Advanced Manufacturing of 2-Cyclopenten-1-One: A Strategic Upgrade for Global Supply Chains
Advanced Manufacturing of 2-Cyclopenten-1-One: A Strategic Upgrade for Global Supply Chains
The global demand for high-purity cyclic enones as versatile building blocks in organic synthesis continues to surge, driven by the expanding pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is detailed in patent CN1289452C, which discloses a novel and industrially advantageous method for producing 2-cyclopenten-1-one. This specific compound serves as a critical precursor for various functional materials and active pharmaceutical ingredients, making its efficient manufacture a priority for supply chain stability. The patented process addresses long-standing inefficiencies in dehydrohalogenation reactions by introducing a specialized solvent system that dramatically simplifies downstream processing. By leveraging a combination of high-boiling amide solvents and optional aromatic co-solvents, the technology ensures that the target molecule can be isolated with exceptional purity while enabling the regeneration of valuable reaction media. For R&D directors and procurement strategists, understanding the nuances of this methodology is essential for evaluating potential suppliers who can offer both technical excellence and economic viability in the competitive landscape of fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
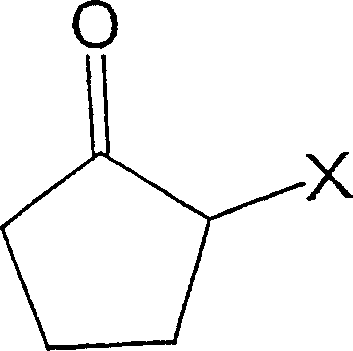
Historically, the synthesis of 2-cyclopenten-1-one from 2-halocyclopentanone precursors has been plagued by significant technical hurdles that impede scalable manufacturing. Traditional protocols often relied on heating the halogenated substrate in solvents like N,N-diethylaniline, a method notorious for delivering disappointingly low yields and failing to produce the target ketone in commercially viable quantities. Another common approach utilized N,N-dimethylformamide (DMF) as the reaction medium; however, this presented a severe thermodynamic challenge during the workup phase. Since the boiling point of DMF is dangerously close to that of 2-cyclopenten-1-one, separating the product from the solvent via standard distillation became an energy-intensive and technically difficult operation, often resulting in product degradation or contamination. Furthermore, industrial implementations of these older methods struggled with the accumulation of halide salts within the solvent matrix, making solvent recycling nearly impossible and driving up waste disposal costs substantially. The reliance on expensive N-alkylformanilide compounds in some prior art further exacerbated the economic burden, rendering these processes unattractive for large-scale commercial adoption where margin compression is a constant threat.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally reengineers the reaction environment to overcome these separation and efficiency bottlenecks. By shifting to an amide-based solvent system with a specifically defined boiling point range of 180 to 240°C, such as N-methylpyrrolidone (NMP), the process creates a substantial thermal gap between the solvent and the product. This physical property difference is the cornerstone of the new method's success, allowing for the straightforward fractional distillation of 2-cyclopenten-1-one under reduced pressure without dragging over significant amounts of solvent. The introduction of an aromatic co-solvent with a boiling point below 145°C, such as toluene, adds another layer of process optimization by facilitating the removal of inorganic halide byproducts through simple aqueous washing, as the aromatic layer is immiscible with water. This dual-solvent strategy not only enhances the reaction kinetics but also establishes a closed-loop system where both the high-boiling amide and the aromatic components can be recovered and reused multiple times. Consequently, this approach transforms a previously wasteful and difficult purification sequence into a streamlined, continuous operation that aligns perfectly with modern green chemistry principles and cost-reduction mandates.
Mechanistic Insights into Base-Promoted Dehydrohalogenation
At the heart of this synthesis lies a classic yet optimized dehydrohalogenation mechanism, where the removal of a hydrogen halide molecule from the 2-halocyclopentanone ring generates the desired alpha,beta-unsaturated ketone functionality. The reaction proceeds through an elimination pathway, likely following an E2 mechanism, where a base abstracts the acidic alpha-proton adjacent to the carbonyl group while the halide leaving group departs simultaneously. The presence of a lithium salt catalyst, such as lithium bromide or lithium chloride, plays a subtle but critical role in this transformation by coordinating with the carbonyl oxygen, thereby increasing the acidity of the alpha-proton and stabilizing the transition state. This Lewis acid activation lowers the activation energy required for the elimination step, allowing the reaction to proceed efficiently at moderate temperatures ranging from 90 to 110°C. The choice of base is equally pivotal, with carbonates like potassium carbonate or lithium carbonate serving as effective proton scavengers that neutralize the generated hydrogen halide without inducing unwanted side reactions such as polymerization or ring opening. The synergy between the lithium catalyst and the carbonate base ensures high conversion rates while maintaining the structural integrity of the sensitive cyclopentenone ring throughout the reaction duration.
Beyond the primary transformation, the mechanistic design of this process inherently controls the impurity profile, which is a paramount concern for pharmaceutical intermediate manufacturers. The use of mild reaction conditions prevents the thermal degradation of the product, which is prone to polymerization at elevated temperatures, a common issue in conventional high-heat methods. Furthermore, the specific solvent polarity provided by the amide system helps to solubilize the organic intermediates while keeping the inorganic halide salts precipitated or separable in the aqueous wash phase. This phase separation capability is crucial for minimizing the carryover of halide ions into the final product, which could otherwise catalyze decomposition during storage or interfere with downstream coupling reactions. The addition of polymerization inhibitors like hydroquinone, as noted in the experimental examples, provides an additional safety net against radical-mediated oligomerization, ensuring that the final distillate consists predominantly of the monomeric 2-cyclopenten-1-one. This rigorous control over the reaction environment translates directly into a cleaner crude product, reducing the load on final purification columns and enhancing the overall throughput of the manufacturing facility.
How to Synthesize 2-Cyclopenten-1-One Efficiently
The practical execution of this synthesis requires precise control over stoichiometry and thermal parameters to maximize yield and solvent recovery. The process begins with the preparation of a reaction vessel charged with the high-boiling amide solvent, the optional aromatic co-solvent, the lithium catalyst, and the carbonate base, all heated to the optimal reaction temperature before the substrate is introduced.
- Prepare a reaction system containing a high-boiling amide solvent (180-240°C) and optionally an aromatic co-solvent, along with a base and lithium salt catalyst.
- Heat the mixture to 90-110°C and slowly add 2-halocyclopentanone to initiate the dehydrohalogenation reaction while neutralizing generated acid.
- Upon completion, recover the aromatic solvent via vacuum concentration, then isolate the pure 2-cyclopenten-1-one product through reduced pressure distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend far beyond simple chemical yield metrics. The most significant advantage lies in the dramatic reduction of operating expenses associated with solvent consumption and waste management. Unlike traditional processes where solvents are often discarded after a single use due to salt contamination, this novel system allows for the extensive regeneration of high-value amide solvents through vacuum concentration. This capability effectively decouples production volume from raw material procurement costs, providing a hedge against market volatility in solvent pricing. Moreover, the simplified workup procedure eliminates the need for complex extraction trains or energy-intensive azeotropic distillations, resulting in shorter batch cycle times and increased asset utilization rates for manufacturing plants. These operational efficiencies collectively contribute to a more resilient supply chain capable of meeting tight delivery schedules without compromising on product quality or regulatory compliance.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the elimination of expensive, single-use solvent systems and the reduction of energy loads during purification. By utilizing a solvent pair where the components have distinct boiling points and solubility profiles, the process avoids the costly entrapment of product in mother liquors that plagues DMF-based methods. The ability to recycle the bulk of the reaction medium means that the variable cost per kilogram of product decreases significantly as production scale increases. Furthermore, the avoidance of transition metal catalysts in favor of inexpensive lithium salts removes the necessity for expensive heavy metal scavenging steps, which are often required to meet strict pharmaceutical impurity limits. This streamlining of the downstream processing train results in substantial cost savings that can be passed down to the customer or reinvested into capacity expansion.
- Enhanced Supply Chain Reliability: From a logistics and sourcing perspective, the raw materials required for this synthesis are commodity chemicals with robust global availability, mitigating the risk of supply disruptions. The reliance on standard bases like potassium carbonate and widely available solvents like toluene and NMP ensures that production is not bottlenecked by exotic reagents with long lead times. Additionally, the robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process tolerates minor variations in feedstock quality without catastrophic failure. This reliability is critical for maintaining continuous supply to downstream API manufacturers who operate on just-in-time inventory models. The simplified purification also reduces the likelihood of batch failures due to off-specification purity, ensuring a consistent flow of high-quality intermediate to the market.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions and waste disposal intensify, this process offers a clear pathway to sustainable manufacturing. The closed-loop solvent recovery system minimizes the volume of hazardous waste generated per unit of product, aligning with increasingly stringent environmental protection laws. The absence of heavy metal catalysts simplifies the effluent treatment process, reducing the burden on wastewater treatment facilities and lowering compliance costs. Furthermore, the scalability of the dehydrohalogenation reaction is proven, as the heat transfer and mixing requirements are manageable even in large-scale reactors. This makes the technology suitable for rapid scale-up from pilot plant to multi-ton commercial production, allowing suppliers to respond quickly to surges in market demand without requiring massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-cyclopenten-1-one, derived directly from the proprietary data and process capabilities.
Q: Why is the new solvent system superior to traditional DMF methods?
A: Traditional methods using N,N-dimethylformamide (DMF) suffer from difficult separation because the solvent's boiling point is too close to the product. The new method utilizes high-boiling amide solvents (180-240°C) which create a significant boiling point gap, allowing for easy fractional distillation and high-purity isolation without complex extraction steps.
Q: How does this process improve cost efficiency for large-scale production?
A: The process enables the regeneration and reuse of expensive amide solvents through simple vacuum concentration. Additionally, the inclusion of low-boiling aromatic solvents like toluene facilitates easier removal of halide salts via aqueous washing, reducing waste disposal costs and raw material consumption significantly.
Q: What represents the critical quality control parameter in this synthesis?
A: Maintaining strict temperature control between 90°C and 110°C is crucial to balance reaction kinetics with product stability. Furthermore, the efficient removal of halide byproducts ensures the final 2-cyclopenten-1-one meets stringent purity specifications required for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyclopenten-1-One Supplier
As the pharmaceutical and fine chemical industries evolve, the need for partners who can bridge the gap between innovative patent technology and commercial reality has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing the technical expertise to implement complex synthetic routes like the dehydrohalogenation process described in CN1289452C with precision and consistency. Our facility is equipped with state-of-the-art reactor systems capable of handling the specific thermal and vacuum requirements of this synthesis, ensuring that every batch meets the rigorous standards expected by global innovators. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, demonstrating our ability to support your projects from early-stage development through to full-scale market launch. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every shipment, guaranteeing that our 2-cyclopenten-1-one performs flawlessly in your downstream applications.
We invite you to engage with our technical team to explore how our optimized manufacturing capabilities can drive value for your organization. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how our efficient solvent recovery and streamlined processing can reduce your total cost of ownership. We encourage potential clients to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your unique project requirements. Whether you require small quantities for R&D validation or metric tons for commercial manufacturing, NINGBO INNO PHARMCHEM is prepared to deliver reliable supply, technical support, and the highest standards of quality assurance to ensure your success in the competitive global marketplace.
