Revolutionizing Zoledronic Acid Production: High-Yield Phase Transfer Catalysis for Commercial Scale
Revolutionizing Zoledronic Acid Production: High-Yield Phase Transfer Catalysis for Commercial Scale
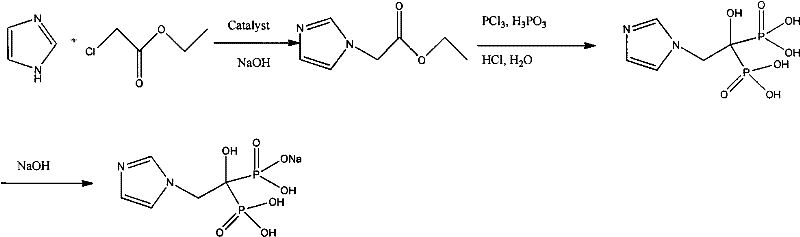
The pharmaceutical industry constantly seeks robust synthetic routes for critical oncology therapeutics, and the production of Zoledronic acid stands as a prime example where process innovation drives market viability. Patent CN102070668A introduces a transformative methodology utilizing a specialized phase transfer catalyst to prepare Zoledronic acid and its sodium salt, addressing long-standing inefficiencies in bisphosphonate manufacturing. This technical breakthrough is particularly significant for global supply chains, as it promises to elevate the total synthesis yield to approximately 60%, a figure that represents a substantial leap forward compared to historical benchmarks. By leveraging quaternary ammonium salts within a refined one-pot synthesis framework, this approach not only accelerates reaction kinetics but also ensures superior product consistency, making it an essential reference for any entity seeking a reliable API intermediate supplier. The implications of this technology extend beyond mere chemistry; they redefine the economic and operational feasibility of producing high-purity pharmaceutical intermediates on a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bisphosphonate medicines like Zoledronic acid has been plagued by significant technical bottlenecks, primarily revolving around the physical properties of the reaction melt and suboptimal conversion rates. Prior art, including domestic patents such as CN1472215A and CN1693308A, attempted to mitigate these issues through stepwise synthesis or the use of specific solvents like diglyme, yet these efforts often resulted in disappointing final yields ranging merely from 20% to 30%. The core challenge lies in the tendency of the reaction mixture to cure or become excessively viscous, creating a heterogeneous system that impedes mass transfer and limits the extent of reaction. Furthermore, traditional methods often require complex multi-step procedures that increase the accumulation of impurities and necessitate rigorous purification protocols, thereby inflating production costs and extending lead times. These inefficiencies create a fragile supply chain where raw material consumption is high relative to output, posing a severe constraint for procurement managers aiming for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast to these legacy processes, the novel approach detailed in the patent employs a strategic one-pot synthesis facilitated by a highly effective phase transfer catalyst, fundamentally altering the reaction landscape. By introducing quaternary ammonium compounds, specifically benzyltriethylammonium chloride (TEBA), into the alkaline reaction medium, the method successfully bridges the interface between organic and aqueous phases, dramatically enhancing the interaction between imidazole and alpha-halogen acetic ester. This catalytic intervention allows the reaction to proceed fully at ambient temperature, eliminating the need for energy-intensive heating cycles while simultaneously preventing the viscosity issues that hinder conventional routes. The result is a streamlined process where the total synthesis yield is improved by about 60%, effectively doubling the output per batch compared to standard techniques. This efficiency gain is not merely a laboratory curiosity but a production innovation that offers profound significance for the industrial scaling of Zoledronic acid, ensuring high product quality and yield with a simplified operational footprint.
Mechanistic Insights into Phase Transfer Catalyzed Alkylation
The efficacy of this synthesis route hinges on the precise mechanistic role of the phase transfer catalyst in facilitating the nucleophilic substitution reaction between imidazole and ethyl chloroacetate. In the absence of a catalyst, the reaction between the solid or poorly soluble imidazole salt and the organic ester is kinetically sluggish due to phase separation. The quaternary ammonium cation acts as a molecular shuttle, transporting the reactive imidazole anion from the aqueous or solid interface into the organic phase where the alkylating agent resides. This transport mechanism drastically reduces the activation energy required for the formation of the intermediate imidazole-ethyl acetate derivative, allowing the reaction to reach completion within a shortened timeframe of approximately two hours under mild stirring conditions. Moreover, the catalyst stabilizes the transition state, minimizing side reactions that typically generate difficult-to-remove impurities, thus contributing to the high purity profile required for clinical-grade active pharmaceutical ingredients.
Following the initial alkylation, the subsequent phosphorylation step involving phosphorous acid and phosphorus trichloride is equally critical and benefits from the homogeneous environment established by the catalyst. The controlled addition of phosphorus trichloride under ambient conditions prevents localized exotherms that could degrade the sensitive imidazole ring, while the presence of the catalyst ensures uniform distribution of reactants throughout the solvent matrix, typically 1,4-dioxane. Upon hydrolysis with water and concentrated hydrochloric acid, the intermediate undergoes cleavage to form the final bisphosphonic acid structure. The choice of methanol as a precipitation agent in the final isolation step exploits the differential solubility of the product, allowing for the efficient removal of inorganic salts and residual organic byproducts. This meticulous control over each mechanistic stage ensures that the final Zoledronic acid meets stringent purity specifications, a key requirement for reducing lead time for high-purity pharmaceutical intermediates in regulated markets.
How to Synthesize Zoledronic Acid Efficiently
The practical implementation of this synthesis route requires strict adherence to stoichiometric ratios and operational parameters to maximize the benefits of the phase transfer catalysis system. The process begins with the dissolution of imidazole in a suitable solvent like 1,4-dioxane, followed by the addition of sodium hydroxide to generate the reactive nucleophile in situ. The critical addition of the phase transfer catalyst, optimally at a mass ratio of 0.15 to 0.5 relative to imidazole, precedes the dropwise introduction of ethyl chloroacetate, ensuring a controlled exotherm and complete conversion. Subsequent steps involve the careful management of phosphorus reagents and acidic hydrolysis, culminating in a crystallization process that leverages methanol to isolate the product as a high-quality solid. For detailed operational specifics, safety protocols, and exact reagent quantities, please refer to the standardized synthesis guide below.
- Dissolve imidazole in 1,4-dioxane solvent under alkaline conditions using sodium hydroxide, then add the phase transfer catalyst (e.g., TEBA) and ethyl chloroacetate to initiate the alkylation reaction.
- Introduce phosphorous acid and phosphorus trichloride to the reaction mixture to form the phosphonate intermediate, followed by hydrolysis with water and concentrated hydrochloric acid.
- Filter the reaction mixture, concentrate the filtrate under reduced pressure, and induce crystallization by adding methanol to isolate high-purity Zoledronic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phase transfer catalyzed synthesis offers compelling strategic advantages that directly impact the bottom line and operational resilience. The most immediate benefit is the drastic improvement in atomic economy and raw material utilization; by doubling the reaction yield from typical levels of 20-30% to approximately 60%, the process effectively halves the quantity of starting materials required to produce a single kilogram of finished API intermediate. This reduction in material intensity translates directly into significant cost savings, as fewer tons of imidazole, esters, and phosphorus reagents need to be sourced, transported, and processed. Furthermore, the one-pot nature of the reaction eliminates the need for intermediate isolation and purification steps, which are often the most labor-intensive and waste-generating stages of chemical manufacturing. This simplification not only lowers utility consumption but also reduces the volume of hazardous waste requiring disposal, aligning with increasingly strict environmental compliance standards.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction of solvent usage through the one-pot strategy significantly lower the variable costs associated with production. By avoiding complex multi-step sequences, manufacturers can reduce labor hours and equipment occupancy time, leading to a more lean and cost-effective manufacturing model. The high yield ensures that fixed costs are amortized over a larger volume of product, further driving down the unit cost of goods sold and enhancing margin potential for downstream drug developers.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route, which operates effectively at ambient temperature and tolerates standard industrial reagents, minimizes the risk of batch failures due to thermal runaway or sensitivity issues. This reliability ensures consistent output volumes, allowing supply chain planners to forecast inventory levels with greater accuracy and reduce the need for safety stock buffers. Additionally, the use of widely available phase transfer catalysts like TEBA mitigates the risk of supply disruptions associated with exotic or proprietary reagents, securing the continuity of supply for critical oncology medications.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial application prospects in mind, allowing for seamless transition from pilot plant to multi-ton commercial production without significant re-engineering. The simplified workup procedure, which relies on filtration and crystallization rather than complex chromatography, generates less solvent waste and facilitates easier recycling of mother liquors. This green chemistry approach not only reduces the environmental footprint but also streamlines regulatory approvals by presenting a cleaner impurity profile and a more sustainable manufacturing lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phase transfer catalyzed synthesis for Zoledronic acid. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is crucial for R&D teams evaluating process transfer and for procurement specialists assessing vendor capabilities. The answers reflect the consensus on best practices for maximizing yield and purity while maintaining operational safety and efficiency in a GMP environment.
Q: What is the primary advantage of using a phase transfer catalyst in Zoledronic acid synthesis?
A: The use of a phase transfer catalyst, specifically quaternary ammonium salts like TEBA, significantly accelerates the reaction rate and improves the total synthesis yield to approximately 60%, effectively doubling the output compared to conventional methods which typically achieve only 20-30% yield.
Q: How does this method address the viscosity issues common in bisphosphonate synthesis?
A: By employing a one-pot synthesis strategy with optimized solvent systems like 1,4-dioxane and specific phase transfer agents, the method mitigates the curing and high-viscosity problems often encountered in traditional bisphosphonate reactions, ensuring smoother processing and easier product isolation.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's good industrial application prospect due to its one-pot nature, high product quality, and substantial yield improvement, making it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Zoledronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible commercial reality through expert process engineering and rigorous quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high-yield potential of the phase transfer catalyzed route is fully realized in our manufacturing facilities. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that monitor every batch for impurities, ensuring that the Zoledronic acid we supply meets the exacting standards required for global pharmaceutical registration. We understand that consistency is key in the supply of oncology intermediates, and our advanced infrastructure is designed to deliver this reliability without compromise.
We invite forward-thinking pharmaceutical companies and generic drug manufacturers to collaborate with us to leverage this advanced synthesis technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this high-yield process for your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your product quality and your bottom line. Let us be your trusted partner in navigating the complexities of modern API manufacturing.
