Advanced Synthesis and Commercial Scale-up of 1,3,6-Trialkylhexahydro-1,3,6-triazocin-2-one
Introduction to Novel Aprotic Polar Solvents
The chemical industry is constantly seeking alternatives to traditional solvents that balance performance with safety and environmental compliance. Patent CN1058488C, granted in late 2000, introduces a significant breakthrough in this domain with the disclosure of 1,3,6-trialkylhexahydro-1,3,6-triazocin-2-one. This novel class of compounds represents a new generation of aprotic polar solvents designed to overcome the limitations of established chemicals like N-methyl-2-pyrrolidone (NMP) and 1,3-dimethyl-2-imidazolidinone (DMI). The core innovation lies in the unique eight-membered heterocyclic ring structure containing three nitrogen atoms, which imparts exceptional solvency power for a wide range of high polymers including polyamides, polyvinyl chloride, and phenolic resins. For R&D directors and procurement specialists, understanding the synthesis and application potential of this molecule is crucial for developing next-generation formulations that require high thermal stability and low toxicity profiles.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the fine chemical and polymer processing industries have relied heavily on five-membered cyclic ureas and lactams, specifically 1,3-dimethyl-2-imidazolidinone and N-methyl-2-pyrrolidone. While these solvents possess excellent dissolving capabilities, they suffer from critical drawbacks that impact both operational safety and product quality. N-methyl-2-pyrrolidone, for instance, has been flagged for causing chromosomal aberrations, raising severe regulatory and health concerns for large-scale manufacturing environments. Furthermore, 1,3-dimethyl-2-imidazolidinone exhibits poor oxidation resistance at elevated temperatures, limiting its utility in high-temperature polymerization reactions or processes requiring prolonged thermal exposure. These deficiencies necessitate costly containment measures and frequent solvent replacement, driving up the total cost of ownership for chemical manufacturers who depend on reliable, high-purity solvent systems for their production lines.
The Novel Approach
The methodology outlined in the patent data presents a transformative alternative by expanding the ring size to an eight-membered 1,3,6-triazocine structure. This structural modification significantly enhances the thermal stability and oxidation resistance of the solvent compared to its five-membered counterparts. The new compound maintains strong polarity, ensuring it remains an effective solvent for difficult-to-dissolve polymers and organic intermediates. By shifting the molecular architecture, the inventors have created a substance that avoids the specific toxicological pitfalls associated with NMP while delivering superior performance metrics. This approach not only mitigates regulatory risks but also opens new avenues for process optimization where higher operating temperatures were previously prohibited by solvent degradation, thereby offering a strategic advantage for companies looking to future-proof their chemical supply chains.
Mechanistic Insights into Solvent-Free Cyclization
The synthesis of 1,3,6-trialkylhexahydro-1,3,6-triazocin-2-one relies on a sophisticated yet efficient cyclization strategy that minimizes waste and maximizes atom economy. The process begins with the preparation of the key linear precursor, N,N',N''-trialkyldiethylenetriamine, which serves as the nitrogen-rich backbone for the final heterocycle. This intermediate is typically generated through the alkylation of diethylenetriamine derivatives or the reaction of alkylamines with dihaloethanes. The subsequent cyclization step involves reacting this triamine with a carbonyl source such as urea, phosgene, or carbon dioxide. When urea is employed, the reaction proceeds through a distinct two-stage thermal profile. Initially, the mixture is heated to a moderate temperature range to form a urea adduct, followed by a high-temperature decomposition phase that drives the intramolecular ring closure. This mechanism ensures the formation of the stable eight-membered ring without the need for exotic catalysts or harsh reagents that could introduce difficult-to-remove impurities.

Controlling impurities in this synthesis is achieved through precise thermal management and distillation protocols. The patent specifies that the initial reaction with urea should occur between 100°C and 155°C to prevent premature decomposition of the urea itself, which would lead to side reactions and reduced yields. Once the urea intermediate is formed, the temperature is ramped significantly higher, ideally between 210°C and 240°C, to facilitate the elimination of ammonia and the final cyclization. This high-temperature step is critical; if the temperature is too low, the reaction kinetics are insufficient, but if it exceeds 300°C, equipment integrity becomes a concern. The resulting crude product is then purified via distillation, a method that effectively separates the high-boiling target molecule from any unreacted starting materials. Notably, the unreacted N,N',N''-trialkyldiethylenetriamine can be recovered from the initial distillate fractions and recycled back into the process, creating a closed-loop system that drastically reduces raw material consumption and waste generation.
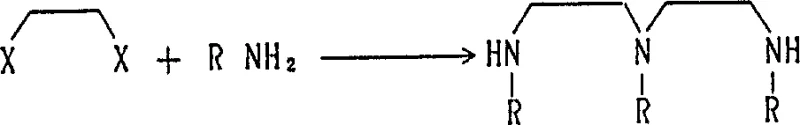
How to Synthesize 1,3,6-Trimethylhexahydro-1,3,6-triazocin-2-one Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal gradients to ensure optimal yield and purity. The patent data provides a clear roadmap for converting simple alkylamines and dihaloethanes into the valuable cyclic urea solvent. The process is particularly advantageous because it can be conducted under solvent-free conditions, eliminating the need for additional volatile organic compounds that would otherwise require removal and disposal. By adhering to the specified molar ratios—typically using a slight excess of the triamine relative to urea—manufacturers can suppress the formation of oligomeric by-products. The following guide outlines the standardized operational steps derived from the patent examples, serving as a foundational protocol for pilot plant trials and commercial production scaling.
- Synthesize N,N',N''-trialkyldiethylenetriamine by reacting alkylamine with 1,2-dihaloethane at 80-150°C, followed by neutralization and distillation.
- React the triamine intermediate with urea at 100-155°C to form a urea intermediate, monitoring ammonia release.
- Raise temperature to 180-260°C to induce decomposition and cyclization, then purify the final product via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers tangible benefits that extend beyond mere chemical performance. The ability to produce high-value solvents through a streamlined, solvent-free process translates directly into improved margin structures and supply security. Traditional solvent manufacturing often involves complex multi-step extractions and extensive solvent recovery units, which represent significant capital expenditure and operational overhead. In contrast, the methodology described in CN1058488C simplifies the production train, reducing the number of unit operations required to reach the final specification. This simplification not only lowers the barrier to entry for manufacturing but also enhances the reliability of supply by minimizing the number of potential failure points in the production line. Furthermore, the versatility of the R-group allows for the tuning of physical properties such as viscosity and boiling point, enabling suppliers to offer customized grades tailored to specific client applications without retooling entire production facilities.
- Cost Reduction in Manufacturing: The most significant economic driver for this process is the elimination of external solvents during the cyclization reaction. By running the reaction neat (solvent-free), manufacturers avoid the costs associated with purchasing, storing, and recovering large volumes of auxiliary solvents. Additionally, the recovery and reuse of unreacted amine intermediates mean that raw material utilization rates are exceptionally high. This circular approach to material usage significantly lowers the variable cost per kilogram of the final product, providing a competitive pricing advantage in the global specialty chemicals market without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as alkylamines and dihaloethanes, are commodity chemicals with well-established global supply chains. Unlike specialized catalysts or rare earth metals that might be subject to geopolitical supply shocks, these feedstocks are readily available from multiple sources. This abundance ensures that production schedules can be maintained consistently, reducing the risk of stockouts for downstream customers. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in raw material quality, further stabilizing the supply chain against upstream variability.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process is inherently cleaner. The absence of auxiliary solvents reduces the facility's VOC (Volatile Organic Compound) emissions profile, simplifying compliance with increasingly stringent environmental regulations. The high boiling point of the final product also reduces evaporation losses during storage and transport. Scalability is facilitated by the fact that the reaction does not require extreme pressures; for many alkyl variants, atmospheric pressure is sufficient, allowing for the use of standard glass-lined or stainless steel reactors. This ease of scale-up from laboratory to multi-tonne production ensures that supply can grow in tandem with market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,3,6-trialkylhexahydro-1,3,6-triazocin-2-one. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this solvent into their existing workflows or product portfolios. The answers reflect the practical realities of scaling this chemistry from benchtop experiments to industrial manufacturing environments.
Q: What are the primary advantages of 1,3,6-trialkylhexahydro-1,3,6-triazocin-2-one over NMP?
A: Unlike N-methyl-2-pyrrolidone (NMP), which is associated with chromosomal aberrations and reproductive toxicity, this novel cyclic urea derivative offers a safer toxicological profile while maintaining excellent solvency for high polymers like polyamides and PVC.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the patent describes a robust solvent-free methodology that eliminates the need for complex solvent recovery systems. The process utilizes standard distillation for purification and allows for the recycling of unreacted amines, making it highly suitable for commercial scale-up.
Q: What represent the key applications for this chemical intermediate?
A: Beyond its primary function as a high-performance aprotic polar solvent for polymer processing and organic synthesis, it serves as a critical intermediate in the manufacture of detergents and surfactants due to its unique amphiphilic structural properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,6-Trialkylhexahydro-1,3,6-triazocin-2-one Supplier
As the demand for safer, high-performance aprotic solvents continues to rise, partnering with an experienced CDMO is essential for navigating the complexities of commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific thermal requirements of this cyclization process, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We understand that transitioning to a new solvent involves validation and testing, and our technical team is prepared to support you through every stage of that journey.
We invite you to engage with our technical procurement team to discuss how this innovative solvent technology can optimize your specific applications. Whether you require a Customized Cost-Saving Analysis for switching from NMP or need detailed route feasibility assessments for new derivative structures, we are ready to provide the data you need. Please contact us today to request specific COA data and explore how our manufacturing capabilities can drive efficiency and value in your supply chain.
