Advanced Biocatalytic Synthesis of R-Mandelic Acid for Scalable Pharmaceutical Manufacturing
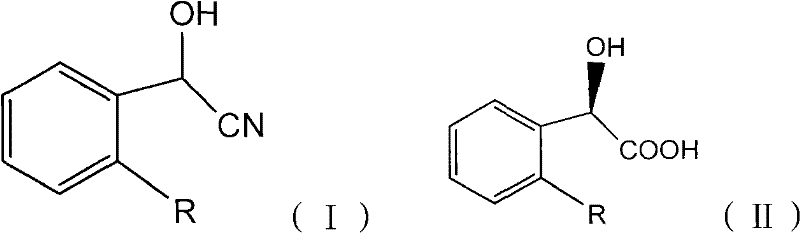
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for producing chiral intermediates, and patent CN101701243B presents a groundbreaking solution for the synthesis of R-mandelic acid and its derivatives. This technology leverages a highly specific mutant strain, Alcaligenes faecalis CCTCC No: M 208168, to catalyze the enantioselective hydrolysis of racemic mandelonitrile. Unlike traditional chemical methods that struggle with toxicity and low stereoselectivity, this biocatalytic approach operates under mild physiological conditions, achieving an enantiomeric excess (ee) of greater than 99%. For R&D directors and procurement managers alike, this represents a significant shift towards greener chemistry that does not compromise on purity or yield. The ability to produce high-value chiral acids like R-o-chloromandelic acid with such precision opens new avenues for the synthesis of beta-lactam antibiotics and other critical therapeutics.
Furthermore, the robustness of the Alcaligenes faecalis strain ensures consistent performance across multiple fermentation batches, addressing a common pain point in biomanufacturing regarding supply continuity. The patent details a comprehensive optimization of the fermentation medium, utilizing cost-effective components like ammonium acetate and yeast extract, which directly translates to lower raw material costs. By integrating this biocatalytic step into existing production lines, manufacturers can drastically simplify their downstream processing, eliminating the need for complex chiral chromatography or toxic resolving agents. This report analyzes the technical merits and commercial viability of this patented process, positioning it as a cornerstone for reliable pharmaceutical intermediate supplier strategies in the modern market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active R-mandelic acid has been plagued by significant technical and economic hurdles inherent to conventional chemical synthesis and resolution techniques. Asymmetric synthesis methods, such as those employing Jacobsen catalysts, often suffer from prohibitively high costs due to the expense of chiral ligands and the difficulty in recovering these precious metal complexes for reuse. Moreover, the enantiomeric purity achieved through these chemical routes frequently ranges between 65% and 85% ee, necessitating additional, costly purification steps to meet the stringent requirements of pharmaceutical grade materials. Alternatively, optical resolution of racemic mandelic acid via diastereomeric salt formation is limited by a theoretical maximum yield of 50%, effectively wasting half of the starting material and generating substantial amounts of chemical waste. These traditional processes also typically require harsh reaction conditions, including extreme temperatures and organic solvents, which pose safety risks and increase the environmental footprint of the manufacturing facility.
The Novel Approach
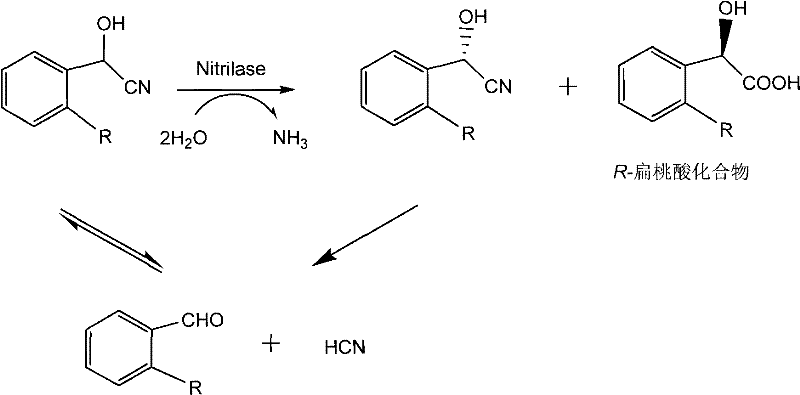
In stark contrast, the biocatalytic method disclosed in the patent utilizes a specialized nitrilase enzyme to selectively hydrolyze the R-enantiomer of mandelonitrile, bypassing the inefficiencies of chemical resolution. This enzymatic pathway operates in an aqueous environment at a neutral to slightly alkaline pH of 8.0 to 8.5, eliminating the need for volatile organic compounds and reducing the hazard profile of the reaction. The use of whole cells containing the nitrilase simplifies the catalyst preparation process, as it avoids the complex and yield-reducing steps of enzyme purification. As illustrated in the reaction scheme below, the substrate is converted directly into the desired chiral acid with ammonia as the only byproduct, streamlining the atom economy of the process.
This novel approach not only enhances the stereochemical purity to over 99% ee but also offers superior scalability. The patent highlights that the mutant strain exhibits significantly higher specific activity compared to wild-type strains reported in prior art, allowing for higher substrate loading and faster reaction kinetics. For a reliable pharmaceutical intermediate supplier, this means the ability to meet large-volume demands without the bottleneck of low-yield chemical steps. The transition from chemical to biological catalysis here represents a paradigm shift, enabling cost reduction in pharmaceutical intermediate manufacturing through improved efficiency and reduced waste disposal costs.
Mechanistic Insights into Nitrilase-Catalyzed Hydrolysis
The core of this technology lies in the unique catalytic mechanism of the nitrilase enzyme derived from Alcaligenes faecalis CCTCC No: M 208168. Nitrilases function by hydrolyzing the carbon-nitrogen triple bond of the nitrile group directly to the corresponding carboxylic acid and ammonia, without forming an amide intermediate. This direct hydrolysis is crucial for preventing the accumulation of mandelonitrile amide, which could complicate downstream purification. The enzyme exhibits strict stereoselectivity, recognizing and binding only the R-configured mandelonitrile within the racemic mixture. This selectivity is governed by the precise spatial arrangement of amino acid residues in the enzyme's active site, which creates a chiral environment that sterically hinders the S-enantiomer from undergoing hydrolysis. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters such as pH and temperature to maintain enzyme stability and activity.
Additionally, the reaction system must account for the chemical equilibrium between mandelonitrile and its decomposition products, benzaldehyde and hydrogen cyanide, as shown in the mechanistic diagram. Under the controlled pH conditions of 8.0 to 8.5, the equilibrium is shifted to favor the nitrile form, ensuring sufficient substrate availability for the enzyme. However, if the pH deviates significantly, the spontaneous decomposition of mandelonitrile can occur, leading to the loss of substrate and the generation of toxic cyanide species. The patented process mitigates this risk by maintaining a tight pH control window and utilizing a buffer system that stabilizes the substrate. This careful management of reaction chemistry ensures that the impurity profile remains clean, with minimal formation of benzaldehyde or S-mandelic acid, thereby facilitating easier isolation of the final high-purity pharmaceutical intermediate.
How to Synthesize R-Mandelic Acid Efficiently
Implementing this biocatalytic route requires a disciplined approach to fermentation and bioconversion to maximize the potential of the Alcaligenes faecalis strain. The process begins with the cultivation of the microorganism in a specifically optimized medium designed to induce high levels of nitrilase expression. Key to this step is the addition of inducers such as n-butyronitrile or caprolactam during the early growth phase, which triggers the metabolic pathways responsible for enzyme production. Following fermentation, the biomass is harvested and suspended in a phosphate buffer, creating a ready-to-use biocatalyst suspension. The subsequent hydrolysis reaction is then conducted by adding the racemic mandelonitrile substrate, where the temperature and pH are strictly monitored to ensure optimal enzyme performance. Detailed standardized synthesis steps see the guide below.
- Cultivate Alcaligenes faecalis CCTCC No: M 208168 in optimized fermentation medium containing ammonium acetate and n-butyronitrile inducer at 30°C for 20-24 hours to maximize nitrilase activity.
- Prepare the reaction system by suspending the harvested enzyme-containing cells in a phosphate buffer solution adjusted to pH 8.0-8.5.
- Add racemic mandelonitrile substrate to the buffer at a concentration of 10-50 mmol/L and maintain the temperature between 30-40°C to achieve high conversion rates and optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic technology offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials; instead of sourcing expensive chiral catalysts or resolving agents, manufacturers can rely on fermentation-derived biocatalysts produced from inexpensive carbon and nitrogen sources. This shift reduces dependency on volatile chemical markets and stabilizes the cost structure of the final product. Furthermore, the mild reaction conditions eliminate the need for specialized corrosion-resistant reactors required for strong acid or base hydrolysis, allowing for the utilization of standard stainless steel equipment. This compatibility with existing infrastructure accelerates the timeline for technology transfer and commercial scale-up.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and toxic organic solvents results in substantial cost savings throughout the production lifecycle. By avoiding the use of heavy metals, the downstream purification process is significantly streamlined, as there is no need for expensive metal scavenging steps or rigorous testing for residual metal contaminants. The high conversion rate and specific activity of the enzyme mean that less biomass is required per unit of product, reducing the overall consumption of fermentation resources. Additionally, the ability to operate at ambient pressure and moderate temperatures lowers energy consumption for heating and cooling, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of the Alcaligenes faecalis strain ensures consistent production quality, minimizing the risk of batch failures that can disrupt supply schedules. Since the biocatalyst is produced via fermentation, scaling up production capacity is a matter of increasing fermenter volume rather than sourcing scarce chemical reagents. This scalability provides a buffer against market fluctuations and ensures a steady supply of high-purity pharmaceutical intermediates even during periods of high demand. The genetic stability of the mutant strain further guarantees that the process performance remains constant over long-term production runs, securing the continuity of supply for downstream drug manufacturers.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns perfectly with green chemistry principles, generating significantly less hazardous waste compared to traditional chemical synthesis. The aqueous nature of the reaction reduces the volume of organic solvent waste that requires treatment or incineration, lowering disposal costs and environmental liability. The byproduct of the reaction is ammonia, which can be easily managed or utilized, unlike the complex salt wastes generated during diastereomeric resolution. This eco-friendly profile not only aids in meeting regulatory compliance standards but also enhances the corporate sustainability image, a factor increasingly valued by global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic process. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational parameters and expected outcomes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolio. The responses highlight the specific advantages of the mutant strain and the optimized reaction conditions that define the success of this method.
Q: What are the advantages of using Alcaligenes faecalis CCTCC No: M 208168 over traditional chemical synthesis?
A: Unlike traditional asymmetric synthesis which often requires expensive chiral catalysts and yields moderate enantiomeric excess (65-85% ee), this biocatalytic method utilizes a highly active mutant strain that achieves >99% ee under mild aqueous conditions, significantly reducing heavy metal contamination risks and purification costs.
Q: How does this process address the theoretical 50% yield limit of kinetic resolution?
A: While kinetic resolution theoretically limits yield to 50% of the racemate, the high specific activity of the nitrilase (up to 997 U/L) ensures rapid and complete conversion of the R-enantiomer, minimizing side reactions and allowing for efficient recycling of the unreacted S-enantiomer or its conversion via racemization strategies in downstream processing.
Q: Is this biocatalytic method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability in 15L fermenters with robust biomass growth (2.64 g/L) and stable enzyme production. The use of whole cells eliminates the need for expensive enzyme purification, and the mild reaction conditions (20-60°C, pH 8.0-8.5) reduce energy consumption and equipment corrosion, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-Mandelic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in the production of high-value chiral intermediates like R-mandelic acid. As a leading CDMO partner, we possess the technical expertise to adapt and optimize such advanced biological routes for commercial production. Our facilities are equipped to handle the commercial scale-up of complex pharmaceutical intermediates, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the highest international standards for optical purity and chemical identity. Our commitment to quality ensures that the transition from laboratory scale to industrial manufacturing is seamless and reliable.
We invite potential partners to engage with our technical procurement team to discuss how this biocatalytic technology can be tailored to your specific supply chain needs. By collaborating with us, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this enzymatic process. We encourage you to request specific COA data and route feasibility assessments to verify the suitability of our R-mandelic acid for your synthetic applications. Together, we can drive innovation and efficiency in the pharmaceutical supply chain, ensuring the timely delivery of critical intermediates for life-saving medications.
