Advanced Catalytic Oxidation Route for Heliotropine: Technical Upgrade and Commercial Scalability
The pharmaceutical and fragrance industries constantly seek robust synthetic pathways that balance high purity with economic efficiency, and Patent CN1871230A presents a significant breakthrough in the production of heliotropine and its derivatives. This intellectual property discloses a novel, high-yield process for synthesizing compounds of formula (IV), which serve as critical building blocks in the manufacture of fine fragrances and active pharmaceutical ingredients. The technology addresses long-standing inefficiencies in traditional aldehyde synthesis by introducing a streamlined sequence that bypasses the need for rigorous intermediate purification, thereby enhancing overall process throughput. By leveraging a specific chloromethyl-to-acetate-to-alcohol transformation followed by catalytic oxidation, the method achieves superior selectivity and yield compared to historical benchmarks. For R&D directors and procurement specialists, understanding this methodology is essential for evaluating potential supply chain optimizations and cost reduction strategies in fine chemical manufacturing. The following analysis dissects the technical merits and commercial implications of this patented route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-methylenedioxybenzaldehyde, commonly known as heliotropine or piperonal, has relied on several established but flawed methodologies that struggle with specificity and environmental impact. Traditional routes often commence with 1,2-methylenedioxybenzene, employing reagents such as gaseous HCl, zinc chloride, and formaldehyde to generate piperonyl chloride, which is subsequently reacted with hexamethylenetetramine in the Sommelet reaction. This classical approach is fraught with difficulties, including the generation of substantial waste streams and the requirement for complex hydrolysis steps that often result in suboptimal yields. Furthermore, alternative transposition processes like the Vilsmeier-Haack reaction necessitate the use of phosphorus oxychloride and N-alkylformanilides, introducing corrosive hazards and demanding stringent safety protocols that escalate operational costs. Another known pathway involves the oxidative decarboxylation of mandelic acid derivatives using nitric acid, a method that poses significant safety risks due to the exothermic nature of nitration and the handling of strong oxidizers. Collectively, these conventional techniques are characterized by non-specific side reactions, low atom economy, and the burdensome necessity for multiple intermediate purification stages, which collectively erode profit margins and extend production lead times.
The Novel Approach
In stark contrast to these legacy methods, the process outlined in CN1871230A introduces a highly specific and efficient three-step sequence that fundamentally alters the production landscape for these aldehyde derivatives. The innovation lies in the strategic conversion of a chloromethyl derivative (I) into an acetyl derivative (II) using an alkaline acetate, rather than attempting direct hydrolysis which is prone to failure. This acetylation step serves as a protective and activating maneuver, allowing for a subsequent hydrolysis to the alcohol (III) with exceptional efficiency, achieving yields between 90% and 96% compared to the less than 80% ceiling of direct NaOH hydrolysis. 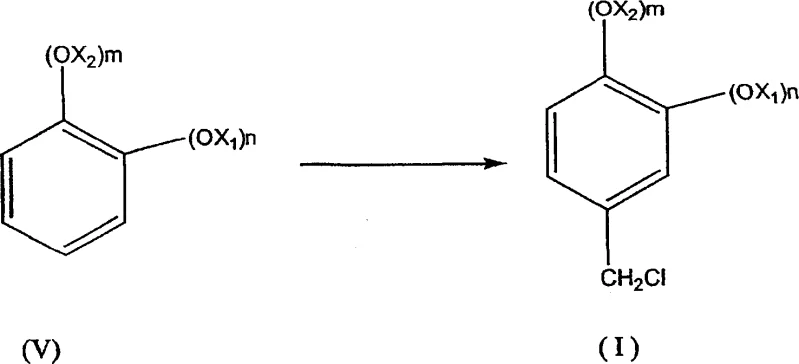 This improvement is critical because it minimizes the formation of by-products that could otherwise poison the catalysts in the final oxidation stage. The final transformation involves the catalytic oxidation of the alcohol (III) to the desired aldehyde (IV) using air or oxygen in the presence of supported metal catalysts, a green chemistry approach that eliminates the need for stoichiometric toxic oxidants. By enabling the use of crude intermediates without isolation, this novel approach drastically simplifies the operational workflow, reduces solvent consumption, and ensures a consistent supply of high-purity end products suitable for sensitive applications in perfumery and medicine.
This improvement is critical because it minimizes the formation of by-products that could otherwise poison the catalysts in the final oxidation stage. The final transformation involves the catalytic oxidation of the alcohol (III) to the desired aldehyde (IV) using air or oxygen in the presence of supported metal catalysts, a green chemistry approach that eliminates the need for stoichiometric toxic oxidants. By enabling the use of crude intermediates without isolation, this novel approach drastically simplifies the operational workflow, reduces solvent consumption, and ensures a consistent supply of high-purity end products suitable for sensitive applications in perfumery and medicine.
Mechanistic Insights into Catalytic Oxidation and Intermediate Stability
The core mechanistic advantage of this synthesis lies in the careful management of functional group interconversions to preserve catalyst activity during the final oxidation step. The initial chloromethylation of the benzodioxole substrate, as depicted in the reaction schemes, generates a reactive benzylic chloride which is inherently unstable towards direct nucleophilic attack by strong bases like sodium hydroxide due to competing elimination or polymerization side reactions. By intercepting this intermediate with acetate ions in a biphasic system, the process forms a stable ester linkage that withstands the reaction conditions better than the free alcohol would during the initial substitution phase. 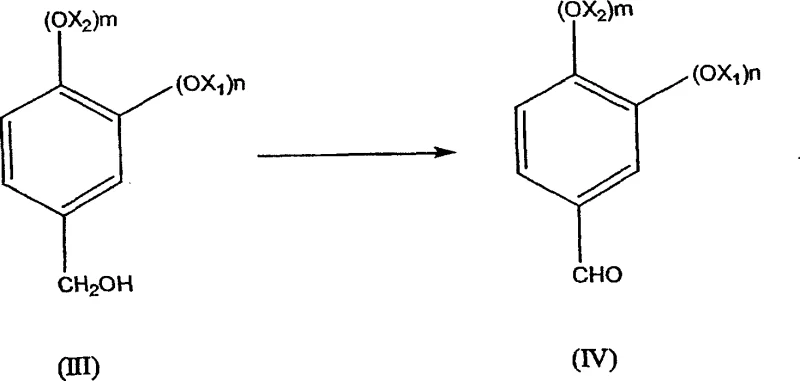 Following the formation of the acetate, the hydrolysis step is conducted under controlled basic conditions with phase transfer catalysts, ensuring complete conversion to the benzylic alcohol while maintaining the integrity of the methylenedioxy ring. The subsequent oxidation mechanism relies on heterogeneous catalysis, typically employing Ruthenium or Platinum supported on carbon, which facilitates the dehydrogenation of the alcohol in the presence of molecular oxygen. The basic environment provided by sodium hydroxide is crucial here, as it enhances the reaction rate by facilitating the formation of alkoxide species that are more readily oxidized, yet the pH is managed to prevent aldol condensation of the product aldehyde. This delicate balance ensures that the catalyst surface remains active and free from fouling by acidic by-products or polymeric tars, which are common failure modes in less optimized oxidation processes.
Following the formation of the acetate, the hydrolysis step is conducted under controlled basic conditions with phase transfer catalysts, ensuring complete conversion to the benzylic alcohol while maintaining the integrity of the methylenedioxy ring. The subsequent oxidation mechanism relies on heterogeneous catalysis, typically employing Ruthenium or Platinum supported on carbon, which facilitates the dehydrogenation of the alcohol in the presence of molecular oxygen. The basic environment provided by sodium hydroxide is crucial here, as it enhances the reaction rate by facilitating the formation of alkoxide species that are more readily oxidized, yet the pH is managed to prevent aldol condensation of the product aldehyde. This delicate balance ensures that the catalyst surface remains active and free from fouling by acidic by-products or polymeric tars, which are common failure modes in less optimized oxidation processes.
Furthermore, the impurity control mechanism is intrinsically built into the choice of the acetate intermediate pathway. Direct hydrolysis of chloromethyl derivatives often leads to the formation of ether-linked dimers or polymeric residues that are difficult to separate and can irreversibly bind to noble metal catalysts, rendering them inactive. The acetate route effectively suppresses these side reactions by modulating the nucleophilicity of the reaction medium during the substitution phase. The use of a biphasic toluene-water system further aids in impurity management by partitioning organic soluble by-products away from the aqueous catalytic phase where the oxidation occurs. This phase separation capability allows for the continuous removal of water generated during oxidation and prevents the accumulation of inorganic salts that could precipitate and foul the reactor internals. Consequently, the process maintains high selectivity for the aldehyde functionality, minimizing the over-oxidation to carboxylic acids which is a prevalent issue in aerobic oxidations of benzylic alcohols. The result is a crude product profile that is exceptionally clean, requiring only simple distillation and crystallization to achieve pharmaceutical-grade purity levels exceeding 99%.
How to Synthesize Heliotropine Efficiently
The synthesis of heliotropine via this patented route offers a practical blueprint for manufacturers aiming to optimize their production lines for both yield and safety. The process begins with the chloromethylation of 1,3-benzodioxole using paraformaldehyde and hydrochloric acid in a solvent like toluene, conducted at mild temperatures to ensure high selectivity for the mono-chloromethylated product. Following the separation of the aqueous acid layer, the organic phase containing the crude chloromethyl derivative is not purified but is instead directly introduced into a reactor containing aqueous sodium acetate. This telescoping of steps eliminates the need for drying or distilling the intermediate, saving significant energy and time. The mixture is heated to facilitate the substitution reaction, forming the acetate ester in situ. Subsequently, sodium hydroxide and a quaternary ammonium salt are added to the same vessel to effect hydrolysis, converting the ester to the corresponding alcohol with high conversion rates. Finally, the alcohol solution is subjected to catalytic oxidation using air sparging and a supported platinum or ruthenium catalyst under basic conditions. This sequence exemplifies modern process intensification, where multiple chemical transformations are integrated into a minimal number of unit operations to maximize efficiency.
- Perform chloromethylation of the benzodioxole substrate using formaldehyde and hydrochloric acid in an organic solvent to form the chloromethyl derivative.
- React the chloromethyl derivative with an alkaline acetate in a biphasic system to generate the acetyl ester intermediate without isolation.
- Hydrolyze the ester intermediate using sodium hydroxide and a phase transfer catalyst to form the alcohol, followed by catalytic oxidation with air to yield the final aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic advantages that extend beyond mere chemical yield improvements. The elimination of intermediate isolation steps fundamentally alters the cost structure of production by reducing the consumption of solvents, energy, and labor associated with distillation and drying operations. This streamlining of the workflow directly contributes to cost reduction in fine chemical manufacturing, allowing suppliers to offer more competitive pricing without compromising on quality standards. Moreover, the reliance on air as the terminal oxidant replaces expensive and hazardous stoichiometric oxidizing agents, further driving down raw material costs and simplifying waste disposal logistics. The robustness of the catalytic system ensures consistent batch-to-batch reproducibility, which is critical for maintaining reliable supply chains for downstream customers in the fragrance and pharmaceutical sectors who demand strict adherence to specifications.
- Cost Reduction in Manufacturing: The process achieves significant economic savings by removing the necessity for purifying the chloromethyl and acetyl intermediates, which traditionally account for a large portion of processing time and solvent usage. By utilizing crude reaction mixtures directly in subsequent steps, the plant throughput is increased, and the capital expenditure on separation equipment is minimized. Additionally, the use of heterogeneous catalysts that can potentially be recovered and reused adds another layer of cost efficiency, reducing the dependency on precious metal inputs. The overall simplification of the process flow means lower utility costs for heating and cooling, as well as reduced waste treatment expenses due to the absence of heavy metal salts or corrosive acid wastes typical of older methods.
- Enhanced Supply Chain Reliability: The use of commercially available starting materials such as 1,3-benzodioxole and paraformaldehyde ensures that the supply chain is not vulnerable to the bottlenecks associated with exotic or custom-synthesized reagents. The reaction conditions are mild and operate at atmospheric pressure with air, reducing the risk of unplanned shutdowns due to equipment failure or safety incidents related to high-pressure hydrogenation or hazardous oxidants. This operational stability guarantees a steady output of high-purity heliotropine, enabling procurement teams to secure long-term contracts with confidence. The ability to scale this process from pilot batches to multi-ton production without significant re-engineering further strengthens supply continuity, making it an ideal candidate for meeting growing global demand.
- Scalability and Environmental Compliance: From an environmental perspective, this route aligns with green chemistry principles by utilizing molecular oxygen from air, producing water as the only by-product of the oxidation step, and avoiding the generation of chlorinated waste streams associated with Vilsmeier-type reagents. The biphasic nature of the reaction facilitates easier product separation and solvent recycling, minimizing the environmental footprint of the manufacturing facility. Regulatory compliance is simplified as the process avoids the use of restricted substances and generates less hazardous waste, reducing the administrative burden and liability for the manufacturer. This sustainability profile is increasingly becoming a deciding factor for multinational corporations seeking responsible suppliers, thereby opening up new market opportunities for producers who adopt this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these details is crucial for assessing the feasibility of integrating this process into existing manufacturing infrastructures.
Q: What is the primary advantage of the acetate intermediate route over direct hydrolysis?
A: The acetate intermediate route significantly improves the yield of the alcohol precursor to over 90%, whereas direct hydrolysis of the chloromethyl derivative with NaOH typically fails to exceed 80% yield and generates by-products that interfere with subsequent oxidation catalysts.
Q: Which catalysts are suitable for the final oxidation step in this process?
A: The process utilizes heterogeneous catalysts such as Ruthenium, Platinum, Palladium, or Gold supported on carbon or alumina. Raney Nickel is also effective, allowing for the use of air or oxygen as a green oxidant in a basic aqueous-organic medium.
Q: Can the intermediates be isolated during the synthesis?
A: No, the process is designed to avoid intermediate purification. The crude organic phase from the chloromethylation step can be used directly for acetylation, and the hydrolysis can proceed in the same biphasic system, streamlining the workflow and reducing solvent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heliotropine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic technologies to meet the evolving demands of the global fine chemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1871230A are fully realized in practical, large-scale operations. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of heliotropine or related aldehyde intermediate meets the exacting standards required by the fragrance and pharmaceutical industries. Our commitment to technical excellence allows us to navigate the complexities of catalytic oxidation and intermediate handling with precision, delivering products that consistently perform in downstream applications.
We invite you to collaborate with us to leverage these technological advancements for your specific supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can enhance your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in process chemistry can become a strategic asset for your organization.
