Advanced Synthesis of Panipenem Intermediates via Safe Iodination for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic pathways for carbapenem antibiotics, balancing efficacy with manufacturing safety and cost-efficiency. Patent CN102351860B discloses a groundbreaking synthesis method for Panipenem and its key intermediates, addressing critical bottlenecks in existing technologies. This innovation pivots away from the traditional reliance on hazardous azide reagents and prohibitively expensive transition metal catalysts, introducing a streamlined iodination-based activation strategy. By replacing p-nitrobenzyl protecting groups with standard benzyl moieties and utilizing mild potassium carbonate systems for cyclization, this methodology offers a superior profile for industrial application. For R&D directors and supply chain leaders, this represents a pivotal opportunity to optimize the production of high-purity carbapenem intermediates while mitigating regulatory and safety risks associated with energetic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Panipenem has been plagued by significant safety and economic hurdles inherent to prior art methodologies. Conventional Route One relies heavily on iodobenzene diacetate, a reagent whose preparation necessitates substantial quantities of benzene, posing severe environmental hazards and operator health risks due to benzene's carcinogenicity. Furthermore, Conventional Route Two, while avoiding some benzene usage, introduces high-danger azido reagents such as p-n-C12H25C6H4SO2N3, which are notoriously unstable and unsuitable for large-scale amplification due to explosion risks. Additionally, both traditional methods frequently employ expensive trifluoromethanesulfonic acid tert-butyldimethyl silyl ester (TBSOTf) and costly rhodium acetate catalysts, drastically inflating the cost of goods sold (COGS) and complicating the supply chain with reliance on precious metals.
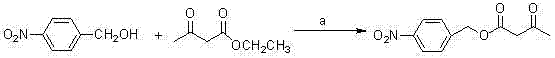
The Novel Approach
The novel approach detailed in the patent fundamentally re-engineers the synthetic logic by activating the methylene group through a safe and efficient iodization method. This strategy completely bypasses the need for high-risk azides or high-cost diphenyl iodoacetic acid derivatives. A key innovation lies in the substitution of p-nitrobenzyl alcohol and p-nitrobenzyl chloroformate with significantly cheaper and more accessible benzyl alcohol and benzyl chloroformate. This switch not only lowers raw material costs but also simplifies the deprotection steps later in the sequence. The process utilizes relatively cheap and gentle anhydrous potassium carbonate in a dimethyl sulfoxide system for cyclization, avoiding the need for expensive acetic acid rhodium reagents. This results in a reaction profile that is both environmentally friendlier and economically superior for commercial manufacturing.
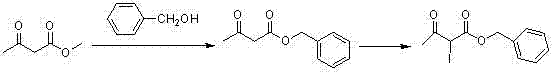
Mechanistic Insights into Iodination-Mediated Methylene Activation
The core mechanistic breakthrough of this synthesis lies in the strategic activation of the methylene position using anhydrous sodium iodide and trimethylchlorosilane. In traditional diazo-transfer methods, the generation of reactive intermediates often requires harsh conditions or unstable precursors. In contrast, this iodination protocol generates a highly reactive iodide intermediate under mild conditions, which subsequently couples efficiently with the beta-lactam fragment. The use of anhydrous zinc chloride as a Lewis acid promoter facilitates the coupling reaction with high stereoselectivity, ensuring the preservation of the critical chiral centers required for biological activity. This mechanism avoids the formation of toxic byproducts associated with azide decomposition, leading to a cleaner reaction mixture and simplified downstream purification processes.
Impurity control is inherently enhanced by the stability of the reagents employed. The replacement of the p-nitrobenzyl (PNZ) group with the benzyl (Bn) group eliminates the potential for nitro-reduction side reactions that can complicate the impurity profile. Furthermore, the cyclization step utilizing potassium carbonate and DMSO operates under thermodynamic control that favors the formation of the desired bicyclic carbapenem core without the epimerization often seen with stronger bases or transition metal catalysts. The final deprotection and amidation steps are conducted under mild aqueous conditions, preserving the integrity of the sensitive beta-lactam ring. This rigorous control over reaction parameters ensures that the final API intermediate meets stringent purity specifications required for parenteral antibiotic formulations.
How to Synthesize Panipenem Intermediates Efficiently
The synthesis of these critical carbapenem intermediates involves a sequential multi-step process beginning with the condensation of methyl acetoacetate and benzyl alcohol. The procedure advances through iodination, coupling with protected azetidinone, and subsequent cyclization and deprotection stages. Each step is optimized for high yield and operational simplicity, utilizing common industrial solvents like toluene, acetonitrile, and ethyl acetate. The detailed standardized synthesis steps, including specific molar ratios and temperature controls for each transformation, are outlined in the technical guide below to ensure reproducibility and quality consistency.
- Activation of methylene group in methyl acetoacetate derivatives using anhydrous sodium iodide and trimethylchlorosilane.
- Coupling with beta-lactam fragments using zinc chloride catalysis followed by cyclization with potassium carbonate.
- Final deprotection and amidation to yield the target carbapenem structure without hazardous azide reagents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, this patented methodology offers transformative advantages by decoupling production from volatile raw material markets and hazardous logistics. The elimination of rhodium catalysts removes a major cost driver and supply bottleneck, as precious metal prices are subject to significant geopolitical fluctuation. Moreover, the shift away from explosive azide reagents drastically reduces insurance premiums and safety compliance costs associated with storage and transport. The use of benzyl alcohol instead of p-nitrobenzyl alcohol leverages a commodity chemical supply chain that is robust, globally available, and significantly less expensive, ensuring long-term supply continuity even during market disruptions.
- Cost Reduction in Manufacturing: The substitution of expensive reagents like diphenyl iodoacetic acid and rhodium acetate with inexpensive anhydrous sodium iodide and potassium carbonate leads to substantial raw material cost savings. Additionally, the use of trimethylchlorosilane instead of bulky silylating agents like TBSOTf further reduces reagent expenses. The simplified workup procedures, which avoid complex heavy metal scavenging steps required for rhodium removal, lower processing costs and waste treatment expenses, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: By relying on widely available commodity chemicals such as benzyl alcohol and sodium iodide, the supply chain becomes far more resilient to shortages. Traditional routes dependent on specialized nitro-aromatics or custom-synthesized azides face higher risks of vendor lock-in and delivery delays. This novel route allows for multi-sourcing of key starting materials, ensuring that production schedules remain uninterrupted and lead times for high-purity pharmaceutical intermediates are minimized.
- Scalability and Environmental Compliance: The absence of high-energy azides makes this process inherently safer for scale-up from pilot plant to multi-ton commercial production. The reduced use of toxic benzene and the elimination of heavy metal catalysts simplify wastewater treatment and align with increasingly strict global environmental regulations. This green chemistry profile facilitates faster regulatory approval for manufacturing sites and reduces the environmental footprint of the facility, supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, cost, and scalability. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs.
Q: Why is this synthesis method safer than conventional Panipenem routes?
A: This method eliminates the use of high-danger azido compounds (like p-n-C12H25C6H4SO2N3) and expensive rhodium catalysts, replacing them with stable iodide reagents and mild base systems.
Q: How does this route reduce manufacturing costs?
A: Costs are reduced by substituting expensive diphenyl iodoacetic acid and rhodium acetate with cheap anhydrous sodium iodide, and by using benzyl alcohol instead of costly p-nitrobenzyl alcohol.
Q: Is this process suitable for large-scale production?
A: Yes, the avoidance of explosive azides and the use of common solvents like toluene and acetonitrile make the process highly scalable and compliant with industrial safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Panipenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global antibiotic market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. We are committed to delivering high-purity Panipenem intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex organic syntheses with the highest standards of safety and quality assurance.
We invite you to collaborate with us to leverage this cost-effective and safe synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can drive value and security for your pharmaceutical projects.
