Advanced Catalytic Oxidation Route for Commercial Scale Heliotropine Production
The global demand for high-purity aromatic aldehydes continues to drive innovation in fine chemical synthesis, particularly for key compounds like heliotropine, widely known as piperonal. Patent CN100528858C introduces a transformative manufacturing methodology that addresses long-standing inefficiencies in producing 3,4-methylenedioxybenzaldehyde derivatives. This technical disclosure outlines a robust, three-stage sequence that begins with a chloromethyl derivative and proceeds through an acetate intermediate to the final aldehyde, bypassing the need for rigorous intermediate isolation. For R&D Directors and Procurement Managers seeking a reliable flavor & fragrance intermediate supplier, this process represents a significant leap forward in operational efficiency. By leveraging air oxidation with supported noble metal catalysts, the technology minimizes the reliance on stoichiometric oxidants that generate excessive waste streams. The strategic implementation of this pathway allows for a seamless transition from laboratory scale to industrial production, ensuring that supply chain stakeholders can depend on consistent quality and availability. The following analysis dissects the mechanistic advantages and commercial implications of this patented approach, highlighting its potential to redefine cost structures in the specialty chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of heliotropine has been plagued by processes that are either environmentally burdensome or economically inefficient due to low selectivity. Traditional routes, such as the Sommelet reaction, involve the treatment of piperonyl chloride with hexamethylenetetramine, a method that frequently results in complex reaction mixtures requiring extensive downstream purification. Furthermore, older methodologies often rely on harsh oxidizing agents like nitric acid or chromium-based reagents, which pose significant safety hazards and create difficult-to-manage waste disposal challenges for environmental compliance officers. The Vilsmeier-Haack reaction, while effective for formylation, often lacks the specificity required for high-volume manufacturing, leading to variable yields that complicate inventory planning for supply chain heads. These conventional methods typically necessitate the isolation and drying of intermediates, adding multiple unit operations that increase energy consumption and capital expenditure. Consequently, manufacturers utilizing these legacy technologies face higher production costs and longer lead times, making it difficult to compete in a market that demands both agility and sustainability. The accumulation of by-products in these older routes also complicates the impurity profile, requiring additional recrystallization steps that further erode overall process yield.
The Novel Approach
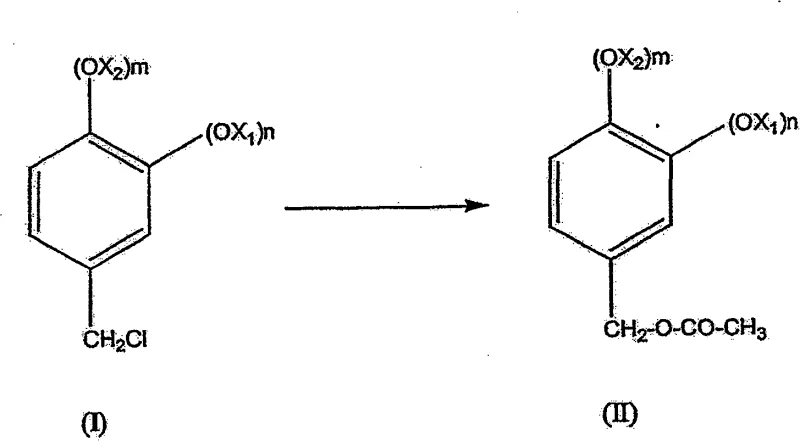
In stark contrast, the methodology described in CN100528858C offers a streamlined alternative that prioritizes atom economy and process intensification. By converting the chloromethyl starting material directly into an acetate ester using alkaline acetates, the process establishes a stable intermediate that is less prone to side reactions compared to direct hydrolysis attempts. This acetate pathway serves as a protective strategy, ensuring that the subsequent hydrolysis to the alcohol occurs with high fidelity and minimal formation of tars or polymeric by-products. The true innovation lies in the telescoping capability, where the crude alcohol solution can be fed directly into the oxidation reactor without intermediate workup, drastically reducing solvent usage and processing time. This approach not only simplifies the equipment footprint required for production but also enhances the safety profile by avoiding the handling of unstable intermediates in large quantities. For procurement teams, this translates to a more predictable manufacturing cycle, as the reduction in unit operations decreases the probability of batch failures or delays. The use of molecular oxygen or air as the terminal oxidant further aligns with green chemistry principles, offering a sustainable advantage that is increasingly valued by downstream pharmaceutical and fragrance clients.
Mechanistic Insights into Catalytic Air Oxidation
The core of this synthetic strategy relies on a sophisticated understanding of phase-transfer catalysis and heterogeneous oxidation mechanisms. In the initial transformation, the chloromethyl derivative reacts with sodium acetate in a biphasic system, where the acetate anion displaces the chloride to form the ester. This step is critical because direct hydrolysis of the chloromethyl group with strong bases often leads to elimination reactions or resinification, which the acetate route effectively suppresses. Following ester formation, the introduction of a phase-transfer catalyst, such as tetrabutylammonium chloride, facilitates the migration of hydroxide ions into the organic phase, enabling rapid hydrolysis of the acetate to the corresponding benzyl alcohol. This mechanistic control ensures that the alcohol is generated in situ with high purity, setting the stage for the final oxidation step. The elimination of transition metal contaminants from the reaction mixture is inherently managed by the choice of reagents, which simplifies the purification of the final active pharmaceutical ingredient or fragrance compound.
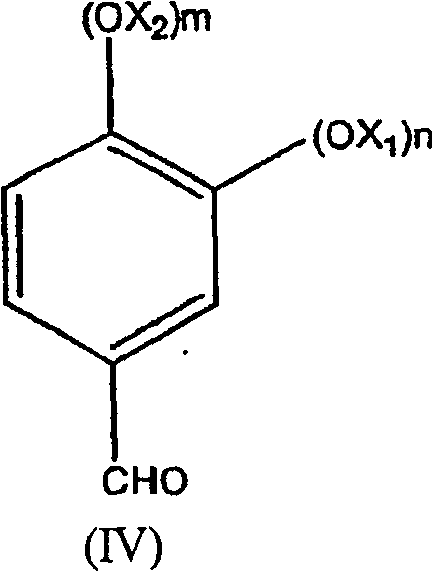
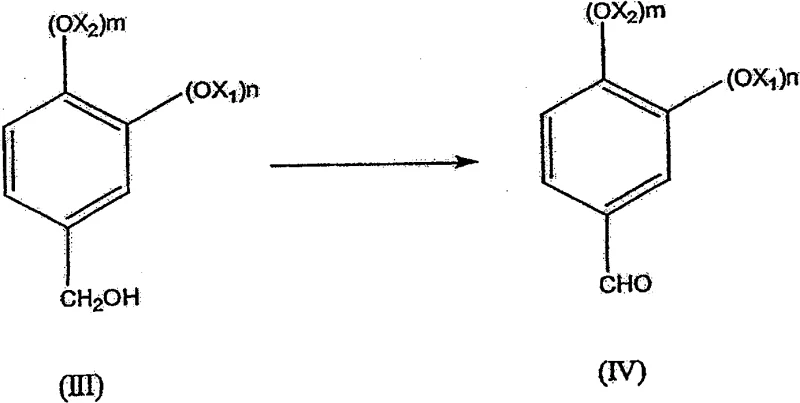
The final oxidation stage utilizes supported platinum or ruthenium catalysts to activate molecular oxygen for the conversion of the benzyl alcohol to the aldehyde. This catalytic cycle operates under basic conditions, where the alkoxide species is more readily oxidized than the neutral alcohol, enhancing the reaction rate significantly. The mechanism involves the adsorption of the alcohol onto the metal surface, followed by dehydrogenation and subsequent reaction with activated oxygen species to release the aldehyde and water. A key advantage for R&D teams is the high selectivity of this system, which prevents the over-oxidation of the aldehyde to the corresponding carboxylic acid, a common issue in non-catalytic oxidation processes. The catalyst stability allows for potential recycling or extended use, contributing to the overall economic viability of the process. By maintaining strict control over pH and oxygen flow rates, the process ensures a consistent impurity profile, which is essential for meeting the stringent specifications required by regulatory bodies in the food and pharmaceutical industries.
How to Synthesize Heliotropine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize the benefits of the telescoped design. The process begins with the preparation of the chloromethyl precursor, which is then reacted with aqueous sodium acetate at elevated temperatures to ensure complete conversion to the acetate ester. Following this, the reaction mixture is treated with sodium hydroxide and a phase-transfer catalyst to effect hydrolysis, generating the alcohol intermediate in the same vessel. The final step involves the addition of the supported metal catalyst and the controlled introduction of air or oxygen to drive the oxidation to completion. Detailed standardized synthesis steps see the guide below.
- Treat chloromethyl derivative with alkaline acetate to form the acetate intermediate without isolation.
- Hydrolyze the acetate intermediate in a biphasic system using sodium hydroxide and a phase transfer catalyst.
- Oxidize the resulting alcohol using air and a supported platinum or ruthenium catalyst to yield the final aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial value propositions for stakeholders focused on cost optimization and supply security. The elimination of intermediate isolation steps significantly reduces the consumption of solvents and energy, leading to a lower overall cost of goods sold without compromising on quality. For procurement managers, this efficiency gain means that pricing structures can be more competitive, allowing for better margin management in volatile raw material markets. The reliance on air as an oxidant removes the need for purchasing and storing hazardous stoichiometric oxidizing agents, thereby reducing insurance costs and regulatory compliance burdens associated with dangerous goods. Furthermore, the robustness of the catalytic system ensures high batch-to-batch consistency, which is critical for maintaining long-term contracts with multinational corporations that require strict adherence to quality agreements.
- Cost Reduction in Manufacturing: The telescoped nature of the synthesis eliminates the need for filtration, drying, and re-dissolution of intermediates, which are traditionally labor and energy-intensive operations. By removing these unit operations, the facility can achieve higher throughput with the existing infrastructure, effectively increasing capacity without capital investment. The reduction in waste generation also lowers disposal costs, contributing to a leaner manufacturing model that aligns with modern sustainability goals. Additionally, the high yield of the oxidation step minimizes the loss of valuable starting materials, ensuring that raw material costs are maximized in the final product output.
- Enhanced Supply Chain Reliability: The use of readily available starting materials and common reagents such as sodium acetate and sodium hydroxide mitigates the risk of supply disruptions caused by specialty chemical shortages. The process is designed to be scalable, allowing manufacturers to ramp up production quickly in response to market demand spikes without extensive process re-validation. This flexibility is crucial for supply chain heads who must navigate the complexities of global logistics and ensure continuous availability for downstream customers. The stability of the intermediates also allows for potential storage strategies that can buffer against short-term fluctuations in production schedules.
- Scalability and Environmental Compliance: The substitution of toxic oxidants with air significantly improves the environmental footprint of the manufacturing process, facilitating easier permitting and regulatory approval in strict jurisdictions. The aqueous workup and reduced solvent usage simplify wastewater treatment requirements, lowering the operational burden on environmental health and safety teams. This green chemistry approach enhances the corporate social responsibility profile of the manufacturer, making the supply chain more attractive to end-users who prioritize sustainable sourcing. The ability to run the process in standard stainless steel reactors without the need for exotic metallurgy further supports easy scale-up from pilot plant to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications to ensure accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of adopting this route for large-scale manufacturing.
Q: How does this process improve upon the Sommelet reaction for heliotropine production?
A: Unlike the Sommelet reaction which often suffers from low specificity and requires complex intermediate purification, this novel route utilizes a direct acetate intermediate pathway that allows for telescoped processing, significantly reducing waste and operational time.
Q: What catalysts are suitable for the final oxidation step in this synthesis?
A: The patent specifies the use of supported noble metal catalysts, particularly ruthenium or platinum on carbon, which facilitate efficient air oxidation under basic conditions, ensuring high conversion rates without over-oxidation to carboxylic acids.
Q: Is intermediate purification required between the hydrolysis and oxidation steps?
A: No, a key advantage of this methodology is the ability to carry forward the crude alcohol product directly into the oxidation stage, eliminating the need for energy-intensive separation and drying processes typically associated with batch synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heliotropine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical innovation, leveraging advanced processes like the one detailed in CN100528858C to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of heliotropine meets the highest industry standards for fragrance and pharmaceutical applications. Our commitment to process excellence allows us to offer a stable supply of high-quality intermediates that support your product development and manufacturing goals.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through our advanced technical capabilities. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can enhance your operational efficiency. Partner with us to secure a reliable source of premium fine chemicals that drive your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
