Advanced Synthesis of 1,3,6-Trialkylhexahydro-1,3,6-triazocin-2-one for Industrial Solvent Applications
Advanced Synthesis of 1,3,6-Trialkylhexahydro-1,3,6-triazocin-2-one for Industrial Solvent Applications
The chemical industry is constantly seeking alternatives to traditional aprotic polar solvents that offer improved safety profiles without compromising performance. Patent CN1111624A introduces a groundbreaking class of compounds known as 1,3,6-trialkylhexahydro-1,3,6-triazocin-2-ones, which represent a significant evolution in solvent technology. These novel cyclic urea derivatives are designed to overcome the critical limitations of established solvents like N-methyl-2-pyrrolidone (NMP) and 1,3-dimethyl-2-imidazolinone (DMI). By leveraging a unique eight-membered ring structure containing three nitrogen atoms, this technology delivers exceptional solvency power for high-performance polymers while mitigating the toxicological risks associated with legacy chemicals. For R&D teams and procurement specialists alike, understanding the synthesis and application of these molecules is key to future-proofing supply chains and enhancing product safety.

As illustrated in the structural diagram above, the core architecture features a robust heterocyclic framework where the alkyl substituents (R) can be tuned from C1 to C8 to optimize physical properties such as viscosity and boiling point. This structural flexibility allows manufacturers to tailor the solvent for specific applications ranging from pharmaceutical intermediates to advanced polymer processing. The patent highlights that these compounds are not merely incremental improvements but represent a distinct chemical class capable of functioning as both reaction media and functional intermediates for surfactants and detergents. For a reliable specialty chemical supplier, mastering the production of this scaffold opens doors to high-value markets demanding stringent purity and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the fine chemical and polymer industries have relied heavily on five-membered cyclic amides and ureas, specifically N-methyl-2-pyrrolidone and 1,3-dimethyl-2-imidazolinone. While effective, these conventional solvents suffer from inherent drawbacks that pose significant operational and regulatory challenges. NMP, for instance, has faced increasing scrutiny due to its classification as a reproductive toxin and its potential to cause chromosomal aberrations, leading to stricter exposure limits and handling requirements in manufacturing facilities. Furthermore, 1,3-dimethyl-2-imidazolinone exhibits poor oxidation resistance at elevated temperatures, limiting its utility in high-heat processes where solvent degradation can lead to product contamination or equipment fouling. These stability issues often necessitate complex purification steps or the use of stabilizers, driving up the total cost of ownership for end-users.
The Novel Approach
The methodology outlined in the patent data proposes a transformative shift by expanding the ring size to an eight-membered triazocine system. This structural expansion fundamentally alters the electronic and steric environment of the molecule, resulting in enhanced thermal stability and reduced toxicity. The synthesis begins with readily available precursors, specifically N,N',N''-trialkyldiethylenetriamines, which are reacted with urea, phosgene, or carbon dioxide.  As shown in the precursor structure above, the linear triamine backbone provides the necessary nitrogen spacing to facilitate the formation of the larger ring upon cyclization. This approach avoids the use of expensive or hazardous catalysts often required in traditional heterocycle synthesis, relying instead on thermal activation to drive the reaction to completion. The result is a cleaner process with fewer by-products, directly addressing the purity concerns of R&D directors working on sensitive API intermediates.
As shown in the precursor structure above, the linear triamine backbone provides the necessary nitrogen spacing to facilitate the formation of the larger ring upon cyclization. This approach avoids the use of expensive or hazardous catalysts often required in traditional heterocycle synthesis, relying instead on thermal activation to drive the reaction to completion. The result is a cleaner process with fewer by-products, directly addressing the purity concerns of R&D directors working on sensitive API intermediates.
Mechanistic Insights into Thermal Cyclization and Urea Condensation
The core of this synthesis lies in a sophisticated two-stage thermal process that converts the linear triamine into the cyclic urea without the need for external coupling agents. In the first stage, the N,N',N''-trialkyldiethylenetriamine reacts with urea at moderate temperatures, typically between 100-155°C. During this phase, a urea intermediate is formed quantitatively, a step that can be precisely monitored by measuring the evolution of ammonia gas. This real-time feedback mechanism allows for tight process control, ensuring that the stoichiometry is optimized before the critical cyclization step begins. The avoidance of strong bases or acids in this step minimizes the formation of salt by-products, simplifying the downstream workup and reducing the load on wastewater treatment systems.
The second stage involves a high-temperature decomposition and cyclization event, where the reaction temperature is ramped to between 200-260°C. At these elevated temperatures, the urea intermediate undergoes intramolecular condensation, closing the ring to form the stable triazocin-2-one structure. 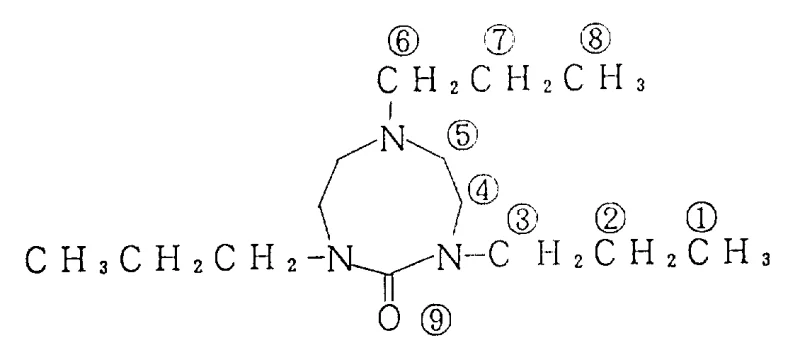 The structural integrity of the final product is confirmed through rigorous spectroscopic analysis, as detailed in the annotated structure above which maps specific proton and carbon environments. This mechanistic pathway ensures that the final solvent possesses a defined impurity profile, free from the heavy metal residues often associated with transition-metal catalyzed routes. For quality assurance teams, this predictable chemistry translates to consistent batch-to-batch reproducibility, a critical factor when scaling up from laboratory grams to commercial metric tons.
The structural integrity of the final product is confirmed through rigorous spectroscopic analysis, as detailed in the annotated structure above which maps specific proton and carbon environments. This mechanistic pathway ensures that the final solvent possesses a defined impurity profile, free from the heavy metal residues often associated with transition-metal catalyzed routes. For quality assurance teams, this predictable chemistry translates to consistent batch-to-batch reproducibility, a critical factor when scaling up from laboratory grams to commercial metric tons.
How to Synthesize 1,3,6-Trialkylhexahydro-1,3,6-triazocin-2-one Efficiently
Implementing this synthesis route requires careful attention to thermal management and stoichiometric ratios to maximize yield and minimize the formation of oligomeric by-products. The process is uniquely suited for continuous flow or large-batch reactor systems due to its solvent-free nature and reliance on thermal energy rather than chemical reagents. Operators must ensure precise temperature control during the transition from the intermediate formation phase to the cyclization phase to prevent urea decomposition before ring closure occurs. The following guide outlines the standardized operational parameters derived from the patent examples to assist technical teams in replicating this high-efficiency process.
- Prepare N,N',N''-trialkyldiethylenetriamine by reacting alkylamine with 1,2-dihaloethane at 80-150°C, followed by neutralization and distillation.
- React the triamine with urea at 100-155°C to form a urea intermediate, monitoring ammonia release to determine endpoint.
- Raise temperature to 200-260°C to induce decomposition and cyclization, then purify the final product via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this novel solvent technology offers compelling advantages that extend beyond mere chemical performance. The ability to produce high-purity solvents using a solvent-free methodology represents a paradigm shift in manufacturing economics, eliminating the need for vast quantities of auxiliary solvents that require recovery and disposal. This reduction in process complexity directly correlates to lower capital expenditure on distillation columns and solvent storage tanks, providing a clear path for cost reduction in specialty chemical manufacturing. Furthermore, the raw materials required, such as alkylamines and urea, are commodity chemicals with robust global supply chains, ensuring long-term availability and price stability for procurement managers.
- Cost Reduction in Manufacturing: The elimination of external solvents in the reaction mixture significantly reduces the energy burden associated with solvent recovery and recycling. By operating under solvent-free conditions, the volumetric efficiency of the reactors is maximized, allowing for greater throughput per batch without increasing facility footprint. Additionally, the unreacted triamine precursor can be recovered via distillation and recycled back into the process, further driving down raw material costs and minimizing waste generation. This closed-loop potential creates a sustainable economic model that insulates the supply chain from volatile raw material pricing.
- Enhanced Supply Chain Reliability: The synthesis relies on fundamental petrochemical building blocks that are produced at massive scales globally, reducing the risk of supply disruptions common with exotic reagents. The robustness of the thermal cyclization process means that production is less sensitive to minor fluctuations in reagent quality, ensuring consistent output even when sourcing from multiple vendors. For supply chain heads, this translates to reduced lead times for high-purity solvents and a more resilient inventory strategy that can withstand market shocks.
- Scalability and Environmental Compliance: The high boiling points of the final products allow for processing at atmospheric pressure for many alkyl variants, removing the need for expensive high-pressure reactor vessels. This simplifies the engineering requirements for commercial scale-up of complex solvents, enabling faster deployment of new production lines. Moreover, the absence of heavy metal catalysts and the potential for near-quantitative conversion align perfectly with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental compliance burden on manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,3,6-trialkylhexahydro-1,3,6-triazocin-2-ones. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the technology's capabilities. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this solvent into existing workflows or developing new formulations that require superior thermal stability and safety.
Q: What are the safety advantages of 1,3,6-trialkylhexahydro-1,3,6-triazocin-2-one over NMP?
A: Unlike N-methyl-2-pyrrolidone (NMP), which is associated with chromosomal aberrations and reproductive toxicity, this novel cyclic urea structure demonstrates superior biocompatibility profiles while maintaining excellent solvency for high polymers.
Q: Can this solvent be produced without additional organic solvents?
A: Yes, the patent describes a highly efficient solvent-free protocol where the reactants themselves serve as the medium, significantly reducing downstream separation costs and environmental waste.
Q: What is the thermal stability range for this new solvent class?
A: The compounds exhibit high thermal stability, with boiling points exceeding 250°C for the methyl derivative, making them suitable for high-temperature polymerization reactions where conventional imidazolinones might degrade.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,6-Trialkylhexahydro-1,3,6-triazocin-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, high-performance solvents is a critical priority for the global chemical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN1111624A can be effectively translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of solvent meets the exacting standards required for pharmaceutical and electronic applications. Our commitment to quality ensures that your downstream processes remain uninterrupted by impurities or variability.
We invite you to collaborate with us to explore how this advanced solvent technology can optimize your specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your production volumes. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how partnering with us can drive efficiency and safety in your supply chain.
