Advanced Manufacturing of Minodronate: A Safe, Scalable, and Cost-Effective Industrial Route
Advanced Manufacturing of Minodronate: A Safe, Scalable, and Cost-Effective Industrial Route
The pharmaceutical industry is constantly seeking robust manufacturing processes that balance high purity with operational safety, particularly for potent active pharmaceutical ingredients (APIs) like Minodronate. Patent CN102020676B introduces a groundbreaking methodology for the preparation of Minodronic acid, a third-generation bisphosphonate used extensively in the treatment of osteoporosis and hypercalcemia associated with malignant tumors. This technical disclosure represents a significant paradigm shift from traditional synthetic routes that rely heavily on hazardous reagents such as sodium cyanide and elemental bromine. By leveraging a streamlined four-step sequence involving condensation, cyclization, hydrolysis, and phosphonation, this innovation offers a pathway that is not only chemically efficient but also environmentally sustainable. For R&D directors and supply chain managers, understanding the nuances of this patent is critical, as it outlines a viable strategy for producing high-purity pharmaceutical intermediates while mitigating the regulatory and safety risks associated with legacy manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Minodronate has been plagued by significant safety and efficiency challenges that hinder large-scale commercial viability. Early methods, such as those disclosed in EP0354806, suffered from low yields and lacked clear sourcing strategies for critical raw materials, making them economically unfeasible for mass production. More concerning are the routes described in literature such as the Chinese Journal of Pharmaceuticals and J. Med. Chem., which utilize sodium cyanide for cyano group substitution. The handling of sodium cyanide requires stringent safety protocols, specialized containment infrastructure, and complex waste treatment systems to neutralize toxic byproducts, all of which drive up operational costs exponentially. Furthermore, alternative pathways like CN101531681 necessitate cryogenic reaction conditions as low as -80°C and the use of volatile, toxic halogenating agents like bromine and carbon tetrachloride. These extreme conditions impose a heavy energy burden on manufacturing facilities and introduce substantial risks regarding worker safety and environmental compliance, rendering such methods unsuitable for modern, green chemistry standards.
The Novel Approach
In stark contrast to these hazardous legacy methods, the process outlined in CN102020676B employs a sophisticated yet practical synthetic strategy that prioritizes safety and controllability. The core innovation lies in the construction of the imidazo[1,2-a]pyridine scaffold through a condensation-cyclization sequence that avoids toxic cyanides entirely. Instead, the process utilizes 2,2-dimethoxyacetaldehyde and triethyl phosphonoacetate in a base-catalyzed condensation, followed by a ring-closure reaction with 2-aminopyridine under mild acidic conditions. This approach not only simplifies the reaction workflow but also operates at moderate temperatures ranging from 20°C to 100°C, eliminating the need for energy-intensive cryogenic cooling. The final phosphonation step is executed using phosphorus trichloride and phosphorous acid in high-boiling solvents like chlorobenzene, ensuring high conversion rates without the generation of hazardous halogenated waste streams typical of bromine-based routes.
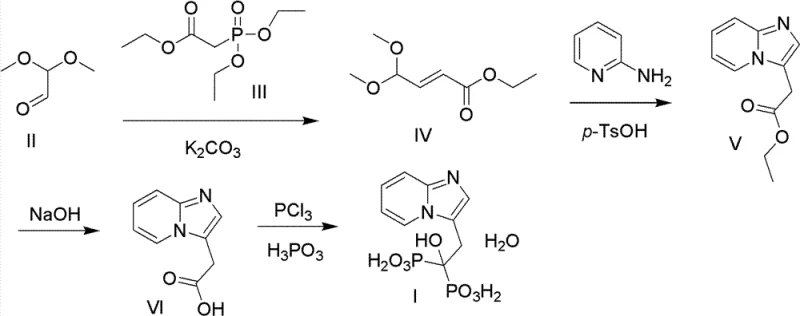
The visual representation of this novel pathway highlights the logical progression from simple starting materials to the complex bisphosphonate structure. By replacing the dangerous cyanide displacement step found in older literature (as seen in comparative diagrams like [图 3]) with a safer hydrolysis and phosphonation sequence, the new method drastically reduces the toxicity profile of the entire manufacturing process. This structural elegance translates directly into operational reliability, allowing for tighter control over impurity profiles and facilitating easier purification of the final API intermediate.
Mechanistic Insights into the Condensation-Cyclization-Phosphonation Sequence
The mechanistic foundation of this synthesis relies on precise control over nucleophilic substitutions and electrophilic aromatic substitutions to build the heterocyclic core. The initial condensation between 2,2-dimethoxyacetaldehyde and triethyl phosphonoacetate is driven by anhydrous potassium carbonate in a non-protonic solvent system such as cyclohexane or n-hexane. This specific choice of base and solvent is critical; the mild inorganic base prevents premature hydrolysis of the acetal group while facilitating the formation of the alpha,beta-unsaturated ester intermediate with high stereoselectivity. Following this, the cyclization step involves the nucleophilic attack of the nitrogen atom in 2-aminopyridine onto the activated double bond of the intermediate. Catalyzed by p-toluenesulfonic acid in a polar acetonitrile-water mixture, this reaction proceeds through a concerted mechanism that efficiently closes the imidazole ring, forming the stable imidazo[1,2-a]pyridine skeleton essential for the biological activity of Minodronate.
Impurity control is inherently built into the reaction conditions specified in the patent. By maintaining the hydrolysis step at temperatures between 70°C and 100°C using sodium hydroxide, the process ensures complete saponification of the ester group without degrading the sensitive heterocyclic ring. The final phosphonation reaction is perhaps the most critical stage for purity, as it introduces the gem-bisphosphonate moiety. Conducted in chlorobenzene at reflux temperatures with phosphorus trichloride and phosphorous acid, this step favors the formation of the desired 1-hydroxy-1,1-bisphosphonate structure while minimizing the formation of mono-phosphonated byproducts or pyrophosphate impurities. The use of concentrated hydrochloric acid in the workup further aids in protonating the phosphonate groups and precipitating the product, allowing for effective removal of inorganic salts and residual organic impurities through simple filtration and recrystallization.
How to Synthesize Minodronate Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and temperature gradients defined in the patent embodiments to ensure optimal yield and purity. The process begins with the preparation of the unsaturated ester, followed by the crucial ring-closure with 2-aminopyridine, which sets the stage for the final functionalization. Detailed operational parameters, including specific solvent volumes and catalyst loading rates, are essential for reproducing the high yields reported in the experimental data. For a comprehensive breakdown of the standardized operating procedures and quality control checkpoints required for this synthesis, please refer to the technical guide below.
- Condense 2,2-dimethoxyacetaldehyde with triethyl phosphonoacetate using potassium carbonate in cyclohexane to form the unsaturated ester intermediate.
- Perform ring-closure reaction with 2-aminopyridine in an acetonitrile-water system catalyzed by p-toluenesulfonic acid to obtain the imidazo[1,2-a]pyridine derivative.
- Hydrolyze the ester intermediate under alkaline conditions using sodium hydroxide to yield the corresponding acetic acid derivative.
- Execute the final phosphonation reaction using phosphorus trichloride and phosphorous acid in chlorobenzene at elevated temperatures to generate Minodronate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this cyanide-free synthesis route offers profound advantages for procurement managers and supply chain directors focused on cost reduction and risk mitigation. The elimination of sodium cyanide and bromine removes the necessity for expensive hazardous material handling licenses, specialized storage facilities, and complex effluent treatment plants required to neutralize toxic waste. This fundamental shift in reagent selection translates into significant operational expenditure savings, as the facility no longer needs to invest in the rigorous safety infrastructure mandated for Class A poisons. Furthermore, the avoidance of cryogenic conditions (-80°C) drastically reduces energy consumption, as the process can be run using standard heating and cooling utilities available in any multipurpose chemical plant, thereby lowering the utility cost per kilogram of produced API.
- Cost Reduction in Manufacturing: The streamlined nature of this four-step process inherently lowers production costs by reducing the number of unit operations and purification stages required. By utilizing readily available and inexpensive starting materials like 2,2-dimethoxyacetaldehyde and triethyl phosphonoacetate, the raw material cost base is significantly optimized compared to routes requiring specialized, low-volume precursors. Additionally, the higher overall yield achieved through mild reaction conditions means less raw material is wasted on side products, directly improving the cost-efficiency of the final Minodronate product without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by relying on commodity chemicals rather than restricted toxic substances. Sodium cyanide is subject to strict government controls and transportation regulations, which can lead to unpredictable delays and supply disruptions. By substituting these with common industrial reagents like potassium carbonate and phosphorus trichloride, manufacturers can secure a more stable and continuous supply of inputs. This reliability ensures consistent production schedules and shorter lead times for delivering high-purity pharmaceutical intermediates to downstream API manufacturers, safeguarding against market volatility.
- Scalability and Environmental Compliance: The environmental footprint of this process is substantially smaller, aligning perfectly with modern green chemistry initiatives and increasingly stringent global environmental regulations. The absence of heavy metal catalysts and persistent organic pollutants simplifies the waste disposal process, reducing the environmental compliance costs associated with hazardous waste incineration. This eco-friendly profile not only facilitates easier regulatory approval for new manufacturing sites but also enhances the brand reputation of the supplier as a responsible partner in the pharmaceutical value chain, making it easier to scale production from pilot batches to multi-ton commercial volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Minodronate synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in patent CN102020676B, offering clarity on how this method outperforms traditional approaches in terms of safety, yield, and scalability. Understanding these details is vital for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines.
Q: Why is the new synthesis route for Minodronate considered safer than previous methods?
A: The novel route described in patent CN102020676B completely eliminates the use of highly toxic sodium cyanide and hazardous bromine reagents found in prior art methods like EP0354806. By replacing these dangerous substances with milder reagents such as potassium carbonate and phosphorus trichloride under controlled conditions, the process significantly reduces operational hazards and environmental compliance burdens.
Q: What are the key reaction conditions for the cyclization step in this process?
A: The cyclization reaction utilizes a polar solvent system, preferably acetonitrile and water, with p-toluenesulfonic acid as the catalyst. The reaction is conducted at moderate temperatures between 70°C and 100°C, which ensures high conversion rates while avoiding the extreme cryogenic conditions (-80°C) required by older synthetic pathways.
Q: How does this method improve scalability for industrial production?
A: This method improves scalability by shortening the reaction steps and utilizing readily available raw materials like 2,2-dimethoxyacetaldehyde. The avoidance of complex purification steps associated with toxic byproducts and the use of standard solvents like chlorobenzene and cyclohexane make the process highly adaptable for large-scale manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Minodronate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN102020676B to meet the evolving demands of the global pharmaceutical market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Minodronate meets the highest international standards for safety and efficacy required for osteoporosis treatments.
We invite potential partners to engage with our technical procurement team to discuss how this innovative, cyanide-free synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your transition to this superior manufacturing process is seamless, compliant, and commercially advantageous.
