxyethyl]-3-[[(3S)-1-(1-iminoethyl)-3-pyrrolidinyl]thio]-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid, represents a critical therapeutic agent with efficacy comparable to imipenem but with a distinct safety profile suitable for pediatric applications. The industrial preparation of such complex beta-lactam structures has historically been plagued by low yields, difficult purification steps, and stability issues during intermediate handling. However, recent technological advancements disclosed in patent CN101885727A offer a transformative approach to this challenge. This patent details a robust methodology that streamlines the synthesis through a telescoped phosphorylation and coupling sequence, effectively bypassing the isolation of unstable intermediates. For global supply chain stakeholders, this innovation signals a potential shift towards more reliable API intermediate supplier capabilities, ensuring consistent availability of high-quality active ingredients.
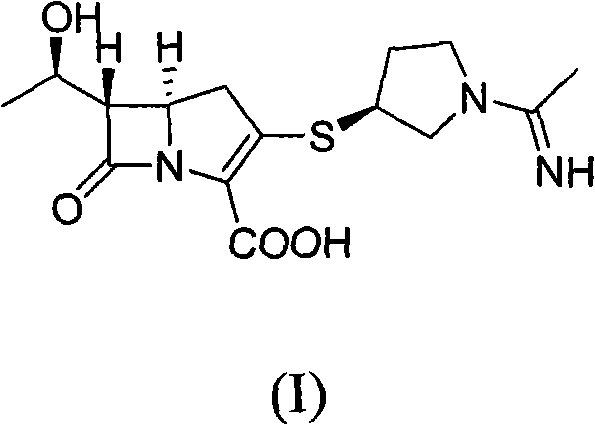
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Panipenem and related carbapenems has relied on stepwise protection and coupling strategies that introduce significant operational bottlenecks. Traditional routes, such as those described in US RE34960 and EP 0161546, typically involve the phosphorylation of the parent nucleus followed by condensation with a side chain to obtain a double-protected intermediate. A major drawback of these legacy processes is the stringent requirement for pH control during the iminoethyl connection step, which restricts operational flexibility and increases the risk of batch failure. Furthermore, when benzyl or substituted benzyl groups are used as protecting groups, the resulting intermediates often exhibit extremely poor solubility in common organic solvents like acetonitrile, methylene chloride, or ethyl acetate. This poor solubility frequently leads to the formation of intractable "soup" compounds within the reaction system, making separation and purification exceptionally difficult. Consequently, subsequent hydrogenation steps often require heating the system above 40°C to dissolve the intermediate, a condition that drastically accelerates the rate of decomposition and severely compromises the overall yield and quality of the final product.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the method disclosed in CN101885727A introduces a highly efficient "one-pot" strategy that fundamentally restructures the synthesis workflow. The core innovation lies in the direct reaction of the starting ketone with phosphorus oxychloride to generate the enol phosphate intermediate (Compound II) in situ, without any isolation step. This reactive species is immediately coupled with the pyrrolidine-thiol side chain (Compound V) to yield the double-protected Panipenem (Compound VI). By eliminating the isolation of the unstable phosphate intermediate, the process minimizes exposure to degradation conditions and significantly reduces the total processing time. Moreover, the optimized solvent system allows the double-protected intermediate to crystallize directly from the reaction mixture in a high-purity solid state, completely avoiding the "soup" formation issues associated with prior art. This streamlined approach not only simplifies the equipment requirements but also drastically lowers the environmental burden by reducing the types and quantities of solvents needed, representing a substantial leap forward in cost reduction in pharmaceutical manufacturing.
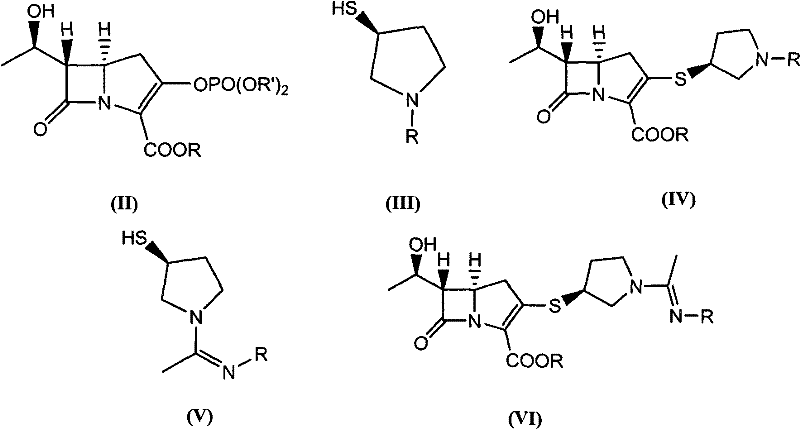
Mechanistic Insights into Telescoped Phosphorylation and Coupling
The chemical elegance of this process is rooted in the precise control of reactivity during the phosphorylation phase. The reaction initiates with the treatment of the carbapenem ketone with phosphoryl chloride in the presence of an organic base, such as diisopropylethylamine or triethylamine, within a high-polarity aprotic solvent like N-methylpyrrolidone (NMP) or dimethylformamide (DMF). Maintaining the reaction temperature between -60°C and 10°C is critical to stabilize the generated enol phosphate species (Compound II), preventing premature hydrolysis or rearrangement. Unlike traditional methods where this intermediate would be isolated—a step prone to yield loss due to instability—the novel protocol leverages its transient existence by immediately introducing the nucleophilic thiol side chain (Compound V). The sulfur atom of the pyrrolidine ring attacks the activated carbon of the enol phosphate, displacing the phosphate group and forming the crucial C-S bond that defines the carbapenem side chain architecture. This telescoped mechanism ensures that the reactive intermediate is consumed as soon as it is formed, driving the equilibrium towards the desired product (Compound VI) and minimizing the formation of phosphorylated byproducts.
Following the coupling reaction, the process employs a strategic crystallization technique to isolate the double-protected intermediate. By adding a crystallization solvent such as methanol or ethanol to the reaction mixture, the solubility of Compound VI is sharply reduced, causing it to precipitate out of the solution while impurities remain dissolved. This "reactive crystallization" step serves as a powerful purification tool, achieving purity levels of up to 99% without the need for column chromatography or complex extraction workflows. The subsequent catalytic hydrogenation step utilizes a buffered aqueous-organic system to remove the protecting groups under mild conditions (0-60°C). The use of a buffer system, such as lutidine hydrochloride or phosphate buffers, maintains the pH between 6.8 and 7.5, which is essential for preserving the sensitive beta-lactam ring during hydrogenolysis. This careful mechanistic orchestration ensures that the final Panipenem product retains its stereochemical integrity and biological activity, meeting the rigorous standards required for a high-purity API intermediate.
How to Synthesize Panipenem Efficiently
The implementation of this synthesis route requires precise adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction vessel under inert atmosphere, followed by the controlled addition of reagents to manage the exothermic nature of the phosphorylation. Operators must monitor the temperature closely during the formation of Compound II to prevent thermal runaway, which could degrade the sensitive carbapenem nucleus. Once the phosphorylation is complete, the addition of the thiol side chain must be performed smoothly to maintain the homogeneity of the reaction mixture. The subsequent crystallization and filtration steps are designed to maximize recovery while minimizing solvent retention in the cake. For a detailed breakdown of the specific molar ratios, solvent volumes, and agitation speeds required for each stage, please refer to the standardized operating procedures below.
- React the carbapenem ketone with phosphoryl chloride in a polar aprotic solvent at low temperature (-60 to 10°C) to generate the enol phosphate intermediate in situ.
- Without isolating the intermediate, directly add the pyrrolidine-thiol side chain compound to the reaction mixture to form the double-protected Panipenem intermediate.
- Perform catalytic hydrogenation on the double-protected intermediate using Pd/C or Pt/C in a buffered aqueous-organic system to yield high-purity Panipenem.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this telescoped synthesis route offers profound benefits for procurement managers and supply chain directors seeking to optimize their sourcing strategies for carbapenem intermediates. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly translates to enhanced supply chain reliability. By removing the isolation step for the unstable enol phosphate intermediate, manufacturers can significantly reduce the number of unit operations required per batch. This reduction in processing steps not only shortens the overall production cycle time but also decreases the dependency on complex equipment setups, thereby lowering the capital expenditure required for facility upgrades. Furthermore, the ability to crystallize the double-protected intermediate directly from the reaction mixture eliminates the need for extensive purification protocols, such as silica gel chromatography, which are often costly and difficult to scale. This operational efficiency ensures a more consistent output of material, mitigating the risks of supply disruptions caused by batch failures or prolonged processing times.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of intermediate isolation and the reduction in solvent consumption. Traditional methods often require large volumes of solvents for extraction and washing during the isolation of unstable intermediates, generating significant waste disposal costs. In contrast, the one-pot nature of this new method minimizes solvent usage and reduces the load on waste treatment facilities. Additionally, the high purity of the crystallized intermediate reduces the need for reprocessing or recycling off-spec material, leading to substantial cost savings in raw material utilization. The avoidance of high-temperature hydrogenation also lowers energy consumption, contributing to a leaner and more cost-effective production model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step syntheses where a failure in any single stage can halt the entire production line. The robustness of this novel route, characterized by its tolerance for variation and simplified workup procedures, significantly de-risks the manufacturing process. The direct crystallization of the intermediate ensures that the material is in a stable, storable form before the final deprotection step, allowing for better inventory management and flexibility in scheduling. This stability is crucial for maintaining a steady flow of materials to downstream formulation partners, ensuring that lead times for high-purity pharmaceutical intermediates are consistently met even during periods of high demand.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, the ability to scale processes with minimal ecological footprint becomes a key differentiator. This synthesis method aligns perfectly with green chemistry principles by reducing the E-factor (mass of waste per mass of product) through solvent minimization and improved atom economy. The use of catalytic hydrogenation with recoverable catalysts like palladium on carbon further supports sustainable practices by avoiding stoichiometric reducing agents that generate heavy metal waste. The scalability of the crystallization step ensures that the process can be seamlessly transferred from pilot plant to commercial scale (100 kgs to 100 MT) without encountering the solubility and mixing issues that often plague batch reactors in traditional carbapenem synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Panipenem synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a clear understanding of the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for large-scale production. The answers highlight the specific advantages in terms of yield, purity, and operational simplicity that distinguish this method from conventional approaches.
Q: How does the new synthesis method improve upon traditional Panipenem production?
A: The novel method eliminates the isolation of the unstable enol phosphate intermediate, allowing for a telescoped 'one-pot' reaction that significantly reduces solvent usage, equipment investment, and processing time compared to conventional multi-step isolation protocols.
Q: What are the purity specifications achievable with this process?
A: Experimental data from the patent indicates that the double-protected intermediate can be crystallized directly from the reaction mixture with purity levels reaching 99%, and the final Panipenem hydrate crystal achieves purity of 97-98% after hydrogenation and crystallization.
Q: Why is the solubility of the intermediate critical in this synthesis?
A: Traditional methods often suffer from poor solubility of benzyl-protected intermediates, leading to 'soup' formation and requiring high-temperature hydrogenation which causes decomposition. This new route utilizes specific solvent systems that allow for direct crystallization and lower-temperature hydrogenation, preserving product integrity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Panipenem Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the global supply of life-saving antibiotics. Our technical team has extensively analyzed the methodologies described in CN101885727A and possesses the expertise to implement this advanced telescoped synthesis at an industrial scale. We understand that transitioning to a new process requires confidence in both the chemistry and the manufacturing capability. Therefore, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to ensure that every batch of Panipenem meets stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing detailed impurity profiling and structural confirmation, guaranteeing that the final product adheres to the highest international pharmacopeial standards.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to explore the full potential of this optimized synthesis route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can drive down costs and secure a sustainable supply chain for Panipenem and other critical carbapenem intermediates, ensuring that patients worldwide have access to effective antibiotic therapies.
