Advanced Biocatalytic Production of R-Mandelic Acid for Global Pharmaceutical Supply Chains
Advanced Biocatalytic Production of R-Mandelic Acid for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable methods for producing chiral intermediates, and patent CN101709323B presents a transformative approach to synthesizing R-mandelic acid. This intellectual property details a sophisticated coupling method that integrates biocatalysis with in-situ separation, specifically utilizing nitrilase enzymes alongside strong basic anion exchange resins. By addressing the critical bottleneck of product inhibition that plagues traditional enzymatic processes, this technology enables the efficient conversion of racemic mandelonitrile into high-purity R-mandelic acid. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate suppliers, this patent outlines a pathway that not only enhances reaction kinetics but also simplifies downstream processing, marking a significant leap forward in the manufacturing of optically active acids used in cephalosporins, penicillins, and anti-obesity drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of optically pure R-mandelic acid has relied heavily on chemical resolution of racemates or asymmetric synthesis, both of which suffer from inherent inefficiencies and economic drawbacks. Chemical resolution methods, while established, are fundamentally limited by a maximum theoretical yield of 50%, meaning half of the valuable starting material is discarded or requires complex recycling streams, driving up raw material costs and waste generation. Furthermore, asymmetric synthesis routes often necessitate the use of expensive transition metal catalysts, such as Jacobsen catalysts, which pose significant challenges regarding metal residue removal and regulatory compliance in pharmaceutical applications. These conventional pathways frequently involve harsh reaction conditions, multiple synthetic steps, and generate substantial environmental pollution, making them increasingly unattractive for modern green chemistry mandates and cost-sensitive supply chains.
The Novel Approach
In stark contrast, the biocatalytic coupling method described in the patent leverages the stereoselectivity of nitrilase derived from strains like Alcaligenes faecalis ZJUTB10 to achieve theoretical conversion rates approaching 100% from racemic substrates. The core innovation lies in the simultaneous addition of strong basic anion exchange resins, such as the HZ202 type, directly into the reaction vessel during the hydrolysis phase. This strategic integration allows for the immediate adsorption of the generated R-mandelic acid onto the resin matrix, effectively pulling the reaction equilibrium forward and shielding the biocatalyst from the inhibitory effects of the accumulating acid product. This approach not only drastically accelerates the reaction rate but also facilitates a simplified work-up procedure where the product is concentrated on the solid phase, ready for elution and crystallization, thereby streamlining the entire manufacturing workflow.
Mechanistic Insights into Nitrilase-Catalyzed Hydrolysis and Resin Coupling
The biochemical foundation of this process rests on the highly specific activity of nitrilase enzymes, which catalyze the direct hydrolysis of the nitrile group in mandelonitrile to the corresponding carboxylic acid without forming an amide intermediate. The patent elucidates that the enzyme exhibits a profound preference for the R-enantiomer, converting racemic mandelonitrile into R-mandelic acid while leaving the S-enantiomer largely untouched or allowing it to racemize under the reaction conditions, thus maximizing atom economy. The reaction is conducted in a buffered aqueous system maintained at a pH between 8.0 and 8.5 and temperatures ranging from 20°C to 60°C, conditions that are mild enough to preserve enzyme integrity yet vigorous enough to ensure rapid turnover. The presence of the anion exchange resin introduces a physicochemical dimension to the catalysis, where the quaternary ammonium functional groups on the resin interact electrostatically with the carboxylate anions of the product.
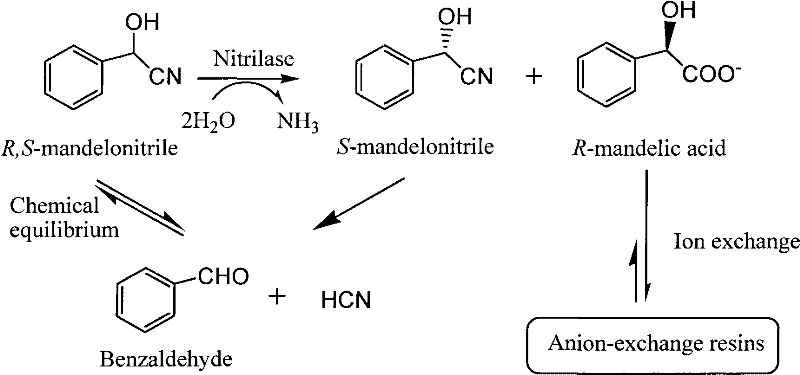
To fully appreciate the efficacy of the separation coupling, one must examine the structure of the resin itself, which acts as a solid-phase scavenger. The patent specifies the use of strong basic anion exchange resins containing either trimethylamine groups (Type I) or dimethyl-β-hydroxy-ethylamine groups (Type II), as illustrated in the chemical structures provided. These functional groups provide the necessary basicity and steric environment to selectively bind the R-mandelic acid anions as they are formed. By sequestering the product onto the solid resin beads, the concentration of free acid in the liquid phase remains low, preventing the feedback inhibition that typically deactivates nitrilase enzymes in batch processes. This mechanism allows for the repeated feeding of substrate (racemic mandelonitrile) into the same reaction vessel, enabling a semi-continuous or fed-batch operation that significantly boosts the volumetric productivity of the biocatalyst.
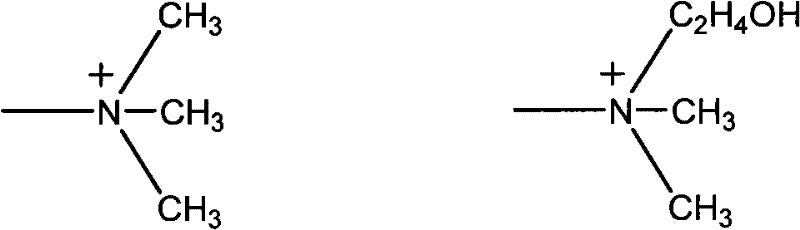
How to Synthesize R-Mandelic Acid Efficiently
Implementing this technology requires precise control over the fermentation of the biocatalyst and the conditioning of the ion exchange resin to ensure optimal performance. The process begins with the cultivation of the Alcaligenes faecalis strain in a defined fermentation medium supplemented with inducers like n-butyronitrile to maximize enzyme expression. Following cell harvest, the resin must be rigorously pre-treated with acid and base cycles to remove impurities and convert it to the appropriate ionic form before being introduced to the bioconversion reactor. The detailed standardized synthesis steps, including specific concentrations, timing for resin addition, and elution protocols, are critical for reproducing the high yields and optical purities reported in the patent documentation.
- Cultivate Alcaligenes faecalis ZJUTB10 to obtain nitrilase-containing cells.
- Prepare strong basic anion exchange resin (e.g., HZ202) by washing and conditioning with acid and base.
- Conduct hydrolysis of racemic mandelonitrile at pH 8.0-8.5 and 20-60°C with simultaneous resin addition.
- Elute the resin with HCl solution to recover R-mandelic acid and regenerate the resin for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic coupling technology translates into tangible strategic advantages regarding cost stability and operational reliability. By eliminating the need for expensive chiral metal catalysts and avoiding the 50% yield ceiling of classical resolution, the process inherently lowers the cost of goods sold (COGS) through superior raw material utilization. The ability to regenerate and reuse the anion exchange resin multiple times further diminishes consumable costs, while the mild aqueous reaction conditions reduce energy consumption associated with heating, cooling, and solvent recovery compared to traditional organic synthesis routes. This efficiency creates a more resilient supply chain capable of meeting high-volume demands for R-mandelic acid without the volatility associated with precious metal markets or complex waste disposal regulations.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for costly and technically demanding heavy metal clearance steps, which are a significant expense in API intermediate manufacturing. Furthermore, the in-situ adsorption mechanism allows for higher substrate loading and reduced reactor volumes to achieve the same output, effectively increasing asset utilization and lowering capital expenditure per unit of production. The qualitative improvement in process efficiency means that manufacturers can achieve substantial cost savings through reduced utility usage and simplified downstream purification trains.
- Enhanced Supply Chain Reliability: Utilizing a robust microbial strain like Alcaligenes faecalis ZJUTB10 ensures a consistent and renewable source of catalytic activity, mitigating the risks associated with the supply of synthetic chiral ligands. The fed-batch capability demonstrated in the patent allows for flexible production scheduling, where substrate can be added as needed to match demand, preventing bottlenecks and ensuring continuous availability of the intermediate. This operational flexibility is crucial for maintaining uninterrupted supply lines to downstream pharmaceutical customers who require just-in-time delivery of critical chiral building blocks.
- Scalability and Environmental Compliance: The process operates primarily in aqueous buffers, significantly reducing the volume of hazardous organic solvents required, which aligns with increasingly stringent global environmental regulations and sustainability goals. The simplicity of the work-up, involving filtration of the resin followed by acid elution and crystallization, is inherently easier to scale from pilot to commercial tonnage than multi-step organic syntheses. This scalability ensures that the technology can support large-scale commercial production of complex pharmaceutical intermediates without compromising on purity or environmental safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic process, derived directly from the experimental data and claims within the patent specification. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing frameworks. The answers reflect the specific capabilities of the nitrilase-resin coupling system as validated by the patent holder's research.
Q: How does the resin coupling technology improve catalytic efficiency?
A: The strong basic anion exchange resin adsorbs the produced R-mandelic acid in situ, effectively removing product inhibition on the nitrilase enzyme and allowing for higher cumulative yields and repeated catalyst usage.
Q: What is the optical purity achievable with this method?
A: Experimental data from the patent indicates that the optical purity (ee value) of the resulting R-mandelic acid consistently exceeds 99% when using optimized resin types like HZ202.
Q: Can the anion exchange resin be reused?
A: Yes, the resin can be regenerated after elution using sodium hydroxide solution, washed to neutrality, and reused in subsequent batches, significantly reducing material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-Mandelic Acid Supplier
As the global demand for chiral pharmaceutical intermediates continues to rise, partnering with a CDMO expert who understands the intricacies of advanced biocatalysis is paramount for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards, providing our partners with the confidence needed to accelerate their drug development pipelines.
We invite you to engage with our technical procurement team to discuss how this innovative biocatalytic technology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient manufacturing route. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial viability.
